
|
시장보고서
상품코드
1920864
의약품 수탁 제조 시장(제5판) : 동향과 예측(-2035년) - 제품 유형별, 원료의약품 유형별, 원료의약품 효력별, FDF별, 제형별, 경구 고형제별, 최종사용자별, 지역별, 주요 기업별Pharmaceutical Contract Manufacturing Market (5th Edition): Trends and Forecast Till 2035 - Distribution by Type of Product Manufactured, Type of API, API Potency, FDF, Dosage Form, Oral Solid, End User, Geographical Regions and Key Players |
||||||
의약품 위탁생산 시장 : 개요
Roots Analysis의 조사에 따르면, 의약품 위탁생산 시장 규모는 2035년까지 예측 기간 동안 CAGR 4.5%로 성장하여 현재 1,003억 달러에서 2035년까지 1,554억 달러에 달할 것으로 추정됩니다.
의약품 위탁생산 시장 : 성장과 동향
위탁생산은 특정 개발 및/또는 생산 업무를 외부 서비스 제공업체에 위탁하는 것을 말합니다. 이는 한 기업이 다른 기업의 라벨이나 브랜드를 사용하여 제품을 개발하는 것을 의미합니다. 제약기업이 가장 많이 위탁하는 업무는 제제 설계, 제형 개발, 임상/상업 생산, 포장, 물류 등이 있습니다.
흥미롭게도, 현대의 신약 후보물질 파이프라인은 점점 더 복잡해지고 있으며, 전문적인 시설, 장비 및 운영 지식이 필요합니다. 만성질환을 앓고 있는 사람들의 수가 크게 증가하면서 혁신적인 의약품에 대한 지속적인 수요가 발생하고 있습니다. 이러한 수요 증가에 대응하기 위해 다양한 중소기업과 일부 대형 제약사들은 생산 활동을 위탁 서비스 제공업체에 아웃소싱하기 시작했습니다. 기술 개발 및 혁신적인 치료법에 대한 시장의 요구도 그 발전을 지속시키고 있습니다.
주목할 만한 점은 CMO(위탁생산기관)가 맞춤형 치료법, 지속적인 제조 공정, 디지털 헬스 통합 기술 도입을 통해 새로운 트렌드에 적응하고 있다는 점입니다. 혁신, 지속가능성, 팀워크의 채택으로 제약 CMO는 의료 분야에 큰 영향을 미치고 향후 몇 년 동안 괄목할만한 성장을 이룰 것으로 예측됩니다.
성장 촉진요인 : 시장 확대의 전략적 기반
바이오의약품, 단일클론항체, ADC(항체-약물 복합체), GLP-1 치료제 등 새로운 치료법에 대한 수요가 증가하면서 고도의 역량을 갖춘 전문 CMO에 대한 아웃소싱을 촉진하고 있습니다. 또한, 주요 바이오의약품의 특허만료와 오리지널 의약품의 가격 책정 문제로 인해 제약사들은 비용 효율적인 위탁생산 전략을 추구할 수 밖에 없습니다. 또한, 만성질환 증가율과 신약에 대한 수요는 확장 가능한 제조 능력과 신속한 시장 출시를 실현하는 CMO에 대한 의존도를 높이고 있습니다.
시장 과제: 발전을 가로막는 심각한 장벽들
의약품 위탁생산 시장은 FDA, EMA 등 규제 당국의 엄격한 규제 준수, 생산 기술 비용 상승 등 성장을 저해하는 심각한 장벽에 직면해 있습니다. 이러한 장벽으로 인해 수탁 제조업체는 높은 컴플라이언스 비용을 부담하고 있습니다. 또한, 세계 무역 불안정으로 인해 원료의약품(API) 및 원자재 공급망에 혼란이 발생하고, 미국 및 유럽에서의 가격 압박이 가중되면서 비용 관리의 어려움이 가중되고 있습니다.
의약품 위탁생산 시장 : 주요 조사 결과
본 보고서는 의약품 위탁생산 시장의 현황을 상세하게 분석하고, 업계 내 잠재적인 성장 기회를 파악합니다. 주요 조사 결과는 다음과 같습니다.
- 현재 시장 상황에서는 전 세계적으로 약 510개 기업이 의약품 위탁생산 서비스를 제공한다고 주장하고 있으며, 그 중 50%가 아시아태평양에 본사를 두고 있습니다.
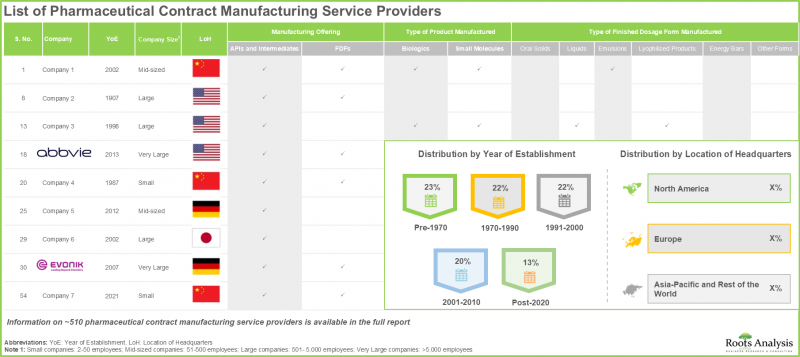
- 의약품 위탁생산 기업의 대다수(약 90%)가 최종 제형 제조 서비스를 제공하고 있으며, 75% 이상의 기업이 충진, 포장, 라벨링 서비스를 제공합니다.
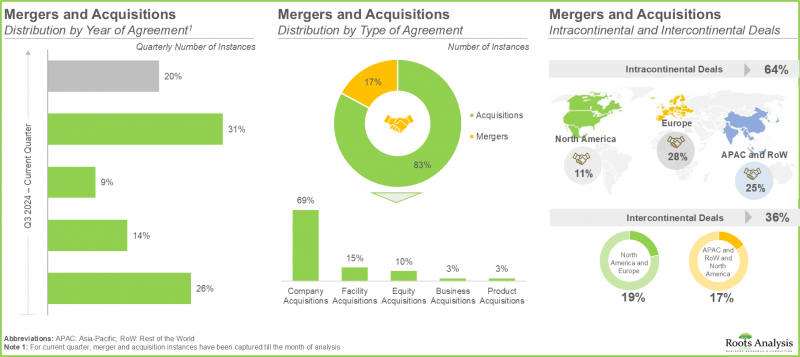
- 인수합병(M&A)를 통해 기업은 원스톱 숍으로서의 지위를 확립하고, 끊임없이 진화하는 시장 상황에서 경쟁 우위를 구축할 수 있습니다.
- 저분자 의약품에 대한 수요 증가에 대응하기 위해 CMO들은 기존 생산 능력과 기술력 확충에 많은 투자를 해왔습니다. 최근 이러한 추세는 유럽에서 가장 두드러지게 나타나고 있습니다.
- 전 세계 의약품 위탁생산 능력은 세계 각지의 다양한 시설에 고르게 분포되어 있습니다. 특히, 대규모 및 초대형 기업이 총 생산능력의 85% 이상을 차지하고 있습니다.
- 자체 생산시설 설립 및 유지에 따른 고가의 설비투자 및 운영비용을 절감하기 위해 의약품 위탁생산 수요는 크게 증가할 것으로 예측됩니다.
- 현재 의약품 위탁생산 시장 점유율의 대부분은 북미가 차지하고 있습니다. 이는 선진적인 의료 인프라가 구축되어 있어 각 사가 광범위한 조사를 수행할 수 있기 때문입니다.
- 경구용 고형제 하위 부문은 비용 효율성, 투여의 용이성, 환자의 높은 순응도로 인해 올해 전체 시장의 대부분을 차지할 것으로 추정됩니다.
- 북미는 전체 의약품 위탁생산 시장에서 큰 비중을 차지할 것으로 예상되며, 이러한 추세는 앞으로도 지속될 것으로 예측됩니다.
- 미국의 의약품 위탁생산 시장은 연평균 4.3%의 성장률을 보일 것으로 예측됩니다. 이는 저분자 화합물에 대한 수요가 증가함에 따라 고도의 제조 능력에 대한 필요성이 증가하고 있기 때문입니다.
- 저분자 화합물에 대한 수요 증가와 더불어 인수합병 및 사업 확장 움직임이 활발해짐에 따라 의약품 CMO 시장은 향후 몇 년 동안 큰 폭의 성장을 이룰 것으로 예측됩니다.
의약품 위탁생산 시장
시장 규모 및 기회 분석은 다음과 같은 매개 변수별로 분류됩니다.
제조 제품 유형별
- 원료의약품(API) 및 중간체
- FDF(완제의약품)
원료의약품 유형별
- 원료의약품용 API
- 제네릭 API
원약 효능별
- 저효능 API
- 고효능 API
FDF 유형별
- 선발품 FDF
- 제네릭 FDF
제형별
- 경구용 고형제
- 액제
- 에멀젼
- 기타 제형
경구용 고형제 유형별
- 정제
- 캡슐
- 기타
제공되는 포장 유형별
- 항아리
- 블리스터 포장
- 바이알
- 프리필드 시린지
- 카트리지
- 앰플
- 경구용 액제용 병
- 기타
사업 규모별
- 임상
- 상업
최종 사용자별
- 소규모
- 중규모
- 대기업 및 초대형 기업
지역별
- 북미
- 미국
- 캐나다
- 유럽
- 벨기에
- 스위스
- 영국
- 독일
- 아일랜드
- 네덜란드
- 이탈리아
- 프랑스
- 스웨덴
- 기타 유럽
아시아태평양 및 기타 지역
- 중국
- 인도
- 일본
- 한국
- 기타 아시아태평양 및 기타 국가
의약품 위탁생산 시장 : 주요 부문
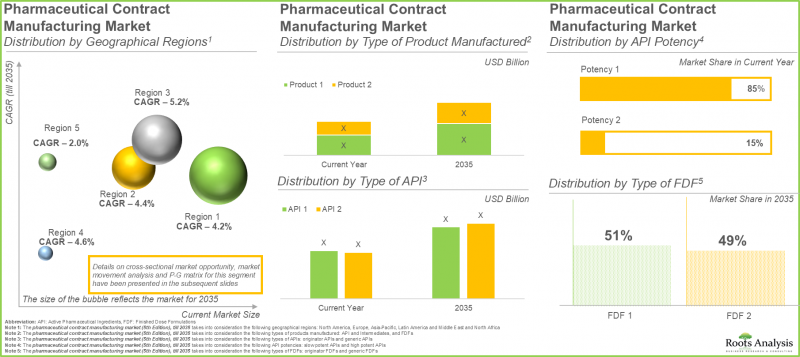
저효능 API 제조가 의약품 위탁생산 시장 주도
현재 저효능 API는 전체 시장의 85%를 차지하고 있습니다. 이는 주로 대규모 생산과 확장 가능한 제조 방식에 기인합니다. 또한, 이러한 API는 제약회사에서 당뇨병, 감염성 질환 등 다양한 질환의 치료에 활용되고 있습니다. 이러한 광범위한 사용으로 인해 전 세계적으로 지속적으로 높은 수요가 발생하고 있습니다. 특히, 고효능 API 부문은 더 빠른 속도로 확대될 것으로 예측됩니다. 표적치료와 정밀의료에 대한 수요 증가가 이러한 확대를 견인하고 있으며, 이러한 치료법은 강력한 치료 효과로 인해 최소 용량으로도 매우 효과적인 것으로 입증되었기 때문입니다.
의약품 위탁생산 분야에서는 경구용 고형제제가 압도적인 수요를 주도하고 있습니다.
경구용 고형제 제조 분야는 의약품 위탁생산 시장 전체 매출의 약 55%를 차지하고 있습니다. 이는 저비용, 환자 편의성, 대량 생산의 효율성 때문입니다. 향후 예측 기간 동안 의약품 위탁생산 시장에서 액상제제 카테고리가 더 큰 성장세를 보일 것으로 예측됩니다.
북미가 의약품 위탁생산 분야 주도
북미는 의약품 위탁생산 시장을 선도하고 있으며, 전체 매출의 약 45%를 차지하고 있습니다. 고도로 발달된 제약 부문, 탄탄한 규제 시스템, 그리고 세계 유수의 제약회사들의 존재가 이 지역의 제약 위탁생산업체에 대한 수요를 견인하고 있습니다.
의약품 수탁생산 시장의 대표적인 진출기업들
- Albemarle
- Aspen Pharmacare
- Bausch Health Sciences
- Catalent
- Delpharm
- Eurofins Scientific
- Evonik Industries
- Fareva
- Fresenius Kabi
- Intas Pharmaceuticals
- Lonza
- Micro Labs
- Nipro Patch
- Patheon
- PiSA Farmaceutica
- Recipharm
- Sandoz
- West Pharmaceutical Services
- Wockhardt
- WuXi AppTec
의약품 위탁생산 시장 : 조사 범위
- 시장 규모 및 기회 분석 : 이 보고서는 의약품 위탁생산 시장에 대해 주요 시장 세분화에 초점을 맞춘 상세한 분석을 제공합니다. 본 보고서는(A) 제품 유형,(B) 원료 유형,(C) 원료 효능,(D) FDF 유형,(E) 제형,(F) 경구용 고형제 유형,(G) 제공되는 포장 유형,(H) 사업 규모,(I) 최종 사용자,(J) 지리적 지역 등 주요 시장 부문에 초점을 맞춘 상세한 분석을 제공합니다. 을 제공합니다.
- 시장 현황: 저분자 의약품 위탁생산 서비스를 제공하는 기업의 현황에 대해 상세하게 소개합니다. 여기에는(A) 설립연도,(B) 제조시설 위치,(C) 제공 서비스 유형,(D) 제조하는 의약품 유형,(E) 사업 규모,(F) 제공 서비스 유형,(G) 제조하는 최종 제형 유형,(H) 제공되는 포장 유형 등이 포함됩니다.
- 기업 프로파일: 북미, 유럽, 아시아태평양의 펩타이드 치료제 제조 시장에 진출한 주요 기업들에 대해(A) 설립연도,(B) 본사 소재지,(C) 제품 포트폴리오,(D) 최근 동향,(E) 향후 전망에 따른 상세한 프로파일을 제공합니다.
- 자체 생산과 외주 생산의 판단 기준: 의약품 개발 기업이 자사 제품을 자체 생산할 것인지, CMO(위탁생산기관)의 서비스를 이용할 것인지를 결정할 때 고려해야 할 다양한 요소들을 강조한 정성적 분석.
- 인수합병(M&A) : 이 분야의 다양한 인수합병을(A) 합의 연도,(B) 합의 형태,(C) 지역,(D) 가장 활발한 진출기업(합의 건수 기준), 소유권 변경 매트릭스, 인수 합의의 주요 가치 동인 등 여러 관련 파라미터를 기준으로 상세하게 분석합니다.
- 최근 확장 동향 : 의약품 위탁생산 분야의 확장 사례에 대한 심층 분석. 확장 시기, 확장 현황, 확장 형태, 시설 위치, 투자 금액, 제조 의약품 유형, 서비스 제공 형태, 주요 진출 기업 등 관련 파라미터를 기준으로 분석.
- 지역별 생산능력 분석 : 북미, 유럽, 아시아태평양, 기타 지역(ROW)의 의약품 위탁생산 시설의 지역별 생산능력에 대한 상세한 분석.
- 생산능력 분석 : 공개된 정보를 바탕으로 이해관계자가 보고한 데이터를 바탕으로 의약품 제조의 총 설비 용량을 추정합니다. A) 제조업체 규모(소형, 중형, 대형, 초대형) 및(B) 지역(북미, 유럽, 아시아태평양, 기타 지역)을 기준으로 가용 생산 능력의 분포를 파악할 수 있습니다.
- 수요 분석 : 저분자 의약품의 연간 수요에 대한 정보에 기반한 추정치를(A) 사업 규모,(B) 원료의약품 유형,(C) 원료의약품 효능,(D) 지역 등 여러 관련 매개변수에 걸쳐 제시합니다.
- 시장 영향 분석 : 세계 의약품 위탁생산 시장의 성장에 영향을 미칠 수 있는 요인에 대한 상세한 분석입니다. 또한,(A) 주요 촉진요인,(B) 잠재적 제약 요인,(C) 새롭게 떠오르는 기회,(D) 기존 과제 파악 및 분석이 특징입니다.
목차
제1장 서문
제2장 조사 방법
제3장 시장 역학
제4장 거시경제 지표
제5장 주요 요약
제6장 서론
- 본 장의 개요
- 써드파티 제조업체 타입
- 의약품 수탁 제조 개요
- 의약품 수탁 제조 진화
- 제약 업계 아웃소싱 필요성
- 의약품 수탁 제조업계 최근 동향
- 계약 제조 조직이 제공하는 서비스
- 계약 제조 조직을 선택할 때에 고려해야 할 중요 점
- 의약품 제조업무 아웃소싱에 수반하는 리스크와 과제
- 향후 전망
제7장 의약품 수탁 제조 조직 : 규제 상황
- 본 장의 개요
- 북미 규제 상황
- 유럽 규제 상황
- 아시아태평양과 세계 기타 지역 규제 상황
- 규제기관으로부터 승인 취득 상황별 의약품 수탁 제조 조직 분석
- 규제 상황 : 지역 벤치마크 분석
제8장 의약품 수탁 제조 조직 : 시장 구도
- 본 장의 개요
- 의약품 수탁 제조 조직 : 시장 구도
제9장 기업 개요 주요 시장 진출기업
- 본 장의 개요
- 북미 의약품 수탁 제조 기관
- Albemarle
- Bausch Health Services
- Catalent
- Patheon
- PiSA Farmaceutica
- West Pharmaceutical Services
- 유럽 의약품 수탁 제조 기관
- Delpharm
- Eurofins Scientific
- Evonik Industries
- Fareva
- Fresenius Kabi
- Lonza
- Recipharm
- Sandoz
- 아시아태평양 및 기타 지역 의약품 수탁 제조 기관
- Aspen Pharmacare
- Intas Pharmaceuticals
- Micro Labs
- Nipro Patch
- WuXi AppTec
- Wockhardt
제10장 제조인가 구입인가 의사 의사결정 프레임워크
- 본 장의 개요
- 전제와 주요 파라미터
- 의약품 수탁 제조업체 : 제조인가 구입인가 의사결정
- 결론
제11장 합병과 인수
- 본 장의 개요
- 인수합병(M&A) 유형
- 의약품 수탁 제조 조직 : 합병과 인수
제12장 최근 확장
- 본 장의 개요
- 확장 유형
- 의약품 수탁 제조 조직 : 최근 확대
제13장 지역 능력 분석
- 본 장의 개요
- 전제와 주요 파라미터
- 의약품 수탁 제조 시설 전체상
제14장 용량 분석
제15장 수요 분석
제16장 시장 영향 분석 : 촉진요인, 억제요인, 기회, 과제
제17장 세계의 의약품 수탁 제조 시장
제18장 의약품 수탁 제조 시장(제품 유형별)
제19장 의약품 수탁 제조 시장(원료의약품 유형별)
제20장 의약품 수탁 제조 시장(원료의약품 효력 별)
제21장 의약품 수탁 제조 시장(FDF 유형별)
제22장 의약품 수탁 제조 시장(제형별)
제23장 의약품 수탁 제조 시장(경구 고형제 별)
제24장 의약품 수탁 제조 시장(포장 형태별)
제25장 의약품 수탁 제조 시장(사업 규모별)
제26장 의약품 수탁 제조 시장(최종사용자별)
제27장 의약품 수탁 제조 시장(지역별)
제28장 의약품 수탁 제조 시장 : 북미
제29장 의약품 수탁 제조 시장 : 유럽
제30장 의약품 수탁 제조 시장 : 아시아태평양
제31장 의약품 수탁 제조 시장 : 기타 지역
제32장 의약품 수탁 제조 시장(주요 기업별)
제33장 사례 연구 : 저분자약과 고분자약/치료법 비교
제34장 결론
제35장 경영진 인사이트
제36장 부록 I : 표 데이터
제37장 부록 II : 기업 및 조직 리스트
LSH 26.02.19Pharmaceutical Contract Manufacturing Market: Overview
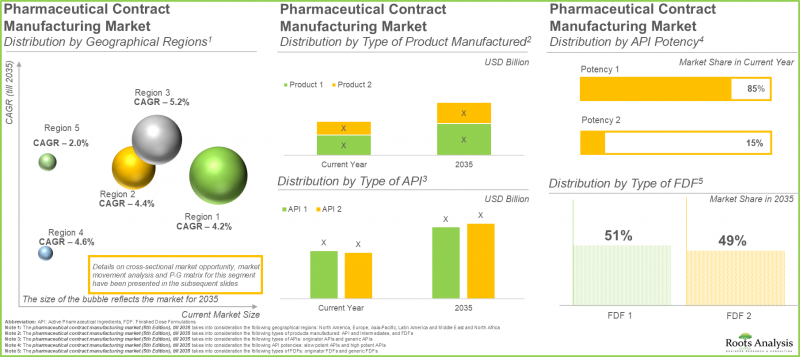
As per Roots Analysis, the pharmaceutical contract manufacturing market is estimated to grow from USD 100.3 billion in the current year to USD 155.4 billion by 2035, at a CAGR of 4.5% during the forecast period, till 2035.
Pharmaceutical Contract Manufacturing Market: Growth and Trends
Contract manufacturing involves outsourcing specific development and / or production tasks to an external service provider. This entails the development of products by one firm, using the label or brand of a different firm. The most frequently outsourced tasks by pharmaceutical companies encompass formulation design, dosage form development, clinical / commercial manufacturing, packaging, and logistics.
Interestingly, the contemporary pipeline of drug candidates is becoming increasingly intricate, necessitating specialized facilities, equipment, and operational knowledge. The number of individuals affected by chronic illnesses has also risen considerably, leading to a continuous need for innovative medications. To address this increasing demand, various smaller companies and some large pharmaceutical firms have started outsourcing their production activities to contract service providers. Technological developments and market need for innovative treatments have also persisted in their development.
Notably, CMOs are adapting to new trends by incorporating customized therapies, ongoing manufacturing methods, and techniques for digital health integration. Through adopting innovation, sustainability, and teamwork, pharmaceutical CMOs will greatly impact the healthcare field and witness notable growth in the coming years.
Growth Drivers: Strategic Enablers of Market Expansion
Increasing demand for biologics, monoclonal antibodies, and novel therapies such as ADCs and GLP-1 medications drive outsourcing to expert CMOs with enhanced capabilities. Further, expiring patents of major biologics and pricing challenges for innovator medications necessitates pharmaceutical firms to pursue cost-effective contract manufacturing strategies. In addition, rising rates of chronic illnesses and the demand for new medications boost dependence on CMOs for scalable manufacturing and quicker time-to-market.
Market Challenges: Critical Barriers Impeding Progress
The pharmaceutical contract manufacturing market faces significant hurdles that hinder its growth including stringent regulatory compliance from regulatory bodies like the FDA and EMA, coupled with rising costs of production technologies. These hurdles burden CMOs with high compliance expenses. Further, global trade instability disrupts supply chains for APIs and raw materials, while pricing pressures in the US and Europe intensify cost management issues.
Pharmaceutical Contract Manufacturing Market: Key Insights
The report delves into the current state of the pharmaceutical contract manufacturing market and identifies potential growth opportunities within industry. Some key findings from the report include:
- The current market landscape features the presence of close to 510 players that claim to offer contract manufacturing services for pharmaceuticals across the world; 50% of these are headquartered in Asia-Pacific.
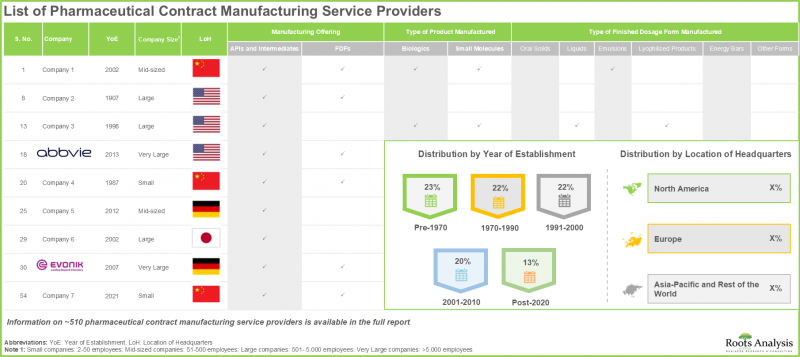
- Majority (~90%) of the pharmaceutical contract manufacturers offer services for producing finished dose formulations; further, >75% players offer services for fill finish / packaging and labeling.
- M&As allow companies to establish themselves as one-stop shops and build a competitive edge in the steadily evolving market landscape.
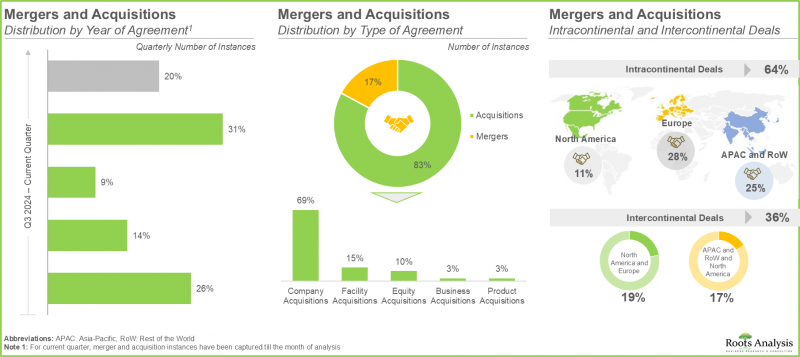
- In order to meet the rising demand for small molecules, CMOs have made significant investments in expanding their existing capacities and capabilities; in the recent past, this trend was most pronounced in Europe.
- The global pharmaceutical contract manufacturing capacity is well distributed across different facilities worldwide; notably, large and very large players account for more than 85% of the total capacity.
- In order to reduce high capital investments and operational costs associated with setting up and maintaining an in-house production facility, the demand for pharmaceutical contract manufacturing is anticipated to rise significantly.
- Currently, majority of the pharmaceutical contract manufacturing market share is captured by North America; this can be attributed to the advanced healthcare infrastructure, enabling players to conduct extensive research.
- The oral solids sub-segment is estimated to capture majority of the overall market share in the current year, owing to their cost-efficiency, ease of administration and high patient compliance.
- North America is expected to capture a significant market share of the overall pharmaceutical contract manufacturing market; this trend is unlikely to change in the future.
- Pharmaceutical contract manufacturing market in the US is expected to grow at CAGR of 4.3%; this is due to the increasing demand for small molecules, consequently fueling the need for advanced manufacturing capabilities.
- Given the rising demand for small molecules, along with the rise in mergers and acquisitions, and expansion activity, the pharmaceutical CMO market is likely to experience substantial growth in the coming years.
Pharmaceutical Contract Manufacturing Market
The market sizing and opportunity analysis has been segmented across the following parameters:
By Type of Product Manufactured
- API & Intermediates
- FDF
By Type of API
- Originator API
- Generic API
By API Potency
- Low Potent API
- High Potent API
By Type of FDF
- Originator FDF
- Generic FDF
By Dosage Form
- Oral Solids
- Liquids
- Emulsions
- Other Dosage Forms
By Type of Oral Solid
- Tablets
- Capsules
- Others
By Type of Packaging Offered
- Bottles
- Blister Packs
- Vials
- Prefilled Syringes
- Cartridges
- Ampoules
- Oral Liquid Bottles
- Others
By Scale of Operation
- Clinical
- Commercial
By End User
- Small
- Mid-sized
- Large and Very Large
By Geographical Regions
- North America
- US
- Canada
- Europe
- Belgium
- Switzerland
- UK
- Germany
- Ireland
- Netherlands
- Italy
- France
- Sweden
- Rest of Europe
Asia-Pacific and Rest of the World
- China
- India
- Japan
- Korea
- Rest of Asia-Pacific and Other Countries
Pharmaceutical Contract Manufacturing Market: Key Segments
The Manufacturing of Low Potent APIs Predominates the Pharmaceutical Contract Manufacturing Market
At present, low potent APIs account for 85% of the total market share. This is mainly due to their large-scale manufacturing and scalable methods. In addition, these APIs are utilized by pharmaceutical companies for treating different illnesses, including diabetes and infectious diseases. This extensive usage results in persistently elevated demand worldwide. Notably, the segment of highly potent APIs is expected to expand at a quicker rate. The increasing need for targeted treatments and precision medications fuels this expansion, as these therapies prove to be very effective even at minimal doses because of their strong therapeutic benefits.
Oral Solid Dosage Forms Dominate in the Pharmaceutical Contract Manufacturing Sector with Unmatched Demand
The oral solid dosage manufacturing sector represents approximately 55% of the total revenue in the pharma contract manufacturing market. This results from their low cost, convenience for patients, and effectiveness in mass production. In the future, the liquids category is expected to demonstrate greater growth in the pharmaceutical contract manufacturing market throughout the forecast .
North America Dominates the Pharmaceutical Contract Manufacturing Domain
North America leads the pharmaceutical contract manufacturing market, capturing approximately 45% of total revenue. The presence of a highly developed pharmaceutical sector, robust regulatory systems, and established leading global drug firms is driving the demand for pharmaceutical contract manufacturers in this region.
Primary Research Overview
The opinions and insights presented in the market report were also influenced by discussions held with senior stakeholders in the industry. The market report includes detailed transcripts of interviews conducted with the following individuals:
- President and Chief Executive Officer, Corporate Strategy and Business Development, Mid-sized Company, US
- Former Chief Executive Officer, Large Company, Switzerland
- Former Vice President Business Development, Marketing, and IP, Mid-sized Company, Israel
- Director, Operations, Mid-sized Company, India
- Former Director of Business Development, Large Company, US
- Former Director, Commercial Development, Cell Therapy, Mid-sized Company, Switzerland
- Former Director, Pharmaceuticals Marketing, Large Company, France
- Digitalization and Artificial Intelligence Manager, Large Company, Switzerland
Example Players in Pharmaceutical Contract Manufacturing Market
- Albemarle
- Aspen Pharmacare
- Bausch Health Sciences
- Catalent
- Delpharm
- Eurofins Scientific
- Evonik Industries
- Fareva
- Fresenius Kabi
- Intas Pharmaceuticals
- Lonza
- Micro Labs
- Nipro Patch
- Patheon
- PiSA Farmaceutica
- Recipharm
- Sandoz
- West Pharmaceutical Services
- Wockhardt
- WuXi AppTec
Pharmaceutical Contract Manufacturing Market: Research Coverage
- Market Sizing and Opportunity Analysis: The report features an in-depth analysis of the pharmaceutical contract manufacturing market, focusing on key market segments, including [A] type of product manufactured, [B] type of API, [C] API potency, [D] type of FDF, [E] dosage form, [F] type of oral solid [G] type of packaging offered [H] scale of operation, [I] end user and [J] geographical regions.
- Market Landscape: A detailed overview of the current market landscape of companies engaged in offering contract manufacturing services for small molecules, along with information on several relevant parameters, such as [A] year of establishment, [B] location of manufacturing facility, [C] type of offering, [D] type of pharmaceutical product manufactured, [E] scale of operation, [F] type of service offered, [G] type of finished dosage form manufactured, [H] type of packaging offered.
- Company Profiles: In-depth profiles of prominent players North America, Europe and Asia-Pacific that are engaged in the peptide therapeutics manufacturing market based on [A] year of establishment, [B] location of headquarters, [C] product portfolio, [D] recent developments and [E] an informed future outlook.
- Make versus Buy Framework: A qualitative analysis highlighting various factors that need to be taken into consideration by pharmaceutical drug developers, while deciding whether to manufacture their respective products in-house or engage the services of a CMO.
- Mergers and Acquisitions: A detailed analysis of the various mergers and acquisitions in this domain, based on several relevant parameters, such as [A] year of agreement, [B] type of agreement, [C] geography, [D] most active players (in terms of number of agreements), ownership change matrix and key value drivers of the acquisition agreement.
- Recent Expansions: A detailed analysis of expansions that have taken place in the pharmaceutical contract manufacturing domain, along with analysis on several relevant parameters, such as [A] year of expansion, [B] status of expansion, [C] type of expansion, [D] location of facility, [E] amount invested, [F] type of pharmaceutical product manufactured, [G] type of service offered and [H] most active players.
- Regional Capability Analysis: A detailed regional capability of pharmaceutical contract manufacturing facilities in different regions (North America, Europe, Asia-Pacific and Rest of the World) across the globe.
- Capacity Analysis: An estimate an estimate of the overall installed capacity for the manufacturing of pharmaceuticals, based on information reported by various industry stakeholders in the public domain, highlighting the distribution of the available capacity, based on [A] size of manufacturer (small, mid-sized, and large and very large) and [B] geography (North America, Europe, Asia-Pacific and Rest of the World).
- Demand Analysis: An informed estimate of the annual demand for small molecule drugs, across several relevant parameters, such as [A] scale of operation, [B] type of API, [C] potency of API and [D] geographical regions.
- Market Impact Analysis: An in-depth analysis of the factors that can impact the growth of the global pharmaceutical contract manufacturing market. It also features identification and analysis of [A] key drivers, [B] potential restraints, [C] emerging opportunities, and [D] existing challenges.
Key Questions Answered in this Report
- Which are the leading companies in pharmaceutical contract manufacturing market?
- Which region dominates the pharmaceutical contract manufacturing market?
- What are the key trends observed in the pharmaceutical contract manufacturing market?
- What factors are likely to influence the evolution of this market?
- What are the primary challenges faced by pharmaceutical contract manufacturers?
- What is the current and future market size?
- What is the CAGR of this market?
- How is the current and future market opportunity likely to be distributed across key market segments?
Reasons to Buy this Report
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
- The report can aid businesses in identifying future opportunities in any sector. It also helps in understanding if those opportunities are worth pursuing.
- The report helps in identifying customer demand by understanding the needs, preferences, and behavior of the target audience in order to tailor products or services effectively.
- The report equips new entrants with requisite information regarding a particular market to help them build successful business strategies.
- The report allows for more effective communication with the audience and in building strong business relations.
Additional Benefits
- Complementary PPT Insights Pack
- Complimentary Excel Data Packs for all Analytical Modules in the Report
- 15% Free Content Customization
- Detailed Report Walkthrough Session with Research Team
- Free Updated report if the report is 6-12 months old or older
TABLE OF CONTENTS
1. PREFACE
- 1.1. Introduction
- 1.2. Report Coverage
- 1.3. Market Segmentation
- 1.4. Key Market Insights
- 1.5. Market Share Insights
- 1.6. Key Questions Asked
2. RESEARCH METHODOLOGY
- 2.1. Chapter Overview
- 2.2. Research Assumptions
- 2.2.1. Market Landscape and Market Trends
- 2.2.2. Market Forecast and Opportunity Analysis
- 2.2.3. Comparative Analysis
- 2.3. Database Building
- 2.3.1. Data Collection
- 2.3.2. Data Validation
- 2.3.3. Data Analysis
- 2.4. Project Methodology
- 2.4.1. Secondary Research
- 2.4.1.1. Annual Reports
- 2.4.1.2. Academic Research Papers
- 2.4.1.3. Company Websites
- 2.4.1.4. Investor Presentations
- 2.4.1.5. Regulatory Filings
- 2.4.1.6. White Papers
- 2.4.1.7. Industry Publications
- 2.4.1.8. Conferences and Seminars
- 2.4.1.9. Government Portals
- 2.4.1.10. Media and Press Releases
- 2.4.1.11. Newsletters
- 2.4.1.12. Industry Databases
- 2.4.1.13. Roots Proprietary Databases
- 2.4.1.14. Paid Databases and Sources
- 2.4.1.15. Social Media Portals
- 2.4.1.16. Other Secondary Sources
- 2.4.2. Primary Research
- 2.4.2.1. Types of Primary Research
- 2.4.2.1.1. Qualitative Research
- 2.4.2.1.2. Quantitative Research
- 2.4.2.1.3. Hybrid Approach
- 2.4.2.2. Advantages of Primary Research
- 2.4.2.3. Techniques for Primary Research
- 2.4.2.3.1. Interviews
- 2.4.2.3.2. Surveys
- 2.4.2.3.3. Focus Groups
- 2.4.2.3.4. Observational Research
- 2.4.2.3.5. Social Media Interactions
- 2.4.2.4. Key Opinion Leaders Considered in Primary Research
- 2.4.2.4.1. Company Executives (CXOs)
- 2.4.2.4.2. Board of Directors
- 2.4.2.4.3. Company Presidents and Vice Presidents
- 2.4.2.4.4. Research and Development Heads
- 2.4.2.4.5. Technical Experts
- 2.4.2.4.6. Subject Matter Experts
- 2.4.2.4.7. Scientists
- 2.4.2.4.8. Doctors and Other Healthcare Providers
- 2.4.2.5. Ethics and Integrity
- 2.4.2.5.1. Research Ethics
- 2.4.2.5.2. Data Integrity
- 2.4.2.1. Types of Primary Research
- 2.4.3. Analytical Tools and Databases
- 2.4.1. Secondary Research
- 2.5. Robust Quality Control
3. MARKET DYNAMICS
- 3.1. Chapter Overview
- 3.2. Forecast Methodology
- 3.2.1. Top-down Approach
- 3.2.2. Bottom-up Approach
- 3.2.3. Hybrid Approach
- 3.3. Market Assessment Framework
- 3.3.1. Total Addressable Market (TAM)
- 3.3.2. Serviceable Addressable Market (SAM)
- 3.3.3. Serviceable Obtainable Market (SOM)
- 3.3.4. Currently Acquired Market (CAM)
- 3.4. Forecasting Tools and Techniques
- 3.4.1. Qualitative Forecasting
- 3.4.2. Correlation
- 3.4.3. Regression
- 3.4.4. Extrapolation
- 3.4.5. Convergence
- 3.4.6. Sensitivity Analysis
- 3.4.7. Scenario Planning
- 3.4.8. Data Visualization
- 3.4.9. Time Series Analysis
- 3.4.10. Forecast Error Analysis
- 3.5. Key Considerations
- 3.5.1. Demographics
- 3.5.2. Government Regulations
- 3.5.3. Reimbursement Scenarios
- 3.5.4. Market Access
- 3.5.5. Supply Chain
- 3.5.6. Industry Consolidation
- 3.5.7. Pandemic / Unforeseen Disruptions Impact
- 3.6. Limitations
4. MACRO-ECONOMIC INDICATORS
- 4.1. Chapter Overview
- 4.2. Market Dynamics
- 4.2.1. Time Period
- 4.2.1.1. Historical Trends
- 4.2.1.2. Current and Forecasted Estimates
- 4.2.2. Currency Coverage
- 4.2.2.1. Major Currencies Affecting the Market
- 4.2.2.2. Factors Affecting Currency Fluctuations on the Industry
- 4.2.2.3. Impact of Currency Fluctuations on the Industry
- 4.2.3. Foreign Currency Exchange Rate
- 4.2.3.1. Impact of Foreign Exchange Rate Volatility on the Market
- 4.2.3.2. Strategies for Mitigating Foreign Exchange Risk
- 4.2.4. Recession
- 4.2.4.1. Assessment of Current Economic Conditions and Potential Impact on the Market
- 4.2.4.2. Historical Analysis of Past Recessions and Lessons Learnt
- 4.2.5. Inflation
- 4.2.5.1. Measurement and Analysis of Inflationary Pressures in the Economy
- 4.2.5.2. Potential Impact of Inflation on the Market Evolution
- 4.2.6. Interest Rates
- 4.2.6.1. Interest Rates and Their Impact on the Market
- 4.2.6.2. Strategies for Managing Interest Rate Risk
- 4.2.7. Commodity Flow Analysis
- 4.2.7.1. Type of Commodity
- 4.2.7.2. Origins and Destinations
- 4.2.7.3. Values and Weights
- 4.2.7.4. Modes of Transportation
- 4.2.8. Global Trade Dynamics
- 4.2.8.1. Import Scenario
- 4.2.8.2. Export Scenario
- 4.2.8.3. Trade Policies
- 4.2.8.4. Strategies for Mitigating the Risks Associated with Trade Barriers
- 4.2.8.5. Impact of Trade Barriers on the Market
- 4.2.9. War Impact Analysis
- 4.2.9.1. Russian-Ukraine War
- 4.2.9.2. Israel-Hamas War
- 4.2.10. COVID Impact / Related Factors
- 4.2.10.1. Global Economic Impact
- 4.2.10.2. Industry-specific Impact
- 4.2.10.3. Government Response and Stimulus Measures
- 4.2.10.4. Future Outlook and Adaptation Strategies
- 4.2.11. Other Indicators
- 4.2.11.1. Fiscal Policy
- 4.2.11.2. Consumer Spending
- 4.2.11.3. Gross Domestic Product
- 4.2.11.4. Employment
- 4.2.11.5. Taxes
- 4.2.11.6. Stock Market Performance
- 4.2.11.7. Cross Border Dynamics
- 4.2.1. Time Period
- 4.3. Conclusion
5. EXECUTIVE SUMMARY
- 5.1. Executive Summary: Market Landscape
- 5.2. Executive Summary: Market Trends
- 5.3. Executive Summary: Market Forecast and Opportunity Analysis
6. INTRODUCTION
- 6.1. Chapter Overview
- 6.2. Type of Third-party Manufacturers
- 6.3. Overview of Pharmaceutical Contract Manufacturing
- 6.4. Evolution of Pharmaceutical Contract Manufacturing
- 6.4.1. Traditional Pharmaceutical Contract Manufacturing Organizations
- 6.4.2. Modern Pharmaceutical Contract Manufacturing Organizations
- 6.5. Need for Outsourcing in the Pharmaceutical Industry
- 6.6. Recent Developments in the Pharmaceutical Contract Manufacturing Industry
- 6.6.1. Integration of Artificial Intelligence
- 6.6.2. Strategic Partnerships and Collaborations
- 6.6.2.1. Strategic Long-Term Alliance
- 6.6.2.2. Flexible Short-Term Partnership Agreements
- 6.6.3. Integrated End-to-End Business Model
- 6.6.4. Dependence on Software Service Providers
- 6.7. Services Offered by Contract Manufacturing Organizations
- 6.8. Key Considerations while Selecting a Contract Manufacturing Organization
- 6.9. Risks and Challenges Associated with Outsourcing Pharmaceutical Manufacturing Operations
- 6.10. Future Perspectives
7. PHARMACEUTICAL CONTRACT MANUFACTURING ORGANIZATIONS: REGULATORY LANDSCAPE
- 7.1. Chapter Overview
- 7.2. Regulatory Landscape in North America
- 7.2.1. Landscape in the US
- 7.2.2. Landscape in Canada
- 7.3. Regulatory Landscape in Europe
- 7.4. Regulatory Landscape in Asia-Pacific and Rest of the World
- 7.4.1. Landscape in China
- 7.4.2. Landscape in India
- 7.4.3. Landscape in Japan
- 7.4.4. Landscape in South Korea
- 7.4.5. Landscape in Australia
- 7.4.6. Landscape in Brazil
- 7.5. Analysis of Pharmaceutical Contract Manufacturing Organizations by Approval Received from Regulatory Authorities
- 7.6. Regulatory Landscape: Regional Benchmarking Analysis
8. PHARMACEUTICAL CONTRACT MANUFACTURING ORGANIZATIONS: MARKET LANDSCAPE
- 8.1. Chapter Overview
- 8.2. Pharmaceutical Contract Manufacturing Organizations: Overall Market Landscape
- 8.2.1. Analysis by Year of Establishment
- 8.2.2. Analysis by Company Size
- 8.2.3. Analysis by Location of Headquarters (Region)
- 8.2.4. Analysis by Location of Headquarters (Country)
- 8.2.5. Analysis by Location of Manufacturing Facility (Region)
- 8.2.6. Analysis by Type of Offering
- 8.2.7. Analysis by Type of Pharmaceutical Product Manufactured
- 8.2.8. Analysis by Scale of Operation
- 8.2.9. Analysis by Type of Service Offered
- 8.2.10. Analysis by Finished Dosage Form Manufactured
- 8.2.11. Analysis by Type of Packaging Offered
9. COMPANY PROFILES: LEADING PLAYERS
- 9.1. Chapter Overview
- 9.2. Pharmaceutical Contract Manufacturing Organizations based in North America
- 9.2.1. Albemarle
- 9.2.1.1. Company Overview
- 9.2.1.2. Financial Information
- 9.2.1.3. Service Portfolio
- 9.2.1.4. Recent Developments and Future Outlook
- 9.2.1. Albemarle
- similar details are presented for other below mentioned players based on information in the public domain
- 9.2.2. Bausch Health Services
- 9.2.3. Catalent
- 9.2.4. Patheon
- 9.2.5. PiSA Farmaceutica
- 9.2.6. West Pharmaceutical Services
- 9.3. Pharmaceutical Contract Manufacturing Organizations based in Europe
- 9.3.1. Delpharm
- 9.3.2. Eurofins Scientific
- 9.3.3. Evonik Industries
- 9.3.4. Fareva
- 9.3.5. Fresenius Kabi
- 9.3.6. Lonza
- 9.3.7. Recipharm
- 9.3.8. Sandoz
- 9.4. Pharmaceutical Contract Manufacturing Organizations based in Asia-Pacific and Rest of the World
- 9.4.1. Aspen Pharmacare
- 9.4.2. Intas Pharmaceuticals
- 9.4.3. Micro Labs
- 9.4.4. Nipro Patch
- 9.4.5. WuXi AppTec
- 9.4.6. Wockhardt
10. MAKE VERSUS BUY DECISION FRAMEWORK
- 10.1. Chapter Overview
- 10.2. Assumptions and Key Parameters
- 10.3. Pharmaceutical Contract Manufacturers: Make versus Buy Decision Making
- 10.3.1. Scenario 1
- 10.3.2. Scenario 2
- 10.3.3. Scenario 3
- 10.3.4. Scenario 4
- 10.4. Concluding Remarks
11. MERGERS AND ACQUISITIONS
- 11.1. Chapter Overview
- 11.2. Types of Mergers and Acquisitions
- 11.3. Pharmaceutical Contract Manufacturing Organizations: Mergers and Acquisitions
- 11.3.1. Analysis by Year of Agreement
- 11.3.2. Analysis by Type of Agreement
- 11.3.3. Analysis by Geography
- 11.3.3.1. Local and International Agreements
- 11.3.3.2. Intracontinental and Intercontinental Agreements
- 11.3.4. Most Active Players: Analysis by Number of Agreements
- 11.3.5. Ownership Change Matrix
- 11.3.6. Analysis by Key Value Drivers
12. RECENT EXPANSIONS
- 12.1. Chapter Overview
- 12.2. Type of Expansions
- 12.3. Pharmaceutical Contract Manufacturing Organizations: Recent Expansions
- 12.3.1. Analysis by Year of Expansion
- 12.3.2. Analysis by Status of Expansion
- 12.3.3. Analysis by Type of Expansion
- 12.3.4. Analysis by Year and Type of Expansion
- 12.3.5. Analysis by Location of Facility (Region)
- 12.3.6. Analysis by Location of Facility (Country)
- 12.3.7. Analysis by Type of Expansion and Location of Facility (Region)
- 12.3.8. Analysis of Amount Invested by Year of Expansion
- 12.3.9. Analysis of Amount Invested by Type of Expansion
- 12.3.10. Analysis by Type of Pharmaceutical Product Manufactured
- 12.3.11. Analysis by Type of Service Offered
- 12.3.12. Most Active Players: Analysis by Number of Expansions
- 12.3.13. Most Active Players: Analysis by Amount Invested
13. REGIONAL CAPABILITY ANALYSIS
- 13.1. Chapter Overview
- 13.2. Assumptions and Key Parameters
- 13.3. Overall Landscape of Pharmaceutical Contract Manufacturing Facilities
- 13.3.1. Pharmaceutical Contract Manufacturing Facilities in North America
- 13.3.2. Pharmaceutical Contract Manufacturing Facilities in Europe
- 13.3.3. Pharmaceutical Contract Manufacturing Facilities in Asia-Pacific
- 13.3.4. Pharmaceutical Contract Manufacturing Facilities in Rest of the World
14. CAPACITY ANALYSIS
- 14.1. Chapter Overview
- 14.2. Key Assumptions and Methodology
- 14.3. Pharmaceutical Contract Manufacturing: Global Production Capacity
- 14.3.1. Analysis by Company Size
- 14.3.2. Analysis by Scale of Operation
- 14.3.3. Analysis by Location of Manufacturing Facility
- 14.3.3.1. Pharmaceutical Contract Manufacturing Capacity in North America
- 14.3.3.2. Pharmaceutical Contract Manufacturing Capacity in Europe
- 14.3.3.3. Pharmaceutical Contract Manufacturing Capacity in Asia-Pacific
- 14.4. Concluding Remarks
15. DEMAND ANALYSIS
- 15.1. Chapter Overview
- 15.2. Key Assumptions and Methodology
- 15.3. Global Demand for Pharmaceutical Contract Manufacturing
- 15.3.1. Demand for Pharmaceutical Contract Manufacturing: Analysis by Scale of Operation
- 15.3.2. Demand for Pharmaceutical Contract Manufacturing: Analysis by Type of API
- 15.3.3. Demand for Pharmaceutical Contract Manufacturing: Analysis by Potency of API
- 15.3.4. Demand for Pharmaceutical Contract Manufacturing: Analysis by Geographical Regions
16. MARKET IMPACT ANALYSIS: DRIVERS, RESTRAINTS, OPPORTUNITIES AND CHALLENGES
- 16.1. Chapter Overview
- 16.2. Market Drivers
- 16.3. Market Restraints
- 16.4. Market Opportunities
- 16.5. Market Challenges
- 16.6. Conclusion
17. GLOBAL PHARMACEUTICAL CONTRACT MANUFACTURING MARKET
- 17.1. Chapter Overview
- 17.2. Assumptions and Methodology
- 17.3. Global Pharmaceutical Contract Manufacturing Market: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 17.3.1. Roots Analysis Perspective on Market Growth
- 17.3.2. Scenario Analysis
- 17.3.2.1. Conservative Scenario
- 17.3.2.2. Optimistic Scenario
- 17.4. Key Market Segments
18. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY TYPE OF PRODUCT MANUFACTURED
- 18.1. Chapter Overview
- 18.2. Key Assumptions and Methodology
- 18.3. Pharmaceutical Contract Manufacturing Market: Distribution by Type of Product Manufactured
- 18.3.1. Pharmaceutical Contract Manufacturing Market for API and Intermediates: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 18.3.2. Pharmaceutical Contract Manufacturing Market for FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 18.4. Data Triangulation and Validation
19. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY TYPE OF API
- 19.1. Chapter Overview
- 19.2. Key Assumptions and Methodology
- 19.3. Pharmaceutical Contract Manufacturing Market: Distribution by Type of API
- 19.3.1. Pharmaceutical Contract Manufacturing Market for Originator APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 19.3.2. Pharmaceutical Contract Manufacturing Market for Generic APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 19.4. Data Triangulation and Validation
20. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY API POTENCY
- 20.1. Chapter Overview
- 20.2. Key Assumptions and Methodology
- 20.3. Pharmaceutical Contract Manufacturing Market: Distribution by API Potency
- 20.3.1. Pharmaceutical Contract Manufacturing Market for Low Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 20.3.2. Pharmaceutical Contract Manufacturing Market for High Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 20.4. Data Triangulation and Validation
21. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY TYPE OF FDF
- 21.1. Chapter Overview
- 21.2. Key Assumptions and Methodology
- 21.3. Pharmaceutical Contract Manufacturing Market: Distribution by Type of FDF
- 21.3.1. Pharmaceutical Contract Manufacturing Market for Originator FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 21.3.2. Pharmaceutical Contract Manufacturing Market for Generic FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 21.4. Data Triangulation and Validation
22. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY DOSAGE FORM
- 22.1. Chapter Overview
- 22.2. Key Assumptions and Methodology
- 22.3. Pharmaceutical Contract Manufacturing Market: Distribution by Dosage Form
- 22.3.1. Pharmaceutical Contract Manufacturing Market for Oral Solids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 22.3.2. Pharmaceutical Contract Manufacturing Market for Liquids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 22.3.3. Pharmaceutical Contract Manufacturing Market for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 22.3.4. Pharmaceutical Contract Manufacturing Market for Other Dosage Forms: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 22.4. Data Triangulation and Validation
23. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY ORAL SOLIDS
- 23.1. Chapter Overview
- 23.2. Key Assumptions and Methodology
- 23.3. Pharmaceutical Contract Manufacturing Market: Distribution by Oral Solids
- 23.3.1. Pharmaceutical Contract Manufacturing Market for Tablets: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 23.3.2. Pharmaceutical Contract Manufacturing Market for Capsules: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 23.3.3. Pharmaceutical Contract Manufacturing Market for Other Solid Doses: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 23.4. Data Triangulation and Validation
24. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY TYPE OF PACKAGING OFFERED
- 24.1. Chapter Overview
- 24.2. Key Assumptions and Methodology
- 24.3. Pharmaceutical Contract Manufacturing Market: Distribution by Type of Packaging Offered
- 24.3.1. Pharmaceutical Contract Manufacturing Market for Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.2. Pharmaceutical Contract Manufacturing Market for Blister Packs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.3. Pharmaceutical Contract Manufacturing Market for Oral Liquid Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.4. Pharmaceutical Contract Manufacturing Market for Vials: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.5. Pharmaceutical Contract Manufacturing Market for Prefilled Syringe: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.6. Pharmaceutical Contract Manufacturing Market for Cartridges: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.7. Pharmaceutical Contract Manufacturing Market for Ampoules: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.3.8. Pharmaceutical Contract Manufacturing Market for Other Types of Packaging: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 24.4. Data Triangulation and Validation
25. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY SCALE OF OPERATION
- 25.1. Chapter Overview
- 25.2. Key Assumptions and Methodology
- 25.3. Pharmaceutical Contract Manufacturing Market: Distribution by Scale of Operation
- 25.3.1. Pharmaceutical Contract Manufacturing Market for Clinical Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 25.3.2. Pharmaceutical Contract Manufacturing Market for Commercial Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 25.4. Data Triangulation and Validation
26. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY END-USER
- 26.1. Chapter Overview
- 26.2. Key Assumptions and Methodology
- 26.3. Pharmaceutical Contract Manufacturing Market: Distribution by End-user
- 26.3.1. Pharmaceutical Contract Manufacturing Market for Small Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 26.3.2. Pharmaceutical Contract Manufacturing Market for Mid-sized Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 26.3.3. Pharmaceutical Contract Manufacturing Market for Large and Very Large Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 26.4. Data Triangulation and Validation
27. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY GEOGRAPHICAL REGIONS
- 27.1. Chapter Overview
- 27.2. Key Assumptions and Methodology
- 27.3. Pharmaceutical Contract Manufacturing Market: Distribution by Geographical Regions
- 27.3.1. Pharmaceutical Contract Manufacturing Market in North America: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.1.1. Pharmaceutical Contract Manufacturing Market in the US: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.1.2. Pharmaceutical Contract Manufacturing Market in Canada: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2. Pharmaceutical Contract Manufacturing Market in Europe: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2.1. Pharmaceutical Contract Manufacturing Market in Germany: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2.2. Pharmaceutical Contract Manufacturing Market in France: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2.3. Pharmaceutical Contract Manufacturing Market in the UK: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2.4. Pharmaceutical Contract Manufacturing Market in Italy: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2.5. Pharmaceutical Contract Manufacturing Market in Spain: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.2.6. Pharmaceutical Contract Manufacturing Market in rest of the Europe: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3.1. Pharmaceutical Contract Manufacturing Market in China: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3.2. Pharmaceutical Contract Manufacturing Market in Japan: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3.3. Pharmaceutical Contract Manufacturing Market in India: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3.4. Pharmaceutical Contract Manufacturing Market in South Korea: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3.5. Pharmaceutical Contract Manufacturing Market in Australia: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.3.6. Pharmaceutical Contract Manufacturing Market in rest of the Asia-Pacific: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.4. Pharmaceutical Contract Manufacturing Market in Latin America
- 27.3.4.1. Pharmaceutical Contract Manufacturing Market in Brazil: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.4.2. Pharmaceutical Contract Manufacturing Market in Argentina: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.4.3. Pharmaceutical Contract Manufacturing Market in rest of the Latin America: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.5. Pharmaceutical Contract Manufacturing Market in Middle East and North Africa
- 27.3.5.1. Pharmaceutical Contract Manufacturing Market in Saudi Arabia: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.5.2. Pharmaceutical Contract Manufacturing Market in Egypt: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.5.3. Pharmaceutical Contract Manufacturing Market in rest of the Middle East and North Africa: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.3.1. Pharmaceutical Contract Manufacturing Market in North America: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 27.4. Market Dynamics Assessment
- 27.4.1. Market Movement Analysis
- 27.4.2. Penetration Growth (P-G) Matrix
- 27.5. Data Triangulation and Validation
28. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET: NORTH AMERICA
- 28.1. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Type of Product Manufactured
- 28.1.1. Pharmaceutical Contract Manufacturing Market in North America for API and Intermediates: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.1.2. Pharmaceutical Contract Manufacturing Market in North America for FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.2. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Type of API
- 28.2.1. Pharmaceutical Contract Manufacturing Market in North America for Generic APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.2.2. Pharmaceutical Contract Manufacturing Market in North America for Innovator APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.3. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Potency of API
- 28.3.1. Pharmaceutical Contract Manufacturing Market in North America for Low Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.3.2. Pharmaceutical Contract Manufacturing Market in North America for High Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.4. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Type of FDF
- 28.4.1. Pharmaceutical Contract Manufacturing Market in North America for Generic FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.4.2. Pharmaceutical Contract Manufacturing Market in North America for Innovator FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.5. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Dosage Form
- 28.5.1. Pharmaceutical Contract Manufacturing Market in North America for Oral Solids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.5.2. Pharmaceutical Contract Manufacturing Market in North America for Liquids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.5.3. Pharmaceutical Contract Manufacturing Market in North America for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.5.4. Pharmaceutical Contract Manufacturing Market in North America for Other Dosage Forms: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.6. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Oral Solids
- 28.6.1. Pharmaceutical Contract Manufacturing Market in North America for Tablets: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.6.2. Pharmaceutical Contract Manufacturing Market in North America for Capsules: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.6.3. Pharmaceutical Contract Manufacturing Market in North America for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.7. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Type of Packaging Offered
- 28.7.1. Pharmaceutical Contract Manufacturing Market in North America for Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.7.2. Pharmaceutical Contract Manufacturing Market in North America for Blisters: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.7.3. Pharmaceutical Contract Manufacturing Market in North America for Parenterals: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.7.4. Pharmaceutical Contract Manufacturing Market in North America for Oral Liquid Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.7.5. Pharmaceutical Contract Manufacturing Market in North America for Others: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.8. Pharmaceutical Contract Manufacturing Market in North America: Distribution by End-user
- 28.8.1. Pharmaceutical Contract Manufacturing Market in North America for Small Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.8.2. Pharmaceutical Contract Manufacturing Market in North America for Mid-sized Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.8.3. Pharmaceutical Contract Manufacturing Market in North America for Large and Very Large Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.9. Pharmaceutical Contract Manufacturing Market in North America: Distribution by Scale of Operation
- 28.9.1. Pharmaceutical Contract Manufacturing Market in North America for Clinical Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 28.9.2. Pharmaceutical Contract Manufacturing Market in North America for Commercial Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
29. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET: EUROPE
- 29.1. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Type of Product Manufactured
- 29.1.1. Pharmaceutical Contract Manufacturing Market in Europe for API and Intermediates: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.1.2. Pharmaceutical Contract Manufacturing Market in Europe for FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.2. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Type of API
- 29.2.1. Pharmaceutical Contract Manufacturing Market in Europe for Generic APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.2.2. Pharmaceutical Contract Manufacturing Market in Europe for Innovator APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.3. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Potency of API
- 29.3.1. Pharmaceutical Contract Manufacturing Market in Europe for Low Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.3.2. Pharmaceutical Contract Manufacturing Market in Europe for High Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.4. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Type of FDF
- 29.4.1. Pharmaceutical Contract Manufacturing Market in Europe for Generic FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.4.2. Pharmaceutical Contract Manufacturing Market in Europe for Innovator FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.5. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Dosage Form
- 29.5.1. Pharmaceutical Contract Manufacturing Market in Europe for Oral Solids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.5.2. Pharmaceutical Contract Manufacturing Market in Europe for Liquids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.5.3. Pharmaceutical Contract Manufacturing Market in Europe for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.5.4. Pharmaceutical Contract Manufacturing Market in Europe for Other Dosage Forms: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.6. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Oral Solids
- 29.6.1. Pharmaceutical Contract Manufacturing Market in Europe for Tablets: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.6.2. Pharmaceutical Contract Manufacturing Market in Europe for Capsules: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.6.3. Pharmaceutical Contract Manufacturing Market in Europe for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.7. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Type of Packaging Offered
- 29.7.1. Pharmaceutical Contract Manufacturing Market in Europe for Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.7.2. Pharmaceutical Contract Manufacturing Market in Europe for Blisters: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.7.3. Pharmaceutical Contract Manufacturing Market in Europe for Parenterals: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.7.4. Pharmaceutical Contract Manufacturing Market in Europe for Oral Liquid Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.7.5. Pharmaceutical Contract Manufacturing Market in Europe for Others: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.8. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by End-user
- 29.8.1. Pharmaceutical Contract Manufacturing Market in Europe for Small Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.8.2. Pharmaceutical Contract Manufacturing Market in Europe for Mid-sized Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.8.3. Pharmaceutical Contract Manufacturing Market in Europe for Large and Very Large Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.9. Pharmaceutical Contract Manufacturing Market in Europe: Distribution by Scale of Operation
- 29.9.1. Pharmaceutical Contract Manufacturing Market in Europe for Clinical Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 29.9.2. Pharmaceutical Contract Manufacturing Market in Europe for Commercial Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
30. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET: ASIA-PACIIFC
- 30.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Type of Product Manufactured
- 30.1.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for API and Intermediates: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.1.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Type of API
- 30.2.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Generic APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.2.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Innovator APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.3. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Potency of API
- 30.3.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Low Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.3.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for High Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.4. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Type of FDF
- 30.4.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Generic FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.4.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Innovator FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.5. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Dosage Form
- 30.5.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Oral Solids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.5.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Liquids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.5.3. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.5.4. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Other Dosage Forms: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.6. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Oral Solids
- 30.6.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Tablets: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.6.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Capsules: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.6.3. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.7. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Type of Packaging Offered
- 30.7.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.7.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Blisters: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.7.3. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Parenterals: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.7.4. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Oral Liquid Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.7.5. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Others: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.8. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by End-user
- 30.8.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Small Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.8.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Mid-sized Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.8.3. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Large and Very Large Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.9. Pharmaceutical Contract Manufacturing Market in Asia-Pacific: Distribution by Scale of Operation
- 30.9.1. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Clinical Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 30.9.2. Pharmaceutical Contract Manufacturing Market in Asia-Pacific for Commercial Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
31. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET: REST OF THE WORLD
- 31.1. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Type of Product Manufactured
- 31.1.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for API and Intermediates: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.1.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.2. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Type of API
- 31.2.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Generic APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.2.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Innovator APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.3. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Potency of API
- 31.3.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Low Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.3.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for High Potent APIs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.4. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Type of FDF
- 31.4.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Generic FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.4.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Innovator FDFs: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.5. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Dosage Form
- 31.5.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Oral Solids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.5.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Liquids: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.5.3. Pharmaceutical Contract Manufacturing Market in Rest of the World for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.5.4. Pharmaceutical Contract Manufacturing Market in Rest of the World for Other Dosage Forms: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.6. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Oral Solids
- 31.6.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Tablets: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.6.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Capsules: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.6.3. Pharmaceutical Contract Manufacturing Market in Rest of the World for Emulsions: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.7. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Type of Packaging Offered
- 31.7.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.7.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Blisters: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.7.3. Pharmaceutical Contract Manufacturing Market in Rest of the World for Parenterals: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.7.4. Pharmaceutical Contract Manufacturing Market in Rest of the World for Oral Liquid Bottles: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.7.5. Pharmaceutical Contract Manufacturing Market in Rest of the World for Others: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.8. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by End-user
- 31.8.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Small Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.8.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Mid-sized Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.8.3. Pharmaceutical Contract Manufacturing Market in Rest of the World for Large and Very Large Companies: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.9. Pharmaceutical Contract Manufacturing Market in Rest of the World: Distribution by Scale of Operation
- 31.9.1. Pharmaceutical Contract Manufacturing Market in Rest of the World for Clinical Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- 31.9.2. Pharmaceutical Contract Manufacturing Market in Rest of the World for Commercial Scale: Historical Trends (Since 2023) and Forecasted Estimates (Till 2035)
- *Detailed information on Chapters 28-31 is available in the Excel Data Packs shared along with the report**
32. PHARMACEUTICAL CONTRACT MANUFACTURING MARKET, BY LEADING PLAYERS
- 32.1. Chapter Overview
- 32.2. Key Assumptions and Methodology
- 32.3. Pharmaceutical Contract Manufacturing Market by Leading Players
- 32.4. Data Triangulation and Validation
33. CASE STUDY: COMPARISON OF SMALL AND LARGE MOLECULES DRUGS / THERAPIES
- 33.1. Chapter Overview
- 33.2. Overview of Small Molecule and Large Molecule Drugs / Therapies
- 33.2.1. Comparison of General Characteristics
- 33.2.2. Comparison of Key Specifications
- 33.2.3. Comparison of Manufacturing Processes
- 33.2.4. Comparison of Key Manufacturing-related Challenges





