
|
시장보고서
상품코드
1690953
만성 정맥 폐색 치료 시장 : 시장 점유율 분석, 산업 동향 및 통계, 성장 예측(2025-2030년)Chronic Venous Occlusions Treatment - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030) |
||||||
 Mordor Intelligence
Mordor Intelligence
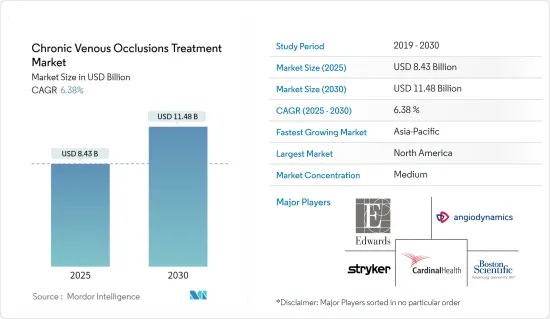
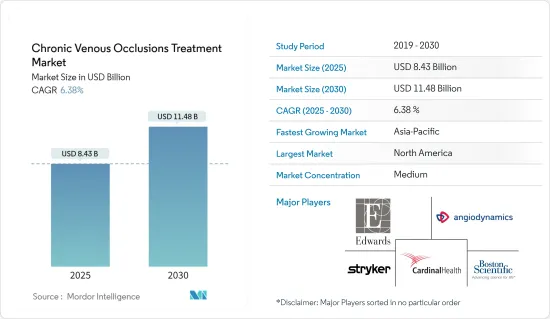
만성 정맥 폐색 치료 시장 규모는 2025년에 84억 3,000만 달러로 추정되고, 예측 기간 2025년부터 2030년까지 CAGR 6.38%로 성장할 전망이며, 2030년에는 114억 8,000만 달러에 달할 것으로 예측됩니다.
만성 정맥 폐색 치료는 만성 정맥 질환의 치료에 사용됩니다. 정맥 폐색은 정맥이 혈전, 근육, 동맥 및 기타 정맥과 같은 주변 구조물에 의해 폐색, 협착 및 압박되는 것입니다. 그 결과 혈액이 쌓이고 역류하여 부위에 통증과 붓기가 생깁니다.
세계 정맥 폐색성 질병의 높은 유병률은 시장 성장을 뒷받침하고 있습니다. 예를 들어, 2024년 5월 International Wound Journal에 게재된 연구에 따르면 만성 정맥 질환의 평균 유병률은 58.4%, 정맥류의 유병률은 22.1%였습니다. 이러한 세계 수준에서 높은 유병률은 시장 성장을 뒷받침하고 있습니다.
주요 시장 기업은 특히 혈관 질환 치료를 위한 저침습 장치의 제품 포트폴리오를 강화하기 위해 인수와 같은 전략적 움직임을 점점 강화하고 있습니다.
예를 들어, 2024년 5월, Siemens Hertiners는 자회사인 Varian을 통해 Innova Basculer의 인수를 결정했습니다. 의료기기 업계에서 유명한 Innova Vascular사는 혈관 질환 치료에 특화된 최첨단 저침습 기기를 전문으로 하고 있습니다. 주요 제품인 라구나 혈전 제거 시스템은 탁월한 유연성, 추적가능성, 다양한 크기로 높은 평가를 받고 있습니다.
이러한 특성을 통해 헬스케어 제공업체는 환자의 프로파일이나 혈전의 변형에 관계없이 정맥 혈전 색전증에 효과적으로 대처할 수 있습니다. 이 인수를 통해 Siemens Hertiners는 혈전 치료에 대한 제품 라인업을 크게 강화하고 예측 기간 동안 시장이 크게 성장할 무대를 마련했습니다.
게다가 제약회사는 정맥부전 치료에 대한 인식을 높이기 위한 이니셔티브를 솔선수범하여 추진하고 있으며, 예측기간 동안 시장 성장을 가속하고 있습니다. 예를 들어, 2023년 7월, LES LABORATOIRES SERVIER는 정맥 질환 계발에 특화된 종합적인 캠페인을 전개했습니다. 헬스케어 전문가나 환자와 밀접하게 협력함으로써, 회사는 정맥부전의 이해를 깊게 하고 증상 관리를 강화하는 것을 목표로 하고 있습니다. 제약 기업에 의한 이러한 협력적인 노력은 앞으로 수년간 시장 성장을 견인할 것으로 보입니다.
이와 같이 정맥질환의 높은 유병률, 정맥질환에 대한 의식의 고조, 신제품 발매 등 상술한 요인 덕분에 시장의 성장은 그 대부분을 견인하고 있습니다. 그러나 치료와 관련된 비용과 위험은 시장 성장을 방해할 수 있습니다.
만성 정맥 폐색 치료 시장 동향
심부정맥 부전 부문이 예측 기간 동안 시장 성장을 견인할 것으로 예측
만성 정맥 폐색 치료는 주로 항생제와 항응고제로 구성됩니다. 이 약은 혈관 내의 혈전을 예방하거나 치료하는 것을 목표로 합니다. 이러한 혈전은 뇌졸중, 심장 마비, 심부정맥 혈전증, 폐 색전증 등 심각한 위험을 초래합니다. 항응고제는 혈전 형성을 예방하고 혈전 용해제는 기존 혈전을 용해시킵니다.
심부정맥혈전증의 관리에 유효한 항응고제에는 에녹사파린, 달테파린, 틴자팔린 등 저분자량 헤파린(LMWH)부터 미분화 헤파린(UFH), 인자Xa 억제제, 직접 트롬빈 억제제, 와파린 등의 비타민 K 길항제까지 다양한 것이 있습니다
만성 정맥 폐색에 맞는 새로운 경구 항응고제의 채용이 증가하고 있다는 것이 이 부문의 성장을 뒷받침하고 있습니다.
바이오시밀러의 등장은 시장 경쟁을 격화시키고 만성 정맥 폐색 치료의 비용을 낮추는 방향에 있습니다. 이 비용 절감은 환자의 편의성을 높이고 이 부문 성장에 직접적인 영향을 미칩니다. 2023년 9월, Fresenius Kabi Canada는 에녹사파린의 바이오시밀러인 ELONOX의 공적 상환 승인을 캐나다 전주에서 취득했습니다. ELONOX. 캐나다 보건부는 ELONOX(에녹사파린 나트륨)를 심부정맥 혈전증을 포함한 혈전 색전증 예방제로 특별히 승인했습니다. 보건 당국에 의한 이러한 상환은 접근성과 적응성 향상에 기여하고 부문의 성장을 더욱 촉진합니다.
심부정맥 혈전증을 대상으로 한 임상 연구에 대한 관심 증가는 부문 성장의 중요한 드라이버가 될 것으로 예상됩니다. Bayer사는 2024년 2월 혁신적인 항-a2 항-플라스민(항-a2ap) 항체인 BAY3018250의 DVT 환자를 대상으로 한 2상 임상시험을 시작했습니다. 본 시험의 결과는 본 항체가 심한 질환에 대한 중요한 치료 수단이 될 가능성을 나타내는 것으로 기대됩니다. 바이엘은 이 결과를 받아 DVT 환자를 대상으로 한 이 항체의 임상시험을 다음 단계로 진행하고자 합니다. 이러한 노력은 환자에게 유망하며,이 부문의 성장을 뒷받침합니다.
따라서 바이오시밀러의 상승, DVT의 임상시험 증가, 바이어의 신약 임상시험과 같은 대기업의 전략적 움직임 등의 요인이 겹쳐 향후 수년간 이 부문의 대폭적인 성장이 예상됩니다.
예측기간 중에는 북미가 시장을 독점
북미가 만성 정맥 폐색 치료 시장을 독점할 전망입니다. 이 시장은 첨단 건강 관리 인프라, 높은 정맥 질환 유병률, 의료기기 및 의약품에 대한 견고한 규제 승인에 의해 지원됩니다.
이 지역 시장은 정맥 질환의 높은 유병률에 견인되어 성장을 이루고 있습니다. 예를 들어, 2023년 4월 미국 국립위생연구소(NIH)는 2,500만 명 이상의 미국인이 정맥류로 고통을 겪고 있으며, 그 중 600만명이 심한 정맥질환에 직면하고 있다고 보고했습니다. 이것은 만성 정맥 질환이 심각한 건강 문제임을 돋보이게 합니다. 이러한 질환에 의한 부담의 증대가 만성 정맥 폐색 치료의 보급을 촉진하고, 시장의 성장에 박차를 가하고 있습니다.
게다가 2023년 10월에 Koninklijke Philips NV가 비악성 장 대퇴부 폐색성 질환을 대상으로 듀오 정맥 스텐트 시스템을 평가한 VIVID 시험 등 진행중인 임상시험이 시장을 더욱 밀어올릴 것입니다. Duo Hybrid 스텐트와 Duo Extend 스텐트로 구성된 Duo 시스템은 장 대퇴 정맥 부문의 다양한 해부학적 및 기계적 요구에 대응합니다. 장비의 효율성과 안전성을 보장하기 위한 이러한 테스트는 시장의 향후 성장에 매우 중요합니다.
또한 심부정맥혈전증 치료용으로 설계된 Pharmascience Canada사의 Pms-rivaroxaban과 같은 제네릭 의약품의 변종이 2023년 11월에 도입됨으로써 시장의 접근성과 적응성이 높아집니다. 이러한 신약 출시는 향후 수년간 시장 성장의 원동력이 될 것으로 보입니다.
심혈관 질환과 정맥 질환의 유병률이 상승하고 있는 것 외에 진행중인 임상시험이나 제품 출시가 기세를 늘리고 있기 때문에 만성 정맥 폐색 치료 시장은 예측기간 중에 크게 성장할 것으로 보입니다.
만성 정맥 폐색 치료 산업 개요
만성 정맥 폐색 치료 시장은 반 고정적이며 여러 기업이 있습니다. 시장 개척 기업은 합병, 인수, 제휴, 공동개발 등 다양한 전략을 구사하여 신약과 신 디바이스를 개발하고 있습니다. 주요 진출기업으로는 Cardinal Health, Stryker, Cook Medical, Boston Scientific Corporation, Edward Lifesciences, AngioDynamics 등이 있습니다.
기타 혜택 :
- 엑셀 형식 시장 예측(ME) 시트
- 3개월간의 애널리스트 서포트
목차
제1장 서론
- 조사 전제조건 및 시장 정의
- 조사 범위
제2장 조사 방법
제3장 주요 요약
제4장 시장 역학
- 시장 개요
- 시장 성장 촉진요인
- 고도의 치료법 및 장치 가용성 증가
- 정맥 폐색성 질환의 유병률 증가
- 시장 성장 억제요인
- 치료에 따른 위험이나 합병증과 연관된 비용 의식
- Porter's Five Forces 분석
- 신규 참가업체의 위협
- 구매자 및 소비자의 협상력
- 공급기업의 협상력
- 대체품의 위협
- 경쟁 기업간 경쟁 관계의 강도
제5장 시장 세분화
- 치료 유형별
- 만성 심부정맥혈전증
- 하지정맥류
- 심부정맥부전
- 기타 치료 유형
- 제품 유형별
- 디바이스
- 치료제
- 지역별
- 북미
- 미국
- 캐나다
- 멕시코
- 유럽
- 독일
- 영국
- 프랑스
- 이탈리아
- 스페인
- 기타 유럽
- 아시아태평양
- 중국
- 일본
- 인도
- 호주
- 한국
- 기타 아시아태평양
- 중동 및 아프리카
- GCC
- 남아프리카
- 기타 중동 및 아프리카
- 남미
- 브라질
- 아르헨티나
- 기타 남미
- 북미
제6장 경쟁 구도
- 기업 프로파일
- Cardinal Health
- Stryker
- Cook Medical
- Boston Scientific Corporation
- Edward Lifesciences
- AngioDynamics
- SIGVARIS GROUP
- Johnson & Johnson Inc.(Janssen Global Services LLC)
- Penumbra Inc.
- Becton, Dickinson and Company
제7장 시장 기회 및 향후 동향
AJY 25.04.09The Chronic Venous Occlusions Treatment Market size is estimated at USD 8.43 billion in 2025, and is expected to reach USD 11.48 billion by 2030, at a CAGR of 6.38% during the forecast period (2025-2030).
Chronic venous occlusion treatment is used to treat chronic venous diseases. Venous occlusion is when veins are blocked, narrowed, or compressed by nearby structures such as clots, muscles, arteries, or other veins. This can result in blood pooling and flowing backward, causing pain and swelling in the area.
The high prevalence of venous occlusion diseases globally drives the market's growth. For instance, a study published in the International Wound Journal in May 2024 revealed that the mean prevalence of chronic venous diseases was 58.4%, and the prevalence of varicose veins was 22.1%. Such a high prevalence of disease at the global level propels the market's growth.
Major market players are increasingly making strategic moves, such as acquisitions, to bolster their product portfolios, particularly in minimally invasive devices for vascular disease treatment.
For instance, in May 2024, Siemens Healthineers, through its subsidiary Varian, finalized the acquisition of Innova Vascular. Innova Vascular, a prominent player in the medical device industry, specializes in cutting-edge, minimally invasive devices tailored for vascular disease treatment. The flagship product, the Laguna Thrombectomy System, is lauded for its exceptional flexibility, trackability, and diverse range of sizes.
These attributes enable healthcare providers to address venous thromboembolism across patient profiles and clot variations effectively. With this acquisition, Siemens Healthineers has significantly bolstered its offerings for blood clot treatments, setting the stage for substantial growth in this market during the forecast period.
Additionally, pharmaceutical companies are increasingly spearheading initiatives to raise awareness about treating venous insufficiency to propel market growth during the forecast period. For example, in July 2023, LES LABORATOIRES SERVIER rolled out a comprehensive campaign dedicated to venous disease awareness. By closely collaborating with healthcare professionals and patients, the company aims to deepen the understanding of venous insufficiency and enhance symptom management. These concerted efforts from pharmaceutical entities are poised to drive market growth in the coming years.
Thus, owing to the abovementioned factors, such as the high prevalence of venous diseases, increasing awareness of venous diseases, and the launch of new products, the growth of the market is the majority driving it. However, the cost compilations and risk associated with treatment may hinder the growth of the market.
Chronic Venous Occlusions Treatment Market Trends
The Deep Vein Insufficiency Segment is Expected to Drive the Market Growth During the Forecast Period
Chronic venous occlusion treatment therapeutics primarily consist of antibiotics and anticoagulants. These medications aim to either prevent or treat blood clots within blood vessels. Such clots pose severe risks, including stroke, heart attack, deep vein thrombosis, and pulmonary embolism. Anticoagulants prevent clot formation, while thrombolytics dissolve existing clots.
Effective anticoagulants for managing deep venous thrombosis encompass a range, from low molecular weight heparins (LMWHs) like enoxaparin, dalteparin, and tinzaparin to unfractionated heparin (UFH), factor Xa inhibitors, direct thrombin inhibitors, and vitamin K antagonists such as warfarin.
The segment's growth is propelled by the rising adoption of new oral anticoagulants tailored for chronic venous occlusion.
The advent of biosimilars is poised to intensify market competition, driving down the costs of chronic venous occlusion drugs. This cost reduction enhances patient accessibility, directly influencing the segment's growth. In September 2023, Fresenius Kabi Canada achieved public reimbursement approval for ELONOX, its enoxaparin biosimilar, across all Canadian provinces. ELONOX. Health Canada specifically endorsed ELONOX (enoxaparin sodium) for thromboembolic disorder prophylaxis, including deep vein thrombosis. Such reimbursement by health authorities contributes to increased accessibility and adaptability and further propels segmental growth.
The increasing focus on clinical studies targeting deep vein thrombosis is anticipated to be a significant driver for segmental growth. A case in point is Bayer AG, which, in February 2024, initiated a Phase II clinical trial for BAY3018250, an innovative anti-a2 antiplasmin (anti-a2ap) antibody, in DVT patients. The trial's results are expected to highlight the antibody's potential as a crucial treatment avenue for severe medical conditions. Buoyed by these findings, Bayer is keen to advance the antibody to the next phase of clinical trials, with a specific focus on DVT patients. Such initiatives hold promise for patients and bolster the segment's growth.
Therefore, the confluence of factors, including the rise of biosimilars, an uptick in clinical studies for DVT, and strategic moves by major players, like Bayer's new drug trials, is poised to drive significant growth in the segment in the coming years.
North America Dominates the Market During the Forecast Period
North America is poised to dominate the chronic venous occlusion treatment market. It is buoyed by its advanced healthcare infrastructure, high venous disease prevalence, and robust regulatory approvals for medical devices and drugs.
The region's market is witnessing growth, driven by the high prevalence of venous diseases. For instance, in April 2023, the National Institutes of Health (NIH) reported that over 25 million Americans suffer from varicose veins, with a staggering 6 million facing severe venous disease. This highlights the gravity of chronic venous conditions as a significant health concern. The rising burden of these conditions has driven the adoption of chronic venous occlusion treatments, fueling market growth.
Moreover, ongoing clinical trials, such as the VIVID trial in October 2023, which assessed the Duo venous stent system by Koninklijke Philips N.V. for nonmalignant iliofemoral occlusive disease, are set to propel the market further. The Duo system, comprising the Duo Hybrid and Duo Extend stents, caters to the diverse anatomical and mechanical needs of the iliofemoral venous segment. These trials to ensure device efficacy and safety are pivotal for the market's future growth.
Also, the introduction of generic drug variants, like Pharmascience Canada's Pms-rivaroxaban in November 2023, designed for deep vein thrombosis treatment, enhances market accessibility and adaptability. Such launches will drive the market's growth in the coming years.
Given the rising prevalence of cardiovascular and venous diseases, alongside the momentum from ongoing clinical trials and the launch of products, the chronic venous occlusion treatment market is set for substantial growth during the forecast period.
Chronic Venous Occlusions Treatment Industry Overview
The chronic venous occlusion treatment market is semi-consolidated, with several players. Companies in the market use diverse strategies such as mergers, acquisitions, partnerships, and collaborations to develop new drugs and devices. Some of the major players in the market include Cardinal Health, Stryker, Cook Medical, Boston Scientific Corporation, Edward Lifesciences, and AngioDynamics.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Availability of Advanced Treatments and Devices
- 4.2.2 Increasing Prevalence of Venous Occlusion Diseases
- 4.3 Market Restraints
- 4.3.1 Cost Consciousness Coupled with Risk and Complications Associated with Treatment
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD)
- 5.1 By Treatment Type
- 5.1.1 Chronic Deep Vein Thrombosis
- 5.1.2 Varicose Veins
- 5.1.3 Deep Vein Insufficiency
- 5.1.4 Other Treatment Types
- 5.2 By Product Type
- 5.2.1 Devices
- 5.2.2 Therapeutics
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia-Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Cardinal Health
- 6.1.2 Stryker
- 6.1.3 Cook Medical
- 6.1.4 Boston Scientific Corporation
- 6.1.5 Edward Lifesciences
- 6.1.6 AngioDynamics
- 6.1.7 SIGVARIS GROUP
- 6.1.8 Johnson & Johnson Inc. (Janssen Global Services LLC)
- 6.1.9 Penumbra Inc.
- 6.1.10 Becton, Dickinson and Company



