
|
시장보고서
상품코드
1877351
환자 유래 이종 이식/PDX 모델 시장 : 유형별, 서비스별, 이식 방법별, 종양 유형별, 용도별, 지역별 예측(-2030년)Patient Derived Xenografts/PDX Models Market by Type, Service, Implantation Method, Tumor Type, Application - Global Forecast to 2030 |
||||||
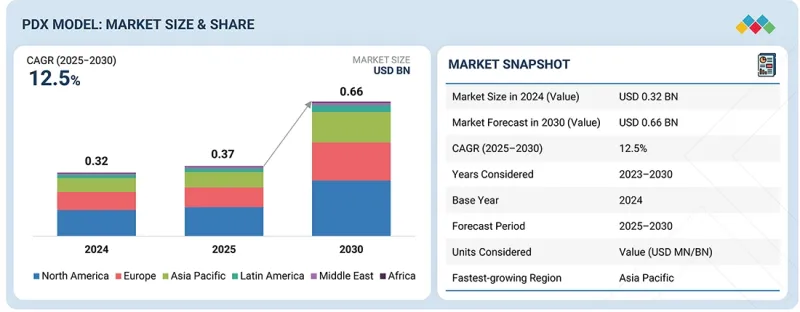
PDX 모델 시장 규모는 예측 기간 동안 CAGR 12.5%를 나타내 2025년 3억 7,000만 달러에서 2030년에는 6억 6,000만 달러에 이를 것으로 전망됩니다.
본 시장의 주요 성장 촉진요인은 암 발생률 증가와 인간 종양 생물학을 충실히 재현하는 효과적인 전임상 모델에 대한 수요 증가입니다. 게다가, 약물 효과 평가, 바이오마커 발견, 맞춤형 치료 접근법의 입안을 목적으로 한 종양학 조사에서 환자 유래 이종 이식/PDX 모델의 이용 확대가 시장 성장을 뒷받침하고 있습니다. 또한, 면역결핍 마우스 모델의 진보와 차세대 시퀀싱 기술의 통합으로 PDX 모델의 전환 가치가 높아지고 있습니다.
| 조사 범위 | |
|---|---|
| 조사 대상 기간 | 2023-2030년 |
| 기준 연도 | 2024년 |
| 예측 기간 | 2025-2030년 |
| 대상 단위 | 금액(10억 달러) |
| 부문 | 이식 방법별, 유형별, 종양 유형별, 용도별, 최종 사용자별 |
| 대상 지역 | 북미, 유럽, 아시아태평양, 라틴아메리카, 중동 및 아프리카 |
그러나 PDX 모델 개발의 고비용성과 시간 소모적 특성, 윤리적 고려 및 모델 재현성의 변화는 시장에서의 광범위한 보급을 여전히 억제하고 있습니다.
동소성 이식 부문은 인간암에서 발견되는 자연적인 종양 환경과 질병 진행을 모방하는 우수한 능력으로 환자 유래 이종 이식/PDX 모델 시장에서 가장 높은 CAGR로 성장할 것으로 예측됩니다. 이 기술은 종양 세포를 원래의 조직 부위에 이식할 수 있어 종양-간질 상호작용, 혈관신생, 전이의 확산을 보다 정확하게 재현할 수 있습니다. 그 결과, 동소성 모델은 다른 모델 유형에 비해 약물의 효능 및 내성 메커니즘을 평가하는 데 더 높은 전환 관련성과 예측 정확도를 제공합니다. 표적요법이나 면역요법의 채용이 증가하고 있는 것도, 치료반응을 평가하는데 있어서 보다 생리학적으로 관련성이 높은 환경을 제공하는 동소성 모델 수요를 더욱 뒷받침하고 있습니다. 또한 영상 진단 기술과 현미 수술 기술의 진보로 동소성 PDX 연구의 재현성과 모니터링이 향상되어 전임상 연구에서 보다 이용하기 쉽고 신뢰성이 높아졌습니다. 이러한 장점은 함께 세계의 PDX 모델 시장에서 동소성 이식 부문의 급속한 성장을 이끌고 있습니다.
환자 유래 이종 이식/PDX 모델 시장에서 소화기(GI) 종양 모델 부문은 가장 큰 점유율을 차지합니다. 이것은 대장암, 위암, 췌장암, 간암을 포함한 소화기계 악성 종양의 세계적인 발생률의 높이에 기인하고 있습니다. 이러한 암은 암 관련 사망의 주요 원인 중 하나이며 종양의 이질성과 진행을 정확하게 재현하는 신뢰성이 높은 전임상 모델에 대한 강한 수요를 만들어내고 있습니다. 소화기계 PDX 모델은 환자 종양의 조직학적·유전적 특징을 충실히 유지하기 때문에 치료 효과나 내성 메커니즘의 예측 정밀도가 높은 평가를 가능하게 합니다. 표적요법, 면역요법, 병용치료 전략의 평가에 있어서 확고한 응용 실적에 의해 전환 종양학 조사의 기반이 되고 있습니다. 게다가 정밀의료에 대한 주목 증가와 소화기 암을 대상으로 한 임상시험 증가가 이러한 모델의 채용을 가속화하고 있습니다. 생착 기술의 지속적인 진보와 분자 프로파일링의 고도화는 소화기계 PDX 모델의 유용성을 더욱 높여 세계 시장에서 주도적 지위를 확고히 하고 있습니다.
미국은 예측 기간 동안 환자 유래 이종 이식/PDX 모델 시장에서 가장 높은 성장률을 나타낼 것으로 전망되고 있습니다. 이 성장은 주로 주요 제약 회사 및 생명 공학 기업의 강력한 존재감, 확립된 연구 인프라 및 광범위한 종양학 연구 이니셔티브에 의해 견인되고 있습니다. 암 이환율의 상승에 더해, 맞춤형 의료나 표적 요법 개발의 보급이 진행되고 있는 것이, PDX 모델에 수요를 대폭 밀어 올리고 있습니다. 또한 전환 연구 및 전임상암 연구에 대한 투자 확대, 학술기관과 산업계의 강력한 연계가 시장 확대를 가속화하고 있습니다.
또한 선진적인 실험 시설의 정비, 종합적인 바이오뱅킹 시스템의 존재, 암치료제 개발에 대한 정부의 지원적 자금 제공이 이 나라 세계 시장에서의 지위를 더욱 강화하고 있습니다. 미국의 규제 환경은 PDX 기반 연구의 신뢰성과 윤리적 컴플라이언스를 보장하면서 혁신을 촉진하고 있습니다. 이러한 요인들이 함께 PDX 모델의 급속한 보급에 공헌하고 북미 세계 시장에서 주도적 입장을 확고하게 하고 있습니다.
본 보고서에서는 세계의 환자 유래 이종 이식/PDX 모델 시장에 대해 조사했으며 모델 유형 및 서비스별, 이식 방법별, 종양 유형별, 용도별, 최종 사용자별, 지역별 동향 및 시장 진출기업 프로파일 등을 정리했습니다.
자주 묻는 질문
목차
제1장 서론
제2장 주요 요약
제3장 중요 인사이트
제4장 시장 개요
- 서론
- 시장 역학
- 미충족 수요(Unmet Needs)와 백스페이스
- 연결된 시장과 부문 간 기회
- Tier 1/2/3 기업의 전략적 움직임
제5장 업계 동향
- Porter's Five Forces 분석
- 생태계 분석
- 밸류체인 분석
- 가격 분석
- 고객의 비즈니스에 영향을 미치는 동향/혼란
- 특허 분석
- 무역 데이터 분석
- 미국 관세가 환자 유래 이종 이식/PDX 모델 시장에 미치는 영향(2025년)
- 거시경제 전망
- 주된 회의와 이벤트(2025-2026년)
- 투자 및 자금조달 시나리오
- 사례 연구 분석
제6장 기술의 진보, AI별 영향, 특허, 혁신 및 미래의 응용
- 주요 신기술
- 보완적 기술
- 기술 로드맵
- 미래의 응용
- AI/생성형 AI가 환자 유래 이종 이식/PDX 모델 시장에 미치는 영향
제7장 지속가능성과 규제상황
- 지역 규제 및 규정 준수
- 지속가능성에 미치는 영향과 규제 정책의 노력
- 인증, 라벨, 환경 기준
제8장 고객정세와 구매행동
- 의사결정 프로세스
- 구매자의 이해관계자와 구매평가기준
- 채용 장벽과 내부 과제
- 최종 이용 산업의 미충족 수요(Unmet Needs)
제9장 환자 유래 이종 이식/PDX 모델 시장, 모델 유형 및 서비스별
- 서론
- PDX 모델
- 서비스
제10장 환자 유래 이종 이식/PDX 모델 시장, 이식 방법별
- 서론
- 피하 이식
- 동위 이식
- 기타
제11장 환자 유래 이종 이식/PDX 모델 시장, 종양 유형별
- 서론
- 위장관 종양 모델
- 부인과 종양 모델
- 호흡기 종양 모델
- 비뇨기 종양 모델
- 혈액종양 모델
- 기타
제12장 환자 유래 이종 이식/PDX 모델 시장, 용도별
- 서론
- 전임상 연구
- 바이오마커 분석
- 전환 연구
- 바이오뱅킹
제13장 환자 유래 이종 이식/PDX 모델 시장, 최종 사용자별
- 서론
- 제약 및 바이오테크놀러지 기업
- CROS 및 CDMOS
- 학술 및 연구 기관
제14장 환자 유래 이종 이식/PDX 모델 시장, 지역별
- 서론
- 북미
- 미국
- 캐나다
- 유럽
- 독일
- 영국
- 프랑스
- 이탈리아
- 스페인
- 기타
- 아시아태평양
- 중국
- 일본
- 인도
- 호주
- 한국
- 기타
- 라틴아메리카
- 브라질
- 멕시코
- 기타
- 중동
- GCC 국가
- 기타
- 아프리카
제15장 경쟁 구도
- 서론
- 주요 진입기업의 전략/강점
- 수익 분석(2022-2024년)
- 시장 점유율 분석(2024년)
- 기업평가 매트릭스 : 주요 진입기업(2024년)
- 기업평가 매트릭스 : 스타트업/중소기업(2024년)
- 기업평가와 재무지표
- 브랜드/제품 비교
- 경쟁 시나리오
제16장 기업 프로파일
- 주요 진출기업
- CHARLES RIVER LABORATORIES
- THE JACKSON LABORATORY
- INOTIV
- JSR CORPORATION
- GENOWAY
- WUXI APPTEC WUXI BIOLOGICS
- BIOCYTOGEN PHARMACEUTICALS CO., LTD.
- TACONIC BIOSCIENCES, INC.
- ONCODESIGN SERVICES(ONCODESIGN PRECISION MEDICINE)
- CHAMPIONS ONCOLOGY, INC.
- PHARMARON
- SYNGENE INTERNATIONAL LIMITED
- LABCORP
- EUROFINS SCIENTIFIC
- VITALSTAR BIOTECHNOLOGY CO., LTD.
- 기타 기업
- CREATIVE ANIMODEL
- JOINN LABORATORIES(CHINA) CO., LTD.
- CREATIVE BIOLABS
- ARAGEN LIFE SCIENCES LTD.
- PHARMTEST SERVICES
- LIDE SHANGHAI BIOTECH CO., LTD.
- CERTIS ONCOLOGY SOLUTIONS
- INNOSER
- SHANGHAI CHEMPARTNER
- IVRS AB
제17장 조사 방법
제18장 부록
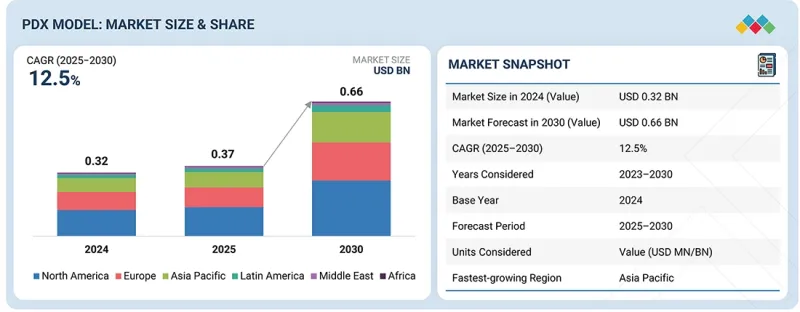
KTH 25.11.28The PDX model market is expected to reach USD 0.66 billion in 2030 from USD 0.37 billion in 2025, at a CAGR of 12.5% during the forecast period. The market is primarily driven by the increasing incidence of cancer and the growing demand for effective preclinical models that closely replicate human tumor biology. The increasing use of patient-derived xenograft (PDX) models in oncology research to evaluate drug efficacy, discover biomarkers, and inform personalized treatment approaches further supports market growth. Additionally, advancements in immunodeficient mouse models and the integration of PDX platforms with next-generation sequencing technologies are enhancing their translational value.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2023-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD billion) |
| Segments | Implantation Method, Type, Tumor Type, Applications, End User |
| Regions covered | North America, Europe, the Asia Pacific, Latin America, the Middle East, and Africa |
However, the high cost and time-intensive nature of PDX model development, along with ethical considerations and variability in model reproducibility, continue to restrain widespread market adoption.
"Orthotopic implantation segment is expected to grow at the highest CAGR during the forecast period"
The orthotopic implantation segment is expected to grow at the highest CAGR in the patient-derived xenograft (PDX) model market owing to its superior ability to mimic the natural tumor environment and disease progression seen in human cancers. This technique allows tumor cells to be implanted at their original tissue site, enabling a more accurate representation of tumor-stroma interactions, angiogenesis, and metastatic spread. As a result, orthotopic models provide higher translational relevance and predictive accuracy for assessing drug efficacy and resistance mechanisms compared to other model types. The increasing adoption of targeted therapies and immunotherapies has further fueled the demand for orthotopic models, as they provide a more physiologically relevant context for evaluating therapeutic responses. Additionally, advancements in imaging modalities and microsurgical techniques have improved the reproducibility and monitoring of orthotopic PDX studies, making them more accessible and reliable for preclinical research. These advantages collectively drive the rapid growth of the orthotopic implantation segment in the global PDX model market.
"Gastrointestinal tumor model segment holds the largest share of the market"
The gastrointestinal (GI) tumor model segment holds the largest share of the patient-derived xenograft (PDX) model market, driven by the high global incidence of gastrointestinal malignancies, including colorectal, gastric, pancreatic, and liver cancers. These cancers are among the leading causes of cancer-related mortality, creating a strong demand for reliable preclinical models that accurately replicate tumor heterogeneity and progression. Gastrointestinal PDX models closely preserve the histological and genetic characteristics of patient tumors, enabling more predictive evaluation of therapeutic efficacy and resistance mechanisms. Their robust application in assessing targeted therapies, immunotherapies, and combination treatment strategies has made them a cornerstone of translational oncology research. Furthermore, the growing emphasis on precision medicine and the rising volume of clinical trials focused on GI cancers have accelerated the adoption of these models. Continuous advancements in engraftment techniques and molecular profiling further enhance the utility of gastrointestinal PDX models, solidifying their leading position in the global market.
"The US is expected to grow at the highest CAGR during the forecast period"
The US is expected to register the highest growth rate in the patient-derived xenograft (PDX) model market during the forecast period. This growth is primarily driven by the strong presence of leading pharmaceutical and biotechnology companies, well-established research infrastructure, and extensive oncology research initiatives. The rising prevalence of cancer, coupled with increasing adoption of personalized medicine and targeted therapy development, is significantly boosting the demand for PDX models. Furthermore, growing investment in translational and preclinical cancer research, along with strong collaborations between academic institutions and industry players, is accelerating market expansion.
Additionally, the availability of advanced laboratory facilities, the presence of comprehensive biobanking systems, and supportive government funding for cancer drug discovery further strengthen the country's position in the global market. The US regulatory environment also promotes innovation while ensuring the reliability and ethical compliance of PDX-based studies. These factors collectively contribute to the rapid adoption of PDX models, reinforcing North America's leadership in the global market.
The primary interviews conducted for this report can be categorized as follows:
- By Respondent: Tier 1 - 25%, Tier 2 - 35%, and Tier 3 - 40%
- By Designation: Managers - 45%, CXOs and Directors - 30%, and Executives - 25%
- By Region: North America - 35%, Europe - 25%, Asia Pacific - 15%, Latin America - 10%, the Middle East - 10%, and Africa - 5%
JSR Corporation (Japan), WuXi AppTec (China), The Jackson Laboratory (US), Charles River Laboratories (US), Taconic Biosciences, Inc. (US), Oncodesign Precision Medicine (France), Inotiv (US), Pharmatest Services (Finland), Hera Biolabs (US), EPO Berlin-Buch GmbH (Germany), XenTech (France), Urosphere (France), Altogen Labs (US), Abnova Corporation (Taiwan), Genesis Biotechnology Group (US), Biocytogen Pharmaceuticals (Beijing) Co., Ltd. (China), Creative Animodel (US), BioDuro (US), Aragen Life Sciences (India), LIDE Shanghai Biotech, Ltd (China), Certis Oncology Solutions (US), InnoSer (Netherlands), IVRS AB (Sweden), Beijing IDMO Co., Ltd. (China), and Shanghai ChemPartner (China) are some of the key companies offering PDX models.
Research Coverage
This research report categorizes the PDX model market by implantation method (subcutaneous implantation, orthotopic implantation, others), type (mouse model, rat model), tumor type (gastrointestinal, gynecological, respiratory, urological, hematological, other tumor models), application (preclinical drug development, biomarker analysis, translational research, biobanks), end user (pharmaceutical & biotechnology companies, contract research organizations (CROs), academic & research institutes), and region (North America, Europe, Asia Pacific, Latin America, Middle East, And Africa).
The report's scope encompasses detailed information about the primary factors, including drivers, restraints, challenges, and opportunities, that influence the growth of the PDX model market. A comprehensive analysis of key industry players has been performed to provide insights into their business overview, product portfolio, key strategies, new product launches, acquisitions, and recent developments related to the PDX model market. This report also includes a competitive analysis of emerging startups in the PDX model industry ecosystem.
Key Benefits of Buying the Report
The report will assist market leaders and new entrants by providing revenue estimates for the overall market and its subsegments. It will also help stakeholders better understand the competitive landscape and gain more insights to position their businesses effectively and develop suitable go-to-market strategies. This report will enable stakeholders to grasp the market's pulse and offer information on key market drivers, restraints, opportunities, and challenges.
The report provides insights into the following pointers:
- Analysis of key drivers (growing demand for personalized medicine, technological advancements in PDX models, rising investments in cancer research), restraints (discontinuation of animal models for clinical trials by the FDA), opportunities (emergence of CRISPR in biomedical research), and challenges (development of alternative animal testing methods) influencing the market growth
- Product Development/Innovation: Detailed insights into newly launched products and technological assessment of the PDX model market
- Market Development: Comprehensive information about lucrative markets and analysis of the market across varied regions
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the PDX model market
- Competitive Assessment: In-depth assessment of market shares, growth strategies, and product offerings of leading players, including JSR Corporation (Japan), WuXi AppTec (China), The Jackson Laboratory (US), Charles River Laboratories (US), Taconic Biosciences, Inc. (US), among others offering products for the PDX model market. Other companies include BioDuro (US), Aragen Life Sciences (India), LIDE Shanghai Biotech, Ltd (China), Certis Oncology Solutions (US), InnoSer (Netherlands), among others, in the PDX model market
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.2.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.2.2 YEARS CONSIDERED
- 1.3 STUDY SCOPE
- 1.3.1 INCLUSIONS & EXCLUSIONS
- 1.3.2 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS & MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: INSIGHTS & STRATEGIC IMPERATIVES
- 2.3 DISRUPTIVE TRENDS SHAPING PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 2.4 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 NORTH AMERICA: PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE AND COUNTRY (2024)
- 3.2 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- 3.3 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SHARE, BY IMPLANTATION, 2025 VS. 2030 (%)
- 3.4 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET SHARE, BY SERVICE, 2024 (%)
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Growing demand for immuno-oncology and complex biologics
- 4.2.1.2 Rising preference for humanized PDX models
- 4.2.1.3 Focus on government-funded initiatives for cancer research
- 4.2.2 RESTRAINTS
- 4.2.2.1 Introduction of 3D printed models
- 4.2.2.2 Ethical concerns and regulatory restrictions
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Increasing preference for personalized medicines in oncology
- 4.2.3.2 Growing demand for CDX models for PDX model vendors
- 4.2.4 CHALLENGES
- 4.2.4.1 Limitations of humanized mouse models
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS & WHITE SPACES
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 BARGAINING POWER OF SUPPLIERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 ECOSYSTEM ANALYSIS
- 5.3 VALUE CHAIN ANALYSIS
- 5.4 PRICING ANALYSIS
- 5.4.1 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY KEY PLAYER, 2022-2024
- 5.4.2 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY IMPLANTATION METHOD, 2022-2024
- 5.4.3 AVERAGE SELLING PRICE TREND OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY TUMOR TYPE, 2022-2024
- 5.4.4 AVERAGE SELLING PRICE OF PATIENT-DERIVED XENOGRAFT (PDX) MODELS, BY REGION, 2024
- 5.5 TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS
- 5.6 PATENT ANALYSIS
- 5.6.1 METHODOLOGY
- 5.6.2 NUMBER OF PATENTS FILED, BY DOCUMENT TYPE
- 5.6.3 LIST OF KEY PATENTS
- 5.7 TRADE DATA ANALYSIS
- 5.7.1 IMPORT DATA FOR HS CODE 106.19.90, 2020-2024
- 5.7.2 EXPORT DATA FOR HS CODE 106.19.90, 2020-2024
- 5.8 IMPACT OF 2025 US TARIFF ON PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 5.8.1 KEY TARIFF RATES
- 5.8.2 PRICE IMPACT ANALYSIS
- 5.8.3 KEY IMPACT ON VARIOUS REGIONS
- 5.8.3.1 North America
- 5.8.3.2 Europe
- 5.8.3.3 Asia Pacific
- 5.8.3.4 Rest of the World
- 5.8.4 IMPACT ON END-USE INDUSTRIES
- 5.8.4.1 Pharmaceutical & biotechnology companies
- 5.8.4.2 CROs & CDMOs
- 5.8.4.3 Academic & research institutes
- 5.9 MACROECONOMIC OUTLOOK
- 5.9.1 INTRODUCTION
- 5.9.2 GDP TRENDS AND FORECAST
- 5.9.3 R&D TRENDS IN GLOBAL HEALTHCARE INDUSTRY
- 5.9.4 R&D TRENDS IN GLOBAL PHARMA INDUSTRY
- 5.10 KEY CONFERENCES & EVENTS, 2025-2026
- 5.11 INVESTMENT & FUNDING SCENARIO
- 5.11.1 VC/PRIVATE EQUITY INVESTMENT TRENDS & STARTUP LANDSCAPE
- 5.12 CASE STUDY ANALYSIS
- 5.12.1 IMAGING-READY ORTHOTOPIC PDX FOR PEDIATRIC BRAIN TUMORS
- 5.12.2 HOST-STRAIN SELECTION USING BREAST CANCER PDX
- 5.12.3 RESISTANCE-ANCHORED PDX PANEL TO DESIGN MOUSE CLINICAL TRIAL
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 KEY EMERGING TECHNOLOGIES
- 6.1.1 NEXT-GENERATION SEQUENCING
- 6.1.2 GENE EDITING
- 6.2 COMPLEMENTARY TECHNOLOGIES
- 6.2.1 MULTI-OMICS
- 6.2.2 MULTI-MODEL PRECLINICAL IMAGING
- 6.3 TECHNOLOGY ROADMAP
- 6.4 FUTURE APPLICATIONS
- 6.5 IMPACT OF AI/GEN AI ON PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 6.5.1 TOP USE CASES & MARKET POTENTIAL
- 6.5.2 CASE STUDIES OF AI IMPLEMENTATION
- 6.5.3 BEST PRACTICES IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS CREATION
- 6.5.4 CLIENTS' READINESS TO ADOPT GENERATIVE AI IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET
- 6.5.5 HIGH READINESS VS. ADOPTION BARRIERS
7 SUSTAINABILITY AND REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS & COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 INDUSTRY STANDARDS
- 7.1.2.1 North America
- 7.1.2.2 Europe
- 7.1.2.3 Asia Pacific
- 7.1.2.4 Latin America
- 7.2 SUSTAINABILITY IMPACT & REGULATORY POLICY INITIATIVES
- 7.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS & BUYING EVALUATION CRITERIA
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM END-USE INDUSTRIES
9 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY MODEL TYPE & SERVICE
- 9.1 INTRODUCTION
- 9.2 PDX MODELS
- 9.2.1 MOUSE MODEL
- 9.2.1.1 Proven efficacy of mouse model in cancer research and drug development to boost segment growth
- 9.2.2 RAT MODEL
- 9.2.2.1 Emergence of advanced technologies in PDX rat models to aid market growth
- 9.2.1 MOUSE MODEL
- 9.3 SERVICES
- 9.3.1 MODEL CREATION & EXPANISON SERVICES
- 9.3.1.1 Need for faster passaging of models to to build study-ready cohorts (P1-P3) to boost segment growth
- 9.3.2 MODEL CHARACTERIZATION SERVICES
- 9.3.2.1 Growing demand for biomarker analysis to drive segment growth
- 9.3.3 CRYOPRESERVATION SERVICES
- 9.3.3.1 Reliable and low-passage biobanking services to offer growth opportunities
- 9.3.4 OTHER PDX SERVICES
- 9.3.1 MODEL CREATION & EXPANISON SERVICES
10 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY IMPLANTATION METHOD
- 10.1 INTRODUCTION
- 10.2 SUBCUTANEOUS IMPLANTATION
- 10.2.1 STANDARDIZED AND SCALABLE PDX MODEL EXPANSION TO DRIVE SEGMENT GROWTH
- 10.3 ORTHOTOPIC IMPLANTATION
- 10.3.1 DEMAND FOR CLINICALLY RELEVANT AND SITE-SPECIFIC PDX MODELS TO BOOST SEGMENT GROWTH
- 10.4 OTHER IMPLANTATION METHODS
11 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY TUMOR TYPE
- 11.1 INTRODUCTION
- 11.2 GASTROINTESTINAL TUMOR MODEL
- 11.2.1 HIGH TISSUE AVAILABILITY AND STANDARDIZED ENGRAFTMENT PROTOCOLS TO FUEL MARKET GROWTH
- 11.3 GYNECOLOGICAL TUMOR MODEL
- 11.3.1 EXPANDING ORTHOTOPIC AND HUMANIZED PDX APPLICATIONS TO DRIVE MARKET
- 11.4 RESPIRATORY TUMOR MODEL
- 11.4.1 INCREASING ADOPTION OF ORTHOTOPIC AND HUMANIZED MODELS TO BOOST SEGMENT GROWTH
- 11.5 UROLOGICAL TUMOR MODEL
- 11.5.1 EXPANSION OF HORMONE-CONTEXT AND IMMUNE-COMPETENT MODELS TO FAVOR MARKET ADOPTION
- 11.6 HEMATOLOGICAL TUMOR MODEL
- 11.6.1 RISING ADOPTION OF HUMANIZED AND RELAPSE-DERIVED MODELS TO DRIVE SEGMENT GROWTH
- 11.7 OTHER TUMOR MODELS
12 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY APPLICATION
- 12.1 INTRODUCTION
- 12.2 PRECLINICAL RESEARCH
- 12.2.1 HIGH IMPORTANCE FOR PDX IN EFFICACY CONFIRMATION TO DRIVE MARKET
- 12.3 BIOMARKER ANALYSIS
- 12.3.1 DEMAND FOR MULTI-OMICS TO AUGMENT MARKET GROWTH
- 12.4 TRANSLATIONAL RESEARCH
- 12.4.1 DEMAND FOR PDX MODELS IN CO-CLINICAL TRIAL DESIGNS TO BOOST SEGMENT GROWTH
- 12.5 BIOBANKING
- 12.5.1 DEMAND FOR PDX MODELS IN SPECIALIZED BANKS TO SPUR MARKET GROWTH
13 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY END USER
- 13.1 INTRODUCTION
- 13.2 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
- 13.2.1 INTEGRATION OF TRANSLATIONALLY RELEVANT PDX WORKFLOWS TO DRIVE MARKET GROWTH
- 13.3 CROS & CDMOS
- 13.3.1 EXPANDED OUTSOURCING THROUGH INTEGRATED WORKFLOWS TO FUEL MARKET GROWTH
- 13.4 ACADEMIC & RESEARCH INSTITUTES
- 13.4.1 ENHANCED INSTITUTIONAL BIOBANKS AND MULTI-OMIC PROFILING TO AUGMENT MARKET GROWTH
14 PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, BY REGION
- 14.1 INTRODUCTION
- 14.2 NORTH AMERICA
- 14.2.1 US
- 14.2.1.1 US to dominate North American PDX models market during study period
- 14.2.2 CANADA
- 14.2.2.1 Growth research and academic activities on oncology to fuel market growth
- 14.2.1 US
- 14.3 EUROPE
- 14.3.1 GERMANY
- 14.3.1.1 Growth in biotechnology industry and strong pharma presence to spur market growth
- 14.3.2 UK
- 14.3.2.1 Increasing focus on cancer research to propel market
- 14.3.3 FRANCE
- 14.3.3.1 Robust research activities in immune-oncology to drive market growth
- 14.3.4 ITALY
- 14.3.4.1 Growing pharmaceutical & biotechnology industry and increasing focus on radiopharmaceuticals to favor market growth
- 14.3.5 SPAIN
- 14.3.5.1 Increasing translational activities to boost market growth
- 14.3.6 REST OF EUROPE
- 14.3.1 GERMANY
- 14.4 ASIA PACIFIC
- 14.4.1 CHINA
- 14.4.1.1 Favorable investments in life sciences research to drive market
- 14.4.2 JAPAN
- 14.4.2.1 Increasing focus on personalized diagnostics products to drive market
- 14.4.3 INDIA
- 14.4.3.1 Favorable initiatives for drug discovery & development to boost market growth
- 14.4.4 AUSTRALIA
- 14.4.4.1 High number of research institutes to favor market growth
- 14.4.5 SOUTH KOREA
- 14.4.5.1 Innovation in health and biotech sectors to support market uptake
- 14.4.6 REST OF ASIA PACIFIC
- 14.4.1 CHINA
- 14.5 LATIN AMERICA
- 14.5.1 BRAZIL
- 14.5.1.1 Increasing investments in biopharmaceutical research to fuel uptake
- 14.5.2 MEXICO
- 14.5.2.1 Expanding healthcare ecosystem to support market growth
- 14.5.3 REST OF LATIN AMERICA
- 14.5.1 BRAZIL
- 14.6 MIDDLE EAST
- 14.6.1 GCC COUNTRIES
- 14.6.1.1 Kingdom of Saudi Arabia
- 14.6.1.1.1 Advanced healthcare sector and prevalence of chronic diseases to boost market
- 14.6.1.2 UAE
- 14.6.1.2.1 Increasing R&D activities and growing demand for personalized medicines to propel market
- 14.6.1.3 Rest of GCC Countries
- 14.6.1.1 Kingdom of Saudi Arabia
- 14.6.2 REST OF MIDDLE EAST
- 14.6.1 GCC COUNTRIES
- 14.7 AFRICA
- 14.7.1 INCREASING FOCUS ON PRECISION MEDICINE INITIATIVES TO SUPPORT MARKET GROWTH
15 COMPETITIVE LANDSCAPE
- 15.1 INTRODUCTION
- 15.2 KEY PLAYERS STRATEGIES/RIGHT TO WIN
- 15.2.1 STRATEGIES ADOPTED BY KEY PLAYERS IN PATIENT-DERIVED XENOGRAFT (PDX) MODELS MARKET, 2022-2025
- 15.3 REVENUE ANALYSIS, 2022-2024
- 15.4 MARKET SHARE ANALYSIS, 2024
- 15.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2024
- 15.5.1 STARS
- 15.5.2 EMERGING LEADERS
- 15.5.3 PERVASIVE PLAYERS
- 15.5.4 PARTICIPANTS
- 15.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2024
- 15.5.5.1 Company footprint
- 15.5.5.2 Region footprint
- 15.5.5.3 Model type & service footprint
- 15.5.5.4 Tumor type footprint
- 15.5.5.5 Implantation method footprint
- 15.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2024
- 15.6.1 PROGRESSIVE COMPANIES
- 15.6.2 RESPONSIVE COMPANIES
- 15.6.3 DYNAMIC COMPANIES
- 15.6.4 STARTING BLOCKS
- 15.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SME PLAYERS, 2024
- 15.6.5.1 Detailed list of key startups/SMEs
- 15.6.5.2 Competitive benchmarking of key startups/SMEs
- 15.7 COMPANY VALUATION & FINANCIAL METRICS
- 15.7.1 FINANCIAL METRICS
- 15.7.2 COMPANY VALUATION
- 15.8 BRAND/PRODUCT COMPARISON
- 15.9 COMPETITIVE SCENARIO
- 15.9.1 DEALS
- 15.9.2 EXPANSIONS
16 COMPANY PROFILES
- 16.1 KEY PLAYERS
- 16.1.1 CHARLES RIVER LABORATORIES
- 16.1.1.1 Business overview
- 16.1.1.2 Products/Services offered
- 16.1.1.3 Recent developments
- 16.1.1.3.1 Deals
- 16.1.1.3.2 Expansions
- 16.1.1.4 MnM view
- 16.1.1.4.1 Key strengths
- 16.1.1.4.2 Strategic choices
- 16.1.1.4.3 Weaknesses & competitive threats
- 16.1.2 THE JACKSON LABORATORY
- 16.1.2.1 Business overview
- 16.1.2.2 Products/Services offered
- 16.1.2.3 Recent developments
- 16.1.2.3.1 Deals
- 16.1.2.3.2 Expansions
- 16.1.2.4 MnM view
- 16.1.2.4.1 Key strengths
- 16.1.2.4.2 Strategic choices
- 16.1.2.4.3 Weaknesses & competitive threats
- 16.1.3 INOTIV
- 16.1.3.1 Business overview
- 16.1.3.2 Products/Services offered
- 16.1.3.3 Recent developments
- 16.1.3.3.1 Expansions
- 16.1.3.4 MnM view
- 16.1.3.4.1 Key strengths
- 16.1.3.4.2 Strategic choices
- 16.1.3.4.3 Weaknesses & competitive threats
- 16.1.4 JSR CORPORATION
- 16.1.4.1 Business overview
- 16.1.4.2 Product/Services offered
- 16.1.4.3 Recent developments
- 16.1.4.3.1 Deals
- 16.1.4.3.2 Expansions
- 16.1.4.4 MnM view
- 16.1.4.4.1 Key strengths
- 16.1.4.4.2 Strategic choices
- 16.1.4.4.3 Weaknesses & competitive threats
- 16.1.5 GENOWAY
- 16.1.5.1 Business overview
- 16.1.5.2 Product/Solutions offered
- 16.1.5.3 MnM view
- 16.1.5.3.1 Key strengths
- 16.1.5.3.2 Strategic choices
- 16.1.5.3.3 Weaknesses & competitive threats
- 16.1.6 WUXI APPTEC + WUXI BIOLOGICS
- 16.1.6.1 Business overview
- 16.1.6.2 Product/Services offered
- 16.1.6.3 Recent developments
- 16.1.6.3.1 Deals
- 16.1.6.3.2 Expansions
- 16.1.7 BIOCYTOGEN PHARMACEUTICALS CO., LTD.
- 16.1.7.1 Business overview
- 16.1.7.2 Product/Services offered
- 16.1.7.3 Recent developments
- 16.1.7.3.1 Product/Service upgrades
- 16.1.7.3.2 Expansions
- 16.1.8 TACONIC BIOSCIENCES, INC.
- 16.1.8.1 Business overview
- 16.1.8.2 Products/Services offered
- 16.1.8.3 Recent developments
- 16.1.8.3.1 Product/Service launches
- 16.1.8.3.2 Deals
- 16.1.8.3.3 Expansions
- 16.1.9 ONCODESIGN SERVICES (ONCODESIGN PRECISION MEDICINE)
- 16.1.9.1 Business overview
- 16.1.9.2 Products/Services offered
- 16.1.10 CHAMPIONS ONCOLOGY, INC.
- 16.1.10.1 Business overview
- 16.1.10.2 Products/Services offered
- 16.1.10.3 Recent developments
- 16.1.10.3.1 Deals
- 16.1.11 PHARMARON
- 16.1.11.1 Business overview
- 16.1.11.2 Products/Services offered
- 16.1.12 SYNGENE INTERNATIONAL LIMITED
- 16.1.12.1 Business overview
- 16.1.12.2 Products/Services offered
- 16.1.13 LABCORP
- 16.1.13.1 Business overview
- 16.1.13.2 Products/Services offered
- 16.1.14 EUROFINS SCIENTIFIC
- 16.1.14.1 Business overview
- 16.1.14.2 Products/Services offered
- 16.1.15 VITALSTAR BIOTECHNOLOGY CO., LTD.
- 16.1.15.1 Business overview
- 16.1.15.2 Products/Services offered
- 16.1.1 CHARLES RIVER LABORATORIES
- 16.2 OTHER PLAYERS
- 16.2.1 CREATIVE ANIMODEL
- 16.2.2 JOINN LABORATORIES (CHINA) CO., LTD.
- 16.2.3 CREATIVE BIOLABS
- 16.2.4 ARAGEN LIFE SCIENCES LTD.
- 16.2.5 PHARMTEST SERVICES
- 16.2.6 LIDE SHANGHAI BIOTECH CO., LTD.
- 16.2.7 CERTIS ONCOLOGY SOLUTIONS
- 16.2.8 INNOSER
- 16.2.9 SHANGHAI CHEMPARTNER
- 16.2.10 IVRS AB
17 RESEARCH METHODOLOGY
- 17.1 RESEARCH DATA
- 17.1.1 SECONDARY DATA
- 17.1.2 KEY OBJECTIVES OF SECONDARY RESEARCH
- 17.1.3 PRIMARY DATA
- 17.2 MARKET ESTIMATION METHODOLOGY
- 17.2.1 COMPANY REVENUE ANALYSIS: BOTTOM-UP APPROACH
- 17.2.2 INSIGHTS FROM PRIMARY EXPERTS
- 17.2.3 TOP-DOWN APPROACH
- 17.3 GROWTH FORECAST
- 17.4 DATA TRIANGULATION
- 17.5 STUDY ASSUMPTIONS
- 17.6 RESEARCH LIMITATIONS
- 17.7 MARKET FORECAST
- 17.8 RISK ANALYSIS
18 APPENDIX
- 18.1 DISCUSSION GUIDE
- 18.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 18.3 CUSTOMIZATION OPTIONS
- 18.4 RELATED REPORTS
- 18.5 AUTHOR DETAILS





