
|
시장보고서
상품코드
1682713
리포솜 개발 및 리포솜 제조 서비스 시장 : 제품 유형별, 사업 규모별, 최종사용자별, 주요 지역별 - 업계 동향과 세계 예측(- 2035년)Liposome Development and Liposome Manufacturing Services Market by Type of Product Formulation, Type of Scale of Operation, Type of End User, and Key Geographical Regions : Industry Trends and Global Forecasts, Till 2035 |
||||||
세계 리포솜 개발 및 리포솜 제조 서비스 시장 규모는 2035년까지 9.7%의 연평균 복합 성장률(CAGR)로 확대되어 23억 4,000만 달러 규모로 성장할 것으로 예상됩니다.
약물의 낮은 용해도와 그에 따른 생체이용률에 미치는 영향은 여전히 일부 의약품 후보물질에 대한 주요 관심사입니다. 최근 동향에 따르면, 시판 중인 약리학적 제제의 약 40%, 개발 중인 의약품 후보물질의 약 90%가 용해성 및 투과성 문제를 가지고 있으며, 이로 인해 생체 이용률이 저하되고 있습니다. 주목할 만한 것은 매년 많은 의약품이 낮은 생체 이용률과 수용성 관련 문제로 인해 시장에 출시되지 못하고 있다는 점입니다. 그 결과, 업계는 이 문제를 완화하기 위해 다양한 도구와 방법을 적극적으로 모색하고 있습니다. 치료 중재의 생체 이용률을 높이기 위해 채택된 다양한 접근법 중 리포좀 및 기타 지질 부형제는 복잡하지만 매우 유망한 치료제의 약물 운반체 역할을 할 수 있기 때문에 의약품 개발자들의 관심을 끌고 있습니다.
리포솜 제조는 매우 복잡하고 자본 집약적인 프로세스이며, 고도의 기술 프로세스(틈새 시장 및 전문적 전문 지식 필요), 인프라 제약, 생산 능력의 제약, 임상 등급 생산 달성의 어려움 등 다양한 과제를 안고 있습니다. 이러한 제제 생산에 수반되는 기술적 복잡성을 고려할 때, 개발자들은 점점 더 서비스 제공업체에 의존하고 있습니다. 리포좀 기반 치료제가 증가함에 따라 리포좀 개발 및 제조 서비스 시장은 가까운 미래에 안정적인 성장을 보일 것으로 예상됩니다.
현재 70개 이상의 기업이 다양한 규모의 리포좀 개발 및 제조 서비스를 제공할 것이라고 합니다.
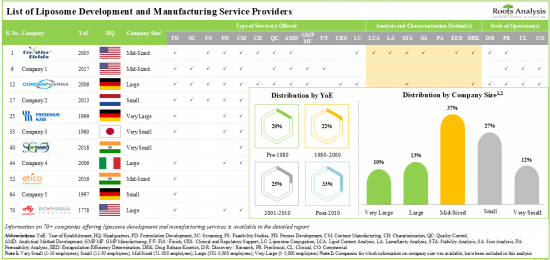
이해관계자들은 전문 지식을 활용하여 고활성 리포솜에 대한 다양한 서비스를 제공합니다. 서비스 제공업체의 약 60%가 리포솜의 안정성 분석 능력을 보유하고 있습니다. 리포솜과 관련된 700여 건의 특허가 다양한 이해관계자별로 출원되어 이 분야에서 탄생한 지적 재산을 보호하고 있습니다.
세계의 리포솜 개발 및 리포솜 제조 서비스 시장에 대해 조사했으며, 시장 개요와 함께 제품 유형별/사업 규모별/최종사용자별/주요 지역별 동향, 시장 진출 기업 프로파일 등의 정보를 전해드립니다.
목차
제1장 서문
제2장 주요 요약
제3장 서론
제4장 시장 구도 : 리포솜 개발 및 리포솜 제조 서비스 제공업체
제5장 기업 개요
제6장 임상시험 분석
제7장 출판물 분석
제8장 특허 분석
제9장 세계 이벤트 분석
제10장 아웃소싱 : GO/NO-GO 프레임워크
제11장 시장 예측과 기회 분석
제12장 결론
제13장 주요 인사이트
제14장 부록 1 : 표 형식 데이터
제15장 부록 2 : 기업 및 단체 리스트
LSH 25.03.27LIPOSOME DEVELOPMENT AND LIPOSOME MANUFACTURING SERVICES MARKET: OVERVIEW
As per Roots Analysis, the global liposome development and liposome manufacturing services market is estimated to grow to USD 2.34 billion by 2035, at a CAGR of 9.7% during the forecast period, till 2035.
The market sizing and opportunity analysis has been segmented across the following parameters:
Type Of Product Formulation
- Therapeutic
- Nutraceutical
Type of Scale of Operation
- Discovery / Research
- Preclinical
- Clinical and Commercial
Type of End User
- Pharmaceutical and Biotechnology,
- Cosmetics
- Food
- Agricultural
- Academic
- Other Industries
Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and North Africa
LIPOSOME DEVELOPMENT AND LIPOSOME MANUFACTURING SERVICES MARKET: GROWTH AND TRENDS
Low drug solubility and its corresponding impact on bioavailability have remained a primary cause of concern for several drug candidates. According to a recent study, approximately 40% of marketed pharmacological products and close to 90% of drug candidates under development, have been associated with solubility and / or permeability issues, resulting in poor bioavailability. It is worth noting that, every year, a large number of drugs fail to reach the market due to poor bioavailability and issues associated with aqueous solubility. As a result, the industry is actively seeking various tools / methods to mitigate this challenge. Amongst the different approaches employed to enhance the bioavailability of therapeutic interventions, liposomes and other lipidic excipients have garnered the attention of drug developers, owing to their ability to act as drug carriers for complex, albeit highly promising therapeutics.
Manufacturing of liposomes is a highly complex and capital-intensive process fraught with a range of challenges, including highly technical processes (which demand niche and specialized expertise), infrastructure limitations, capacity constraints, and challenges in achieving clinical-grade production. Given the technical complexities associated with the manufacturing of such formulations, developers are increasingly relying on service providers. With increase in the number of liposome based therapeutics, the liposome development and manufacturing services market is anticipated to witness a steady growth in the near future.
LIPOSOME DEVELOPMENT AND LIPOSOME MANUFACTURING SERVICES MARKET: KEY INSIGHTS
The report delves into the current state of the liposome development and liposome manufacturing services market and identifies potential growth opportunities within the industry. Some key findings from the report include:
1. Presently, over 70 players claim to offer a wide range of liposome development and manufacturing services across different scales of operation.
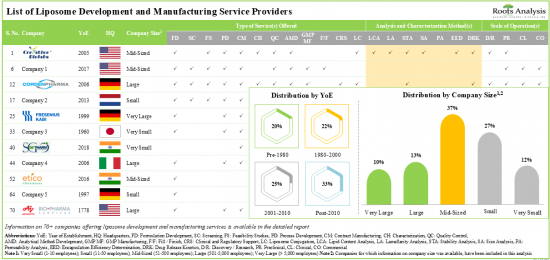
2. Leveraging their expertise, stakeholders offer a variety of services for a myriad of highly potent liposomes; around 60% of the service providers possess the capability for stability analysis of liposomes.
3. Close to 700 patents related to liposomes have been filed by various stakeholders in order to protect the intellectual property generated within this field.
4. The growing interest is also reflected by the events being organized globally; such platforms provide an opportunity to industry stakeholders / researchers to share ideas and develop a better understanding of liposomes.
5. 800+ clinical trials related to liposomes have been registered till date; the majority (38%) of these trials were / are being conducted across various clinical sites based in Europe.
6. Since 2017, more than 6,000+ articles focused on liposomes have been published in high-impact journals, highlighting the substantial efforts undertaken by researchers.
LIPOSOME DEVELOPMENT AND LIPOSOME MANUFACTURING SERVICES MARKET: KEY SEGMENTS
Therapeutic Formulations Occupy the Largest Share of the Liposome Development and Liposome Manufacturing Services Market
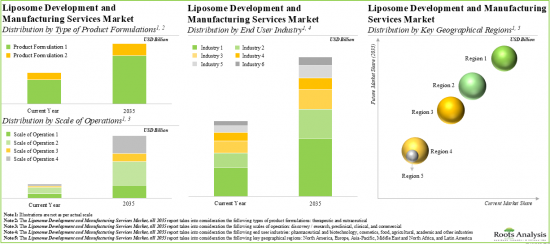
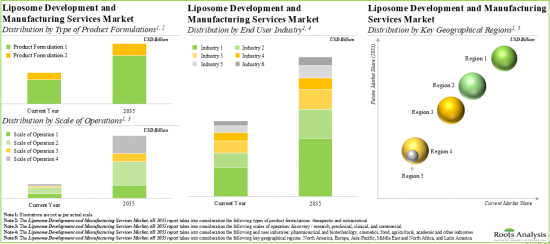
Based on the type of product formulation, the market is segmented into therapeutic and nutraceutical. Currently, the therapeutic formulation holds the maximum share of the liposome development and liposome manufacturing services market. It is worth highlighting that the liposome development and liposome manufacturing services market for nutraceuticals is likely to grow at a relatively higher CAGR.
Clinical Scale is Likely to Dominate the Liposome Development and Liposome Manufacturing Services Market
Based on the scale of operation, the market is segmented into discovery / research, preclinical, clinical and commercial. At present, the clinical scale captures the highest share of the liposome development and liposome manufacturing services market. It is worth highlighting that the discovery / research scale is to drive the market in the future with a relatively higher CAGR.
Pharmaceutical and Biotechnology Industry Occupies the Largest Share of the Liposome Development and Liposome Manufacturing Services Market
Based on the type of end-user, the market is segmented into pharmaceutical and biotechnology, cosmetics, food, agricultural, academic, and other industries. Currently, the pharmaceutical and biotechnology industry holds the maximum share of the liposome development and liposome manufacturing services market. This trend is unlikely to change in the foreseeable future.
North America Accounts for the Largest Share of the Market
Based on key geographical regions, the market is segmented into North America, Europe, Asia-Pacific, Middle East and North Africa, and Latin America. The majority share is expected to be captured by players based in North America. It is worth highlighting that, over the years, the market in Latin America is expected to grow at a higher CAGR.
Example Players in the Liposome Development and Liposome Manufacturing Services Market
- Baxter BioPharma Solutions
- Charles River Laboratories
- Evonik
- Fresenius Kabi
- GEA
- Intertek
- Fujifilm
LIPOSOME DEVELOPMENT AND LIPOSOME MANUFACTURING SERVICES MARKET: RESEARCH COVERAGE
- Market Sizing and Opportunity Analysis: The report features an in-depth analysis of the liposome development and manufacturing services market, focusing on key market segments, including [A] type of product formulation, [B] type of scale of operation, [C] type of end user, and [D] key geographical regions.
- Market Landscape: A comprehensive evaluation of liposome development and manufacturing service providers involved in the liposome development and manufacturing services market, considering various parameters, such as [A] year of establishment, [B] company size (in terms of number of employees), [C] region of headquarters, [D] types of method(s) for liposome preparation, [E] type of service(s) offered, [F] liposome bioconjugation target(s), [G] scalability, [H] liposome analysis and characterization method(s), [I] application(s) of liposomes, [J] scale of operation and [K] end user (s).
- Company Profiles: In-depth profiles of key industry players offering liposome development and manufacturing services across various geographies, focusing on [A] company overviews, [B] financial information (if available), [C] service portfolio, [D] recent developments and [E] an informed future outlook.
- Clinical Trial Analysis: Examination of completed, ongoing, and planned clinical studies of liposomes based on parameters like [A] trial registration year, [B] trial status, [C] trial registration year and patients enrolled, [D] trial phase, [E] type of sponsor / collaborator, [F] study design, [G] leading players (in terms of number of trials conducted), [H] disease indication(s), [I] popular therapeutic areas, [J] type of treatment, [K] emerging focus areas and [L] regional distribution of trials (in terms of number of trials conducted and trial status).
- Publication Analysis: A detailed review of scientific articles related to research on liposomes, based on several relevant parameters, such as [A] year of publication, [B] application area(s), [C] emerging focus areas and [D] the top journals (in terms of number of publications and impact factor).
- Patent Analysis: Detailed analysis of various patents filed / granted for liposomes based on [A] type of patent, [B] patent publication year, [C] granted patents and patent applications, [D] geography, [E] CPC symbols, [F] emerging focus areas, [G] type of organization, [H] leading players (in terms of number of patents granted / filed) and [I] patent characteristics. It also includes a patent benchmarking analysis and a detailed valuation analysis.
- Global Event Analysis: An analysis of recent events, covering webinars, conferences, seminars, workshops, symposiums and summits that were organized for stakeholders in this market, based on several relevant parameters, such as [A] year of event, [B] event platform, [C] type of event, [D] geography, [E] evolutionary trends in event agenda, [F] most active event organizers, [G] active industry and non-industry players (in terms of number of events that each company participated in), [H] seniority level of event speakers, [I] affiliated department of event speakers, [J] most active speakers (in terms of number of events) and [K] a geographical mapping of upcoming events.
- Outsourcing: Go / No-Go Framework: An insightful framework that emphasizes the key indicators and factors that needs to be considered by liposome developers to determine whether to manufacture their respective products in-house or outsource the manufacturing operation to contract service providers.
KEY QUESTIONS ANSWERED IN THIS REPORT
- How many companies are currently engaged in this market?
- Which are the leading companies in this market?
- What factors are likely to influence the evolution of this market?
- What is the current and future market size?
- What is the CAGR of this market?
- How is the current and future market opportunity likely to be distributed across key market segments?
REASONS TO BUY THIS REPORT
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- Stakeholders can leverage the report to gain a deeper understanding of the competitive dynamics within the market. By analyzing the competitive landscape, businesses can make informed decisions to optimize their market positioning and develop effective go-to-market strategies.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
ADDITIONAL BENEFITS
- Complimentary PPT Insights Packs
- Complimentary Excel Data Packs for all Analytical Modules in the Report
- 10% Free Content Customization
- Detailed Report Walkthrough Session with Research Team
- Free Updated report if the report is 6-12 months old or older
TABLE OF CONTENTS
1. PREFACE
- 1.1. Scope of the Report
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Key Questions Answered
- 1.5. Chapter Outlines
2. EXECUTIVE SUMMARY
3. INTRODUCTION
- 3.1. Chapter Overview
- 3.2. Introduction to Liposomes
- 3.3. Formation of Liposomes
- 3.4. Classification of Liposomes
- 3.5. Liposome as a Drug Delivery System
- 3.5.1. Liposomal Drug Delivery Platforms
- 3.5.2. Therapeutic Applications of Liposomes in Drug Delivery
- 3.6. Methods for Liposome Preparation
- 3.7. Post-Preparation Treatments
- 3.7.1. Freeze-Thawing
- 3.7.2. Freeze-Drying
- 3.8. Liposome Analysis and Characterization Methods
- 3.9. Pharmaceutical and Industrial Applications of Liposomes
- 3.9.1. Drug Delivery
- 3.9.2. Gene Delivery
- 3.9.3. Vaccine Delivery
- 3.9.4. Cancer Therapy
- 3.9.5. Agricultural Industry
- 3.9.6. Cosmetics
- 3.9.7. Food Industry
- 3.10. Challenges in Liposome Development and Manufacturing
- 3.11. Need for Outsourcing of Liposome Development and Manufacturing
- 3.12. Future Perspectives
4. MARKET LANDSCAPE: LIPOSOME DEVELOPMENT AND MANUFACTURING SERVICE PROVIDERS
- 4.1. Chapter Overview
- 4.2. Liposome Development and Manufacturing Service Providers Landscape
- 4.2.1. Analysis by Year of Establishment
- 4.2.2. Analysis by Company Size
- 4.2.3. Analysis by Region of Headquarters
- 4.2.4. Analysis by Company Size and Region of Headquarters
- 4.2.5. Analysis by Type of Method(s) Used for Liposome Preparation
- 4.2.6. Analysis by Type of Service(s) Offered
- 4.2.7. Analysis by Liposome Bioconjugation Target(s)
- 4.2.8. Analysis by Scalability
- 4.2.9. Analysis by Liposome Analysis and Characterization Method(s)
- 4.2.10. Analysis by Application(s) of Liposomes
- 4.2.11. Analysis by Type of Service(s) Offered, Application(s) and Product Formulation
- 4.2.12. Analysis by Scale of Operation
- 4.2.13. Analysis by End User(s)
- 4.2.14. Analysis by Scale of Operation and End User(s)
5. COMPANY PROFILES
- 5.1. Chapter Overview
- 5.2. Key Players Based in North America
- 5.2.1. Baxter BioPharma Solutions
- 5.2.1.1. Company Overview
- 5.2.1.2. Financial Information
- 5.2.1.3. Service Portfolio
- 5.2.1.4. Recent Developments and Future Outlook
- 5.2.2. Charles River Laboratories
- 5.2.2.1. Company Overview
- 5.2.2.2. Financial Information
- 5.2.2.3. Service Portfolio
- 5.2.2.4. Recent Developments and Future Outlook
- 5.2.1. Baxter BioPharma Solutions
- 5.3. Key Players Based in Europe
- 5.3.1. Evonik
- 5.3.1.1. Company Overview
- 5.3.1.2. Financial Information
- 5.3.1.3. Service Portfolio
- 5.3.1.4. Recent Developments and Future Outlook
- 5.3.2. Fresenius Kabi
- 5.3.2.1. Company Overview
- 5.3.2.2. Financial Information
- 5.3.2.3. Service Portfolio
- 5.3.2.4. Recent Developments and Future Outlook
- 5.3.3. GEA
- 5.3.3.1. Company Overview
- 5.3.3.2. Financial Information
- 5.3.3.3. Service Portfolio
- 5.3.3.4. Recent Developments and Future Outlook
- 5.3.4. Intertek
- 5.3.4.1. Company Overview
- 5.3.4.2. Financial Information
- 5.3.4.3. Service Portfolio
- 5.3.4.4. Recent Developments and Future Outlook
- 5.3.1. Evonik
- 5.4. Key Players Based in Asia-Pacific
- 5.4.1. Fujifilm
- 5.4.1.1. Company Overview
- 5.4.1.2. Financial Information
- 5.4.1.3. Service Portfolio
- 5.4.1.4. Recent Developments and Future Outlook
- 5.4.1. Fujifilm
6. CLINICAL TRIAL ANALYSIS
- 6.1. Chapter Overview
- 6.2. Scope and Methodology
- 6.3. Liposomes: Clinical Trial Analysis
- 6.3.1. Analysis by Trial Registration Year
- 6.3.2. Analysis by Trial Status
- 6.3.3. Analysis by Trial Registration Year and Trial Status
- 6.3.4. Analysis by Trial Registration Year and Patients Enrolled
- 6.3.5. Analysis by Trial Phase
- 6.3.6. Analysis by Trial Phase and Patients Enrolled
- 6.3.7. Analysis by Type of Sponsor / Collaborator
- 6.3.8. Analysis by Study Design
- 6.3.9. Most Active Industry Players: Analysis by Number of Trials
- 6.3.10. Most Active Non-Industry Players: Analysis by Number of Trials
- 6.3.11. Most Popular Disease Indication(s): Analysis by Number of Trials
- 6.3.12. Most Popular Therapeutic Area(s): Analysis by Number of Trials
- 6.3.13. Analysis by Type of Treatment
- 6.3.14. Word Cloud Analysis: Emerging Focus Areas
- 6.3.15. Analysis by Top Indication(s) and Trial Phase
- 6.3.16. Analysis by Top Indication(s) and Leading Industry Players
- 6.3.17. Geographical Analysis by Number of Clinical Trials
- 6.3.18. Geographical Analysis by Number of Patients Enrolled
7. PUBLICATION ANALYSIS
- 7.1. Chapter Overview
- 7.2. Scope and Methodology
- 7.3. Liposomes: Recent Publications
- 7.3.1. Analysis by Year of Publication
- 7.3.2. Analysis by Application Area(s)
- 7.3.3. Word Cloud Analysis: Emerging Focus Areas
- 7.3.4. Key Journals: Analysis by Number of Publications
- 7.3.5. Key Journals: Analysis by Impact Factor
- 7.3.6. Key Journals: Analysis by Year-wise Number of Publications
8. PATENT ANALYSIS
- 8.1. Chapter Overview
- 8.2. Scope and Methodology
- 8.3. Liposomes: Patent Analysis
- 8.3.1. Analysis by Patent Publication Year
- 8.3.2. Analysis by Granted Patents and Patent Applications
- 8.3.3. Analysis by Geography
- 8.3.4. Analysis by CPC Symbols
- 8.3.5. Word Cloud Analysis: Emerging Focus Areas
- 8.3.6. Analysis by Type of Organization
- 8.3.7. Leading Industry Players: Analysis by Number of Patents
- 8.3.8. Leading Non-Industry Players: Analysis by Number of Patents
- 8.3.9. Leading Individual Assignees: Analysis by Number of Patents
- 8.4. Liposomes: Patent Benchmarking Analysis
- 8.4.1. Analysis by Patent Characteristics
- 8.5. Liposomes: Patent Valuation Analysis
- 8.6. Leading Patents: Analysis by Number of Citations
9. GLOBAL EVENT ANALYSIS
- 9.1. Chapter Overview
- 9.2. Scope and Methodology
- 9.3. Liposome Development and Manufacturing: Global Events Analysis
- 9.3.1. Analysis by Year of Event
- 9.3.2. Analysis by Event Platform
- 9.3.3. Analysis by Type of Event
- 9.3.4. Analysis by Year of Event and Type of Event
- 9.3.5. Analysis by Geography
- 9.3.6. Word Cloud Analysis: Evolutionary Trends in Event Agenda / Key Focus Area
- 9.3.7. Most Active Event Organizers
- 9.4. Liposome Development and Manufacturing: Speaker Analysis
- 9.4.1. Most Active Industry Players: Analysis by Number of Events
- 9.4.2. Most Active Non-Industry Players: Analysis by Number of Events
- 9.4.3. Analysis by Seniority Level of Event Speakers
- 9.4.4. Analysis by Affiliated Department of Event Speakers
- 9.4.5. Most Active Speakers: Analysis by Number of Events
- 9.5. Geographical Mapping of Upcoming Events
- 9.6. Concluding Remarks
10. OUTSOURCING: GO / NO-GO FRAMEWORK
- 10.1. Chapter Overview
- 10.2. Outsourcing: Go / No-Go Framework
- 10.3. Liposome-based Therapeutic Developers Outsourcing: Go / No-Go Framework
- 10.3.1. Key Parameters and Assumptions
- 10.3.2. Methodology
- 10.3.3. Results and Interpretations
- 10.3.3.1. Very Small Companies
- 10.3.3.2. Small Companies
- 10.3.3.3. Mid-Sized Companies
- 10.3.3.4. Large Companies
- 10.3.3.5. Very Large Companies
11. MARKET FORECAST AND OPPORTUNITY ANALYSIS
- 11.1. Chapter Overview
- 11.2. Key Assumptions and Methodology
- 11.3. Global Liposome Development and Manufacturing Services Market, till 2035
- 11.3.1. Liposome Development and Manufacturing Services Market: Distribution by Type of Product Formulation, Current Year and 2035
- 11.3.1.1. Liposome Development and Manufacturing Services Market for Therapeutics, till 2035
- 11.3.1.2. Liposome Development and Manufacturing Services Market for Nutraceuticals, till 2035
- 11.3.2. Liposome Development and Manufacturing Services Market: Distribution by Scale of Operation, Current Year and 2035
- 11.3.2.1. Liposome Development and Manufacturing Services Market for Discovery / Research Scale of Operation, till 2035
- 11.3.2.2. Liposome Development and Manufacturing Services Market for Preclinical Scale of Operation, till 2035
- 11.3.2.3. Liposome Development and Manufacturing Services Market for Clinical Scale of Operation, till 2035
- 11.3.2.4. Liposome Development and Manufacturing Services Market for Commercial Scale of Operation, till 2035
- 11.3.3. Liposome Development and Manufacturing Services Market: Distribution by End User, Current Year and 2035
- 11.3.3.1. Liposome Development and Manufacturing Services Market for Pharmaceutical and Biotechnology Industry, till 2035
- 11.3.3.2. Liposome Development and Manufacturing Services Market for Food Industry, till 2035
- 11.3.3.3. Liposome Development and Manufacturing Services Market for Agricultural Industry, till 2035
- 11.3.3.4. Liposome Development and Manufacturing Services Market for Cosmetic Industry, till 2035
- 11.3.3.5. Liposome Development and Manufacturing Services Market for Academics, till 2035
- 11.3.3.6. Liposome Development and Manufacturing Services Market for Other Industries, till 2035
- 11.3.4. Liposome Development and Manufacturing Services Market: Distribution by Key Geographical Regions, Current Year and 2035
- 11.3.4.1. Liposome Development and Manufacturing Services Market in North America, till 2035
- 11.3.4.2. Liposome Development and Manufacturing Services Market in Europe, till 2035
- 11.3.4.3. Liposome Development and Manufacturing Services Market in Asia-Pacific, till 2035
- 11.3.4.4. Liposome Development and Manufacturing Services Market in Middle East and North Africa, till 2035
- 11.3.4.5. Liposome Development and Manufacturing Services Market in Latin America, till 2035
- 11.3.1. Liposome Development and Manufacturing Services Market: Distribution by Type of Product Formulation, Current Year and 2035

















