
|
시장보고서
상품코드
1872634
조제 약국 시장 : 제품별, 조제 유형별, 치료 용도별, 무균성별, 최종 사용자별 예측(-2030년)Compounding Pharmacy Market by Product (Oral, Topical, Parenteral), Compounding Type (Ingredient Alteration, Dosage Alteration), Therapeutic Application (Pain Management, HRT), Sterility (Sterile), End User (Pediatric, Adult) - Global forecast to 2030 |
||||||
세계의 조제 약국 시장 규모는 2025년 추정 147억 2,000만 달러에서 2030년까지 194억 1,000만 달러에 이를 것으로 예측되며, 2025-2030년에 CAGR로 5.7%를 나타낼 것으로 전망됩니다.
시장의 성장은 주로 개인화된 약물의 동향 증가에 의해 견인됩니다.
| 조사 범위 | |
|---|---|
| 조사 대상 기간 | 2022-2030년 |
| 기준 연도 | 2024년 |
| 예측 기간 | 2025-2030년 |
| 단위 | 100만/10억 달러 |
| 부문 | 제품, 조제 유형, 치료 용도, 무균성, 최종 사용자 |
| 대상 지역 | 북미, 유럽, 아시아태평양, 중동, 아프리카, 라틴아메리카 |
그러나 API, 첨가제 공급 및 CCI/시험에서의 처리 능력의 제약은 시장 성장을 제한할 것으로 예측됩니다.

제품 유형별로 경구 약물은 조제 약국 시장에서 가장 큰 점유율을 차지했습니다.
경구 약물 부문은 개별 환자의 요구에 맞게 개인화 치료의 필요성이 증가함에 따라 세계 시장에서 중요한 지위를 차지합니다. 이 부문은 캡슐, 정제, 로젠지, 용액, 현탁액과 같은 다양한 투약 형태를 포함하며 호르몬 대체 요법, 소아 및 노인 관리, 통증 관리, 소화기 치료, 만성 질환 관리 등에서 널리 사용됩니다. 제형 경구 약물은 약사가 용량 강도, 풍미, 첨가제 및 방출 프로파일을 사용자 정의할 수 있게 하며, 특히 표준 정제를 삼키는 데 어려움이 있는 환자와 정밀한 투여가 필요한 환자에서 복약 규정 준수를 향상시키는 데 기여합니다. 심혈관 질환, 당뇨병, 내분비 장애와 같은 만성 질환 증가는 효능을 극대화하고 부작용을 줄이는 개인화된 경구 치료에 대한 수요를 만들어 이 부문의 성장을 더욱 촉진하고 있습니다. 서방 시스템, 맛 마스킹 기술, 생체이용률 향상 캐리어 등의 기술 혁신은 치료 효과와 환자 경험을 향상시킵니다. FDA 규정과 USP 기준을 준수하면 안전성, 무균성 및 품질을 보장하고 의료 제공업체와 환자 모두에게 신뢰를 구축합니다. 맞춤형 의료에 대한 주목 증가와, 병원 및 전문 클리닉·소매 약국을 통한 이용 가능성의 향상도 있어, 경구약은 수익 성장의 큰 성장 촉진요인으로 계속하고 있습니다. 편의성, 맞춤화 및 효과적인 치료에 중점을 둔 이 부문은 세계의 조제 약국 시장 전체의 발전에 매우 중요한 역할을 합니다.
통증 관리 부문이 2024년 치료 용도 부문 중 가장 큰 점유율을 차지했습니다.
근골격계, 신경병증, 수술 후 환자군에서 급성 및 만성 통증의 세계 유행으로 통증 관리 부문이 치료 용도별 제약 시장에서 가장 큰 점유율을 차지했습니다. 관절염, 요통, 암 관련 통증, 편두통의 증례가 증가함에 따라 비스테로이드성 항염증제(NSAIDs), 오피오이드, 외용 진통제, 복합요법을 포함한 효과적인 통증완화요법에 대한 수요가 높아지고 있습니다. 서방형 제제 및 표적화 전달 시스템과 같은 개인화된 통증 관리 옵션의 사용 확대는 전신 부작용을 줄이면서 효능을 향상시키고 환자의 복약 준수율과 시장 성장을 더욱 촉진하고 있습니다. 경피 패치 및 국소 주사 요법과 같은 기술적 진보는 치료 옵션을 확장하여 환자의 장기 통증 관리를 더욱 편리하게 만들었습니다. 유리한 보험 적용 범위, 건강상의 중요 과제로서 통증에 대한 인식 향상, 노년 인구의 확대가이 부문의 지속적인 성장을 지원합니다. 주요 제약 기업은 신규 진통제, 생물학적 제제, 비오피오이드 대체약의 연구개발을 통해 제품 포트폴리오를 강화하고 보다 큰 시장 점유율 획득을 목표로 하고 있습니다. 안전하고 효과적이며 환자 중심의 통증 관리 솔루션에 대한 수요가 지속적으로 높아지는 가운데 이 치료 부문은 선진국과 신흥국 시장 모두의 지속적인 혁신과 지속적인 수익 증가를 반영하여 주도적 지위를 유지할 것으로 예측됩니다.
북미가 2025-2030년에 세계의 조제 약국 시장에서 가장 큰 시장 점유율을 차지합니다.
미국의 주도로 북미는 세계의 조제 약국 시장에서 계속 주도적인 지위를 유지하고 있습니다. 이 지역의 이점은 견고한 의료 인프라, 엄격한 규제 기준 및 503A/503B 조제 약국의 광범위한 존재와 밀접한 관련이 있습니다. 이러한 약국은 특히 상시된 약품이 불충분한 경우에, 개별화된 약제를 제공하는 데 있어서 필수적인 역할을 담당하고 있습니다. 미국 시장에서는 다양한 치료 요건을 충족시키기 위해 경구용 약물, 외용제, 무균 제제와 같은 광범위한 제제 제품이 제공됩니다. 최근의 동향으로서, 특히 통증 관리, 호르몬 보충 요법, 소아 케어에 있어서 전문적인 제약 수요가 높아지고 있습니다. FDA와 같은 규제 당국은 의약품의 안전성과 효능을 지속적으로 모니터링하고 보장하며 의료 시스템 내에서 신뢰를 구축하기 위해 노력하고 있습니다. 이러한 요인이 결합되어 조제 약국 서비스에 대한 접근의 확대와 향상에 있어서 북미의 중요한 역할이 명시되어 있습니다.
본 보고서에서는 세계의 조제 약국 시장에 대해 조사 분석하여 주요 성장 촉진요인과 억제요인, 경쟁 구도, 미래 동향 등의 정보를 제공합니다.
자주 묻는 질문
목차
제1장 서론
제2장 조사 방법
제3장 주요 요약
제4장 주요 인사이트
- 조제 약국 시장 개요
- 북미의 조제 약국 시장 : 제품별, 국가별(2025년)
- 조제 약국 시장 : 지리적 성장 기회
- 조제 약국 시장 : 신흥 시장과 선진 시장
- 연결된 시장과 부문 간 기회
- 새로운 비즈니스 모델과 생태계의 변화
- VC/프라이빗 에퀴티 투자의 동향과 스타트업의 현상
- 규제 정책의 대처
제5장 시장 개요
- 서론
- 시장 역학
- 성장 촉진요인
- 성장 억제요인
- 기회
- 과제
- Porter's Five Forces 분석
- 주요 이해관계자와 구매 기준
- 거시경제지표
- 의료비 동향
- 세계의 암 부하
- 의약품 연구개발의 영향
- 밸류체인 분석
- 생태계 분석
- 가격 설정 분석
- 조제 약국 제품의 참고 가격 분석 : 주요 기업별(2024년)
- 조제 약국 제품의 가격 분석(2024년) : 지역별
- 주요 컨퍼런스 및 이벤트(2025-2026년)
- 고객사업에 영향을 주는 동향/혼란
- 투자 및 자금조달 시나리오
- 기술 분석
- 주요 기술
- 보완 기술
- 인접 기술
- 조제 약국 시장에 대한 AI/생성형 AI의 영향
- 규제 분석
- 지역별 규제기관, 정부기관, 기타 조직
- 규제 프레임워크
- 조제 약국 시장에 대한 미국 관세의 영향(2025년)
- 서론
- 주요 관세율
- 가격의 영향 분석
- 국가/지역에 미치는 영향
- 제조에 미치는 영향
- 미충족 수요(Unmet Needs)와 백스페이스
제6장 조제 약국 시장 : 제품별
- 서론
- 경구용 의약품
- 국소용 의약품
- 비경구 약물
- 안과용 의약품
- 비강용 의약품
- 직장 투여 약물
- 기타 제품
제7장 조제 약국 시장 : 조제 유형별
- 서론
- 의약품 용량 변경
- 의약품 성분 변경
- 현재 이용할 수 없는 의약품 제조
제8장 조제 약국 시장 : 치료 용도별
- 서론
- 통증 관리
- 호르몬 대체 요법
- 피부 질환
- 영양 보충제
- 기타 치료 용도
제9장 조제 약국 시장 : 무균성별
- 서론
- 멸균
- 비멸균
제10장 조제 약국 시장 : 최종 사용자별
- 서론
- 성인
- 노인
- 소아
제11장 조제 약국 시장 : 지역별
- 서론
- 북미
- 북미의 거시경제 전망
- 미국
- 캐나다
- 유럽
- 유럽의 거시 경제 전망
- 독일
- 영국
- 프랑스
- 이탈리아
- 스페인
- 기타 유럽
- 아시아태평양
- 아시아태평양의 거시 경제 전망
- 중국
- 일본
- 인도
- 호주
- 한국
- 기타 아시아태평양
- 라틴아메리카
- 라틴아메리카의 거시 경제 전망
- 브라질
- 멕시코
- 기타 라틴아메리카
- 중동 및 아프리카
- 중동 및 아프리카의 거시 경제 전망
- GCC 국가
- 기타 중동 및 아프리카
제12장 경쟁 구도
- 서론
- 주요 진입기업의 전략/강점
- 수익 분석(2022-2024년)
- 시장 점유율 분석(2024년)
- 기업 평가 매트릭스 : 주요 기업(2024년)
- 기업 평가 매트릭스 : 스타트업/중소기업(2024년)
- 기업 평가 및 재무 지표
- 브랜드/제품 비교
- 경쟁 시나리오
제13장 기업 프로파일
- 주요 기업
- BAXTER
- EMPOWER PHARMACY
- B. BRAUN SE
- FAGRON
- QUVA PHARMA, INC.
- SCA PHARMA
- NEPHRON PHARMACEUTICALS
- LEITERS HEALTH
- ICON GROUP
- ROSEWAY LABS
- ITH PHARMA
- PENCOL COMPOUNDING PHARMACY
- FORMUL8
- AURORA COMPOUNDING PHARMACY
- 기타 기업
- CUSTOM MED APOTHECARY
- INSTITUTIONAL PHARMACY SOLUTIONS
- FUSION APOTHECARY
- INNOVATION COMPOUNDING PHARMACY
- RX3 COMPOUNDING PHARMACY
- DOUGHERTY'S PHARMACY, INC.
- LORRAINE'S PHARMACY
- WELLS PHARMA
- OLYMPIA PHARMACY
- FRESH THERAPEUTICS
- MEDIOS AG
제14장 부록
KTH 25.11.27The global compounding pharmacy market is projected to reach USD 19.41 billion by 2030 from an estimated USD 14.72 billion in 2025, at a CAGR of 5.7% from 2025 to 2030. The market's growth has mainly been driven by the increasing trend toward personalized doses.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2022-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD) Million/Billion |
| Segments | by Product (Oral, Topical, Parenteral), Compounding Type (Ingredient Alteration, Dosage Alteration), Therapeutic Application (Pain Management, HRT), Sterility (Sterile), End User (Pediatric, Adult) |
| Regions covered | North America, Europe, Asia Pacific, Middle East & Africa, and Latin America |
However, capacity constraints in API, excipient supply, and CCI/testing are expected to limit the market.
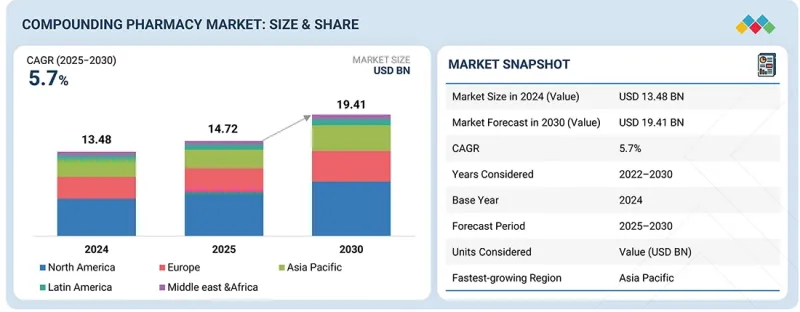
The oral drugs held the highest share in the compounding pharmacy market by product type.
The compounding pharmacy market is segmented by product type into topical drugs, oral drugs, parenteral drugs, rectal drugs, nasal drugs, ophthalmic drugs, and other products. Among these, the oral drugs segment holds a key position in the global market, driven by the increasing need for personalized therapies tailored to individual patient requirements. This segment includes a wide variety of formulations such as capsules, tablets, lozenges, solutions, and suspensions, which are extensively used in hormone replacement therapy, pediatric and geriatric care, pain management, gastrointestinal treatments, and chronic disease management. Compounded oral medications enable pharmacists to customize dosage strengths, flavors, excipients, and release profiles, thereby improving patient compliance-especially among those who have difficulty swallowing standard tablets or who need precise dosing. The rising prevalence of chronic conditions like cardiovascular diseases, diabetes, and endocrine disorders further fuels this segment's growth by creating demand for personalized oral treatments that maximize efficacy and reduce side effects. Technological innovations, including sustained-release systems, taste-masking techniques, and bioavailability-enhancing carriers, improve therapeutic performance and patient experience. Adherence to FDA regulations and USP standards ensures safety, sterility, and quality, building confidence among healthcare providers and patients. Along with the growing emphasis on personalized medicine and increased availability through hospitals, specialty clinics, and retail pharmacies, oral drugs continue to be a major driver of revenue growth. The segment's focus on convenience, customization, and effective therapy highlights its vital role in the overall development of the global compounding pharmacy market.
The pain management segment reported the highest share of the therapeutic application segment in 2024
The pain management segment holds the largest share in the pharmaceutical market by therapeutic application, driven by the global prevalence of acute and chronic pain conditions across musculoskeletal, neuropathic, and post-surgical patient groups. Increasing cases of arthritis, back pain, cancer-related pain, and migraines have boosted demand for effective pain relief therapies, including NSAIDs, opioids, topical analgesics, and combination treatments. The rising use of personalized pain management options, such as sustained-release formulations and targeted delivery systems, improves effectiveness while reducing systemic side effects, further increasing patient adherence and market growth. Technological advances, like transdermal patches and localized injectable therapies, have broadened treatment choices and made long-term pain management more convenient for patients. Favorable insurance coverage, greater awareness of pain as a key health issue, and the expanding elderly population support consistent growth in this segment. Leading pharmaceutical firms are enhancing their portfolios through research into new analgesics, biologics, and non-opioid alternatives, positioning themselves to capture larger market shares. As the need for safe, effective, and patient-focused pain management solutions continues to grow, this therapeutic segment is expected to stay dominant, reflecting sustained revenue increases and ongoing innovation in both developed and emerging markets.
North America accounted for the largest market share in the global Compounding pharmacy market from 2025 to 2030.
North America remains the leading region in the global compounding pharmacy market, mainly due to the United States. The region's dominance is linked to its strong healthcare infrastructure, strict regulatory standards, and a large presence of both 503A and 503B compounding pharmacies. These pharmacies are essential in providing personalized medications, especially where commercially available drugs are insufficient. The US market offers a wide variety of compounded products, including oral, topical, and sterile preparations, to meet different therapeutic needs. Recent trends show a rising demand for specialized compounded medications, particularly in pain management, hormone replacement therapy, and pediatric care. Regulatory agencies like the FDA continue to monitor and ensure the safety and effectiveness of compounded drugs, building trust within the healthcare system. This combination of factors highlights North America's key role in advancing and improving access to compounding pharmacy services.
The primary interviews conducted for this report can be categorized as follows:
- By Respondent: Supply Side-70% and Demand Side-30%
- By Designation: Managers-45%, CXO and Directors-30%, and Executives-25%
- By Region: North America-40%, Europe-25%, the Asia Pacific-25%, Latin America-5%, and the Middle East & Africa-5%
List of Key Companies Profiled in the Report:
Key players in the Compounding pharmacy market include Baxter (US), Empower Pharmacy (US), B. Braun (Germany), Fagron (Netherlands), QuVa Pharma (US), SCA Pharma (US), Nephron Pharmaceuticals (US), Leiters Health (US), Pharmaxo (UK), Icon Group (Australia), Roseway Labs (UK), ITH Pharma (UK), Pencol Compounding Pharmacy (Canada), Formula 8 (South Africa), and Aurora Compounding Pharmacy (Canada)
Research Coverage:
This research report categorizes the compounding pharmacy market by product (oral, topical, parenteral), compounding type (ingredient alteration, dosage alteration), therapeutic application (pain management, HRT), sterility (sterile), and end user (pediatric, adult). The scope of the report includes detailed information about the main factors affecting the growth of the Compounding pharmacy market, such as drivers, restraints, challenges, and opportunities. It also features an in-depth analysis of key industry players, offering insights into their business overview, products, solutions, strategies, collaborations, partnerships, and agreements. The report covers recent developments like new approvals, launches, collaborations, acquisitions, and other updates related to the Compounding pharmacy market.
Key Benefits of Buying the Report:
The report will assist market leaders and new entrants by providing them with the closest estimates of revenue figures for the entire Compounding pharmacy market and its subsegments. It will also help stakeholders better understand the competitive landscape and gain more insights to position their businesses effectively and develop appropriate go-to-market strategies. This report will enable stakeholders to grasp the market's key indicators and offer information on the main market drivers, restraints, opportunities, and challenges.
The report provides insights into the following pointers:
- Analysis of key drivers (Persistent drug shortages in North America and Europe, full enforcement of USP standards, safety push for ready-to-administer and standardized doses, continued demand creating supply gap for GLP-1 drugs, and trend toward personalization of doses), restraints (Resolution of GLP-1 shortages reduces reliance on compounded alternatives, stricter FDA oversight and insanitary conditions enforcement, and limitations on bulk drug substances.), opportunities (Expansion of hospital outsourcing into anesthesia, critical care, ophthalmic & chemo-adjuncts and Radiopharmaceutical compounding standardization), and challenges (Capacity bottlenecks in API, excipient supply, and CCI/testing, and limited quality-system maturity and gaps in stability/BUD evidence generation.) influencing the growth of the market.
- Service Development/Innovation: Detailed insights on upcoming technologies, research & development activities in the compounding pharmacy market
- Market Development: Comprehensive information about lucrative markets - the report analyses the market across varied regions.
- Market Diversification: Exhaustive information about untapped geographies, recent developments, and investments in the compounding pharmacy market
- Competitive Assessment: In-depth assessment of market shares, growth strategies, and product offerings of leading players. A detailed analysis of the key industry players has been done to provide insights into their key strategies, product launches/ approvals, pipeline analysis, acquisitions, partnerships, agreements, collaborations, other recent developments, investment and funding activities, brand/product comparative analysis, and vendor valuation and financial metrics of the Compounding pharmacy market.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.3.4 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
2 RESEARCH METHODOLOGY
- 2.1 RESEARCH DATA
- 2.1.1 SECONDARY DATA
- 2.1.1.1 Objectives of secondary research
- 2.1.1.2 Key data from secondary sources
- 2.1.2 PRIMARY DATA
- 2.1.2.1 Breakdown of primaries (supply- and demand-side participants)
- 2.1.2.2 Key objectives of primary research
- 2.1.1 SECONDARY DATA
- 2.2 MARKET SIZE ESTIMATION
- 2.2.1 GLOBAL MARKET ESTIMATION
- 2.2.1.1 Company revenue analysis (bottom-up approach)
- 2.2.1.2 Revenue share analysis
- 2.2.1.3 MnM repository analysis
- 2.2.1.4 Primary interviews
- 2.2.2 INSIGHTS OF PRIMARY EXPERTS
- 2.2.3 SEGMENTAL MARKET SIZE ESTIMATION (TOP-DOWN APPROACH)
- 2.2.1 GLOBAL MARKET ESTIMATION
- 2.3 MARKET GROWTH RATE PROJECTIONS
- 2.4 DATA TRIANGULATION
- 2.5 STUDY ASSUMPTIONS
- 2.6 RESEARCH LIMITATIONS
- 2.7 RISK ANALYSIS
3 EXECUTIVE SUMMARY
- 3.1 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
- 3.2 STRATEGIC IMPERATIVES FOR KEY STAKEHOLDERS
- 3.2.1 STARTUPS AND INNOVATIVE COMPANIES
- 3.2.2 ESTABLISHED MARKET LEADERS
4 PREMIUM INSIGHTS
- 4.1 COMPOUNDING PHARMACY MARKET OVERVIEW
- 4.2 NORTH AMERICA: COMPOUNDING PHARMACY MARKET, BY PRODUCT AND COUNTRY (2025)
- 4.3 COMPOUNDING PHARMACY MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- 4.4 COMPOUNDING PHARMACY MARKET: EMERGING VS. DEVELOPED MARKETS
- 4.5 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.6 EMERGING BUSINESS MODELS & ECOSYSTEM SHIFTS
- 4.7 VC/PRIVATE EQUITY INVESTMENT TRENDS & STARTUP LANDSCAPE
- 4.8 REGULATORY POLICY INITIATIVES
5 MARKET OVERVIEW
- 5.1 INTRODUCTION
- 5.2 MARKET DYNAMICS
- 5.2.1 DRIVERS
- 5.2.1.1 Persistent drug shortages in North America and Europe
- 5.2.1.2 Enforcement of USP standards
- 5.2.1.3 Push for ready-to-administer (RTA) and standardized doses
- 5.2.1.4 Continued demand-supply gap for GLP-1 drugs
- 5.2.1.5 Trend towards personalization of doses
- 5.2.2 RESTRAINTS
- 5.2.2.1 Resolution of GLP-1 shortages and reduced reliance on compounded alternatives
- 5.2.2.2 Stricter FDA oversight and intensified enforcement of unsanitary conditions violations
- 5.2.2.3 Limitations on bulk drug substances
- 5.2.3 OPPORTUNITIES
- 5.2.3.1 Expansion of hospital outsourcing into anesthesia, critical care, ophthalmic, and chemo-adjuncts
- 5.2.3.2 Radiopharmaceutical compounding standardization
- 5.2.4 CHALLENGES
- 5.2.4.1 Capacity bottlenecks in API, excipient supply, and critical compounding infrastructure testing
- 5.2.4.2 Limited quality-system maturity and gaps in stability/BUD evidence generation
- 5.2.1 DRIVERS
- 5.3 PORTER'S FIVE FORCES ANALYSIS
- 5.3.1 THREAT OF NEW ENTRANTS
- 5.3.2 THREAT OF SUBSTITUTES
- 5.3.3 BARGAINING POWER OF SUPPLIERS
- 5.3.4 BARGAINING POWER OF BUYERS
- 5.3.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.4 KEY STAKEHOLDERS & BUYING CRITERIA
- 5.4.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 5.4.2 KEY BUYING CRITERIA
- 5.5 MACROECONOMICS INDICATORS
- 5.5.1 HEALTHCARE EXPENDITURE TRENDS
- 5.5.2 GLOBAL CANCER BURDEN
- 5.5.3 IMPACT OF PHARMA R&D
- 5.6 VALUE CHAIN ANALYSIS
- 5.7 ECOSYSTEM ANALYSIS
- 5.7.1 ROLE IN ECOSYSTEM
- 5.8 PRICING ANALYSIS
- 5.8.1 INDICATIVE PRICING ANALYSIS OF COMPOUNDING PHARMACY PRODUCTS, BY KEY PLAYER, 2024
- 5.8.2 INDICATIVE PRICING ANAYLSIS OF COMPOUNDING PHARMACY PRODUCTS, BY REGION, 2024
- 5.9 KEY CONFERENCES & EVENTS, 2025-2026
- 5.10 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- 5.11 INVESTMENT & FUNDING SCENARIO
- 5.12 TECHNOLOGY ANALYSIS
- 5.12.1 KEY TECHNOLOGIES
- 5.12.1.1 Automated compounding devices
- 5.12.1.2 Sterile compounding equipment
- 5.12.2 COMPLEMENTARY TECHNOLOGIES
- 5.12.2.1 Quality control & testing systems
- 5.12.2.2 Pharmacy information management software (PIMS)
- 5.12.3 ADJACENT TECHNOLOGIES
- 5.12.3.1 3D printing of pharmaceuticals
- 5.12.3.2 Personalized medicine platforms
- 5.12.1 KEY TECHNOLOGIES
- 5.13 IMPACT OF AI/GEN AI ON COMPOUNDING PHARMACY MARKET
- 5.14 REGULATORY ANALYSIS
- 5.14.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS, BY REGION
- 5.14.2 REGULATORY FRAMEWORK
- 5.15 IMPACT OF 2025 US TARIFF ON COMPOUNDING PHARMACY MARKET
- 5.15.1 INTRODUCTION
- 5.15.2 KEY TARIFF RATES
- 5.15.3 PRICE IMPACT ANALYSIS
- 5.15.4 IMPACT ON COUNTRY/REGION
- 5.15.4.1 US
- 5.15.4.2 Europe
- 5.15.4.3 Asia Pacific
- 5.15.4.4 Rest of the World
- 5.15.5 IMPACT ON MANUFACTURING INDUSTRIES
- 5.16 UNMET NEEDS & WHITE SPACES
6 COMPOUNDING PHARMACY MARKET, BY PRODUCT
- 6.1 INTRODUCTION
- 6.2 ORAL DRUGS
- 6.2.1 INCREASING DEMAND FOR PRECISE AND CUSTOMIZABLE ORAL FORMULATIONS IN UNMET PATIENT POPULATIONS TO DRIVE MARKET
- 6.3 TOPICAL DRUGS
- 6.3.1 RISING DEMAND FOR PERSONALIZED TOPICAL TREATMENTS AND ADVANCED DELIVERY VEHICLES TO FUEL MARKET GROWTH
- 6.4 PARENTERAL DRUGS
- 6.4.1 RISING DEMAND FOR STERILE INJECTABLE THERAPIES AND SUPPLY-CHAIN DISRUPTIONS TO BOOST MARKET GROWTH
- 6.5 OPTHALMIC DRUGS
- 6.5.1 RISING CASES OF EYE-RELATED DISORDERS TO ACCELERATE MARKET GROWTH
- 6.6 NASAL DRUGS
- 6.6.1 ADVANCEMENTS IN NASAL DRUG DELIVERY SYSTEMS AND FAVORABLE REGULATORY POLICIES TO PROPEL MARKET GROWTH
- 6.7 RECTAL DRUGS
- 6.7.1 PERSONALIZED FORMULATIONS AND REGULATORY SUPPORT TO DRIVE GROWTH IN RECTAL COMPOUNDING THERAPIES
- 6.8 OTHER PRODUCTS
7 COMPOUNDING PHARMACY MARKET, BY COMPOUNDING TYPE
- 7.1 INTRODUCTION
- 7.2 PHARMACEUTICAL DOSAGE ALTERATION
- 7.2.1 TAILORED DOSAGE FORMS TO ENHANCE PATIENT ADHERENCE AND CUSTOMIZATION IN COMPOUNDED MEDICATIONS
- 7.3 PHARMACEUTICAL INGREDIENTS ALTERATION
- 7.3.1 INCREASED NEED FOR CUSTOMIZED PHARMACEUTICAL INGREDIENTS TO ENHANCE PATIENT-CENTRIC COMPOUNDING SOLUTIONS
- 7.4 CURRENTLY UNAVAILABLE PHARMACEUTICAL MANUFACTURING
- 7.4.1 PERSISTENT GLOBAL DRUG SHORTAGES AND LIMITATIONS OF LARGE-SCALE PHARMACEUTICAL PRODUCTION TO PROPEL ADOPTION
8 COMPOUNDING PHARMACY MARKET, BY THERAPEUTIC APPLICATION
- 8.1 INTRODUCTION
- 8.2 PAIN MANAGEMENT
- 8.2.1 NEED FOR PERSONALIZED PAIN RELIEF SOLUTIONS FOR CHRONIC AILMENTS TO SUPPORT MARKET
- 8.3 HORMONE REPLACEMENT THERAPIES
- 8.3.1 POPULARITY OF PERSONALIZED HORMONE THERAPIES TO DRIVE STEADY GROWTH IN GLOBAL COMPOUNDING PHARMACY MARKET
- 8.4 DERMAL DISORDERS
- 8.4.1 CUSTOMIZED DERMATOLOGICAL SOLUTIONS TO PROPEL GLOBAL COMPOUNDING PHARMACY MARKET EXPANSION
- 8.5 NUTRITIONAL SUPPLEMENTS
- 8.5.1 PERSONALIZED NUTRACEUTICALS TO OFFER NICHE BUT BETTER GROWTH OPPORTUNITIES IN COMPOUNDING PHARMACIES
- 8.6 OTHER THERAPEUTIC APPLICATIONS
9 COMPOUNDING PHARMACY MARKET, BY STERILITY
- 9.1 INTRODUCTION
- 9.2 STERILE
- 9.2.1 RTU/RTA STERILE PREPARATIONS AND OPHTHALMIC PACKS TO DRIVE RAPID GROWTH IN COMPOUNDING PHARMACIES
- 9.3 NON-STERILE
- 9.3.1 503A/MAGISTRAL NON-STERILE PREPARATIONS TO DRIVE GLOBAL VOLUME AND VALUE LEADERSHIP IN COMPOUNDING PHARMACIES
10 COMPOUNDING PHARMACY MARKET, BY END USER
- 10.1 INTRODUCTION
- 10.2 ADULTS
- 10.2.1 RISING CHRONIC DISEASE BURDEN AND PERSONALIZED THERAPIES TO AUGMENT ADULT SEGMENT GROWTH
- 10.3 GERIATRIC PATIENTS
- 10.3.1 AGING DEMOGRAPHICS AND DOSE-FORM CUSTOMIZATION TO PROPEL GERIATRIC SEGMENT GROWTH
- 10.4 PEDIATRIC PATIENTS
- 10.4.1 CUSTOMIZED DOSAGE FORMS AND PEDIATRIC-SPECIFIC NEEDS TO DRIVE GROWTH
11 COMPOUNDING PHARMACY MARKET, BY REGION
- 11.1 INTRODUCTION
- 11.2 NORTH AMERICA
- 11.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 11.2.2 US
- 11.2.2.1 US to dominate North American compounding pharmacy market during forecast period
- 11.2.3 CANADA
- 11.2.3.1 Personalized treatments, regulatory reinforcement, and supply-gap filling to fuel global compounding pharmacy growth
- 11.3 EUROPE
- 11.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 11.3.2 GERMANY
- 11.3.2.1 Aging demographics, technological advancements, and supportive regulations to drive market
- 11.3.3 UK
- 11.3.3.1 Personalized treatments, chronic disease management, and regulatory support to support market growth
- 11.3.4 FRANCE
- 11.3.4.1 Focus on chronic disease management to fuel growth in French compounding pharmacy market
- 11.3.5 ITALY
- 11.3.5.1 Rising commercial drug development pipeline to spur market growth
- 11.3.6 SPAIN
- 11.3.6.1 Global regulatory advancements and rising R&D activities to drive market
- 11.3.7 REST OF EUROPE
- 11.4 ASIA PACIFIC
- 11.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 11.4.2 CHINA
- 11.4.2.1 Low manufacturing cost and high demand for medicines to favor market growth
- 11.4.3 JAPAN
- 11.4.3.1 Increasing prevalence of chronic diseases and growing geriatric population to fuel demand for customized medications
- 11.4.4 INDIA
- 11.4.4.1 Increasing pharma R&D activities to support market growth
- 11.4.5 AUSTRALIA
- 11.4.5.1 Innovation in clinical practice and regulatory support to accelerate compounding pharmacy growth
- 11.4.6 SOUTH KOREA
- 11.4.6.1 Regulatory innovation and automation to fuel South Korean compounding pharmacy market growth
- 11.4.7 REST OF ASIA PACIFIC
- 11.5 LATIN AMERICA
- 11.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 11.5.2 BRAZIL
- 11.5.2.1 Growing pharmacy & biologics manufacturing sectors and rising R&D sepnding to drive market
- 11.5.3 MEXICO
- 11.5.3.1 Regulatory improvements, high cancer rates, and robust pharmaceutical industry to boost market growth
- 11.5.4 REST OF LATIN AMERICA
- 11.6 MIDDLE EAST & AFRICA
- 11.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 11.6.2 GCC COUNTRIES
- 11.6.2.1 Rising demand for biologics and increasing prevalence of non-communicable diseases to propel market growth
- 11.6.3 REST OF MIDDLE EAST & AFRICA
12 COMPETITIVE LANDSCAPE
- 12.1 INTRODUCTION
- 12.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 12.2.1 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN COMPOUNDING PHARMACY MARKET, 2023-2025
- 12.3 REVENUE ANALYSIS, 2022-2024
- 12.4 MARKET SHARE ANALYSIS, 2024
- 12.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2024
- 12.5.1 STARS
- 12.5.2 EMERGING LEADERS
- 12.5.3 PERVASIVE PLAYERS
- 12.5.4 PARTICIPANTS
- 12.5.5 COMPETITIVE BENCHMARKING: KEY PLAYERS, 2024
- 12.5.5.1 Company footprint
- 12.5.5.2 Region footprint
- 12.5.5.3 Product footprint
- 12.5.5.4 Therapeutic application footprint
- 12.5.5.5 Sterility footprint
- 12.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2024
- 12.6.1 PROGRESSIVE COMPANIES
- 12.6.2 RESPONSIVE COMPANIES
- 12.6.3 DYNAMIC COMPANIES
- 12.6.4 STARTING BLOCKS
- 12.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2024
- 12.6.5.1 Detailed list of key startup/SMEs
- 12.6.5.2 Competitive benchmarking of key startups/SMEs
- 12.7 COMPANY VALUATION & FINANCIAL METRICS
- 12.7.1 FINANCIAL METRICS
- 12.7.2 COMPANY VALUATION
- 12.8 BRAND/PRODUCT COMPARISON
- 12.9 COMPETITIVE SCENARIO
- 12.9.1 PRODUCT LAUNCHES
- 12.9.2 DEALS
- 12.9.3 EXPANSIONS
13 COMPANY PROFILES
- 13.1 KEY PLAYERS
- 13.1.1 BAXTER
- 13.1.1.1 Business overview
- 13.1.1.2 Products offered
- 13.1.1.3 Recent developments
- 13.1.1.3.1 Product launches
- 13.1.1.4 MnM view
- 13.1.1.4.1 Key strengths
- 13.1.1.4.2 Strategic choices
- 13.1.1.4.3 Weaknesses & competitive threats
- 13.1.2 EMPOWER PHARMACY
- 13.1.2.1 Business overview
- 13.1.2.2 Products offered
- 13.1.2.3 Recent developments
- 13.1.2.3.1 Expansions
- 13.1.2.4 MnM view
- 13.1.2.4.1 Key strengths
- 13.1.2.4.2 Strategic choices
- 13.1.2.4.3 Weaknesses & competitive threats
- 13.1.3 B. BRAUN SE
- 13.1.3.1 Business overview
- 13.1.3.2 Products offered
- 13.1.3.3 Recent developments
- 13.1.3.3.1 Product launches
- 13.1.3.4 MnM view
- 13.1.3.4.1 Key strengths
- 13.1.3.4.2 Strategic choices
- 13.1.3.4.3 Weaknesses & competitive threats
- 13.1.4 FAGRON
- 13.1.4.1 Business overview
- 13.1.4.2 Products offered
- 13.1.4.3 Recent developments
- 13.1.4.3.1 Deals
- 13.1.4.4 MnM view
- 13.1.4.4.1 Key strengths
- 13.1.4.4.2 Strategic choices
- 13.1.4.4.3 Weaknesses & competitive threats
- 13.1.5 QUVA PHARMA, INC.
- 13.1.5.1 Business overview
- 13.1.5.2 Products offered
- 13.1.5.3 MnM view
- 13.1.5.3.1 Key strengths
- 13.1.5.3.2 Strategic choices
- 13.1.5.3.3 Weaknesses & competitive threats
- 13.1.6 SCA PHARMA
- 13.1.6.1 Business overview
- 13.1.6.2 Products offered
- 13.1.6.3 Recent developments
- 13.1.6.3.1 Deals
- 13.1.7 NEPHRON PHARMACEUTICALS
- 13.1.7.1 Business overview
- 13.1.7.2 Products offered
- 13.1.7.3 Recent developments
- 13.1.7.3.1 Expansions
- 13.1.8 LEITERS HEALTH
- 13.1.8.1 Business overview
- 13.1.8.2 Products offered
- 13.1.8.3 Recent developments
- 13.1.8.3.1 Expansions
- 13.1.9 ICON GROUP
- 13.1.9.1 Business overview
- 13.1.9.2 Products offered
- 13.1.9.3 Recent developments
- 13.1.9.3.1 Deals
- 13.1.10 ROSEWAY LABS
- 13.1.10.1 Business overview
- 13.1.10.2 Products offered
- 13.1.10.3 Recent developments
- 13.1.10.3.1 Other developments
- 13.1.11 ITH PHARMA
- 13.1.11.1 Business overview
- 13.1.11.2 Products offered
- 13.1.12 PENCOL COMPOUNDING PHARMACY
- 13.1.12.1 Business overview
- 13.1.12.2 Products offered
- 13.1.13 FORMUL8
- 13.1.13.1 Business overview
- 13.1.13.2 Products offered
- 13.1.14 AURORA COMPOUNDING PHARMACY
- 13.1.14.1 Business overview
- 13.1.14.2 Products offered
- 13.1.1 BAXTER
- 13.2 OTHER PLAYERS
- 13.2.1 CUSTOM MED APOTHECARY
- 13.2.2 INSTITUTIONAL PHARMACY SOLUTIONS
- 13.2.3 FUSION APOTHECARY
- 13.2.4 INNOVATION COMPOUNDING PHARMACY
- 13.2.5 RX3 COMPOUNDING PHARMACY
- 13.2.6 DOUGHERTY'S PHARMACY, INC.
- 13.2.7 LORRAINE'S PHARMACY
- 13.2.8 WELLS PHARMA
- 13.2.9 OLYMPIA PHARMACY
- 13.2.10 FRESH THERAPEUTICS
- 13.2.11 MEDIOS AG
14 APPENDIX
- 14.1 DISCUSSION GUIDE
- 14.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 14.3 CUSTOMIZATION OPTIONS
- 14.4 RELATED REPORTS
- 14.5 AUTHOR DETAILS













