
|
시장보고서
상품코드
1876442
CT 모의치료기 시장 : 기술별, 제품 유형별, 용도별, 최종 사용자별 예측(-2030년)CT Simulators Market by Technology, Product Type, Application, End User - Global Forecast to 2030 |
||||||
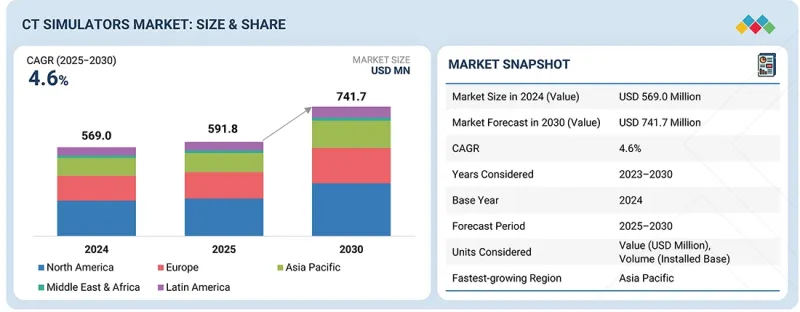
세계의 CT 모의치료기 시장 규모는 2025년 5억 9,810만 달러에서 2030년에는 7억 4,170만 달러에 이를 것으로 예상되며, 예측 기간에 CAGR로 4.6%를 나타낼 것으로 전망됩니다.
| 조사 범위 | |
|---|---|
| 조사 대상 기간 | 2024-2030년 |
| 기준 연도 | 2024년 |
| 예측 기간 | 2025-2030년 |
| 단위 | 100만 달러 |
| 부문 | 제품 유형, 기술, 용도, 최종 사용자, 지역 |
| 대상 지역 | 북미, 유럽, 아시아태평양, 라틴아메리카, 중동 및 아프리카 |
CT 모의치료기 시장은 기술 진보, 질병률 증가 및 의료 인프라에 대한 전략적 투자로 강력한 성장을 보여줍니다. 이러한 요인은 결합되어 CT 모의치료의 효율성, 정확성 및 가용성이 향상되어 진단 및 치료에서 환자 결과를 향상시킵니다. 3D/4D 이미징, 실시간 성능 및 휴대용 솔루션을 포함한 CT 모의치료의 지속적인 혁신으로 진단 정확도와 운영 효율성이 향상되었습니다.
AI와 머신러닝의 통합은 이미지 분석과 치료 계획을 더욱 강화하여 자동 이상 검출, 정밀한 해부학적 매핑, 맞춤형 치료를 가능하게 하여 환자의 결과와 케어의 질을 향상하고 있습니다.
"멀티 슬라이스 CT 모의치료기가 2024년 시장을 독점했습니다."
여러 요인으로 인해 멀티 슬라이스 CT 모의치료기 부문이 CT 모의치료기 시장에 기여합니다. 뛰어난 이미징 능력은 해부학 구조의 고해상도 시각화를 실현하며 정확한 방사선 치료 계획에 필수적입니다. 이것은 주변 건강한 조직에 대한 노출을 줄인 종양의 정밀한 표적화를 보장합니다. 첨단 기술은 스캔 시간을 단축하고 환자 처리 능력과 운영 효율성을 향상시킵니다. 또한 암과 만성 질환의 유병률 증가가 정밀한 진단 및 치료 계획 솔루션에 대한 수요를 뒷받침하고 있습니다. 정확한 지침으로 상세한 이미징에 의존하는 낮은 침습 수술의 채용 증가는 멀티 슬라이스 CT 모의치료기 시장에서의 입지를 더욱 강화하고 있습니다.
"독립 방사선 치료 부문은 2024년 CT 모의치료기 시장에서 가장 큰 점유율을 차지했습니다."
독립 방사선 치료 센터는 증가, 기술 주력, 장비의 높은 가동률로 CT 모의치료기 시장에서 가장 큰 점유율을 차지합니다. International Atomic Energy Agency(IAEA)와 WHO의 DIRAC 데이터베이스에 따르면 세계에 새롭게 설치된 방사선 치료기기의 60-65% 이상이 독립 또는 민간암 센터에 설치되어 있으며, 특히 북미, 유럽, 아시아태평양의 일부에서 현저합니다. 이 센터는 전용 이미징 인프라에 많은 투자를 하고 있으며, 각 시설은 일반적으로 치료 계획과 품질 보증을 위해 하나 이상의 고성능 멀티 슬라이스 CT 모의치료기를 요구합니다.
독립 방사선 치료 센터는 주로 암 치료 워크플로에 주력하고 있으며 방사선 치료 계획에는 정확한 이미징과 모의치료이 필수적입니다. 진단용 CT 스캐너를 부서간에 공유할 수 있는 병원과 달리 방사선치료센터에서는 치료계획에 특화되어 최적화된 전용 CT 모의치료기가 필수가 됩니다. 이러한 시설에서는 암 환자가 안정적으로 유입되는 한편, 병원 특유의 복잡한 부문간의 의존관계는 존재하지 않습니다. 따라서 신속한 영상 획득, 자동 윤곽 묘화, 치료 계획 시스템(TPS)과의 원활한 연계를 통해 소요 시간을 단축하는 전용의 고스루풋 CT 모의치료기를 요구하고 있습니다.
"아시아태평양 부문이 예측 기간에 CT 모의치료기 시장에서 가장 높은 CAGR을 나타낼 것으로 예측됩니다."
아시아태평양은 암, 심혈관 질환, 호흡기 질환 등 만성 질환의 유병률 상승으로 CT 모의치료기 시장에서 가장 빠르게 성장하고 있습니다. 이러한 동향으로 CT 모의치료기를 포함한 첨단 진단 및 치료 계획 기술에 대한 수요가 크게 증가하고 있습니다. AI 통합, 첨단 3D/4D 이미징 기법, 저선량 CT 스캔 등 지속적인 기술 혁신으로 지역 전체에서 CT 모의치료기의 성능과 가용성이 향상되었습니다. 특히 인도나 중국 등 신흥경제권의 의료 인프라에 대한 대규모 투자는 최첨단 의료기술의 신속한 채용을 지원하고 있습니다. 또한, 질병의 조기 발견의 중요성에 대한 사회적 의식 증가는 도시와 지방 모두의 의료 현장에서 첨단 이미징 도구의 사용을 촉진하고 있습니다. 이러한 요인들은 예측 기간 동안 아시아태평양이 CT 모의치료기 시장의 주요 성장 촉진요인이 될 것으로 예측됩니다.
이 보고서는 세계의 CT 모의치료기 시장에 대한 조사 분석을 통해 주요 성장 촉진요인과 억제요인, 경쟁 구도, 미래 동향 등의 정보를 제공합니다.
자주 묻는 질문
목차
제1장 서론
제2장 조사 방법
제3장 주요 요약
제4장 주요 인사이트
- CT 모의치료기 시장 개요
- 아시아태평양의 성장 기회 : 최종 사용자별, 국가별
- CT 모의치료기 시장 : 지리적 성장 기회
제5장 시장 개요
- 서론
- 시장 역학
- 성장 촉진요인
- 성장 억제요인
- 기회
- 과제
- 미충족 수요(Unmet Needs)와 백스페이스
- 연결된 시장과 부문 간 기회
- Tier 1/2/3 기업의 전략적 움직임
제6장 기술, 특허, AI 채용에 의한 파괴적 혁신
- 기술 분석
- 주요 기술
- 보완 기술
- 인접 기술
- 특허 분석
- 미래의 용도
- CT 모의치료기 시장에 대한 AI/생성형 AI의 영향
- 주요 이용 사례와 시장의 장래성
- AI 이용 사례
- CT 모의치료 베스트 프랙티스
- AI 도입의 케이스 스터디
- 연결된 인접 생태계와 시장 기업에 미치는 영향
- 생성형 AI의 채용에 대한 클라이언트의 준비도
- 성공 사례와 실세계에의 응용
제7장 지속가능성과 규제정세
- 지역 규제 및 규정 준수
- 규제기관, 정부기관, 기타 조직
- 규제 프레임워크
- 인증, 라벨, 환경 기준
제8장 고객정세와 구매행동
- 주요 이해관계자와 구매평가기준
- 구매 프로세스의 주요 이해 관계자
- 주요 구매 기준
- 의사결정 프로세스
- 채용 장벽과 내부 과제
- 최종 사용자의 미충족 수요(Unmet Needs)
제9장 업계 동향
- Porter's Five Forces 분석
- 거시경제지표
- 서론
- 의료비와 인프라 전망
- 세계의 CT 업계 동향
- 밸류체인 분석
- 연구개발
- 원재료 조달 및 제조
- 유통, 마케팅 및 판매
- 애프터 서비스
- 공급망 분석
- 주요 기업
- 중소기업
- 최종 사용자
- 생태계 분석
- 가격 설정 분석
- CT 모의치료기의 평균 판매 가격 동향(2022-2024년) : 주요 기업별
- CT 모의치료기의 평균 판매 가격 동향(2022-2024년) : 지역별
- 무역 분석
- HS코드 9022.12의 수입 데이터
- HS코드 9022.12의 수출 데이터
- 주요 컨퍼런스 및 이벤트(2025-2026년)
- 고객사업에 영향을 주는 동향/혁신
- 투자 및 자금조달 시나리오
- 사례 연구 분석
- CT 모의치료기 시장에 대한 미국 관세의 영향(2025년)
- 서론
- 주요 관세율
- 가격의 영향 분석
- 국가/지역에 미치는 영향
- 최종 이용 산업에 대한 영향
제10장 CT 모의치료기 시장 : 기술별
- 서론
- 2D/3D CT 모의치료 기술
- 3D/4D CT 모의치료 기술
제11장 CT 모의치료기 시장 : 제품 유형별
- 서론
- 단층 CT 모의치료기
- 다중층 CT 모의치료기
제12장 CT 모의치료기 시장 : 용도별
- 서론
- 영상 유도 방사선 치료
- 3D CRT
- 근접 치료
- 적응형 방사선 치료/표면 유도 방사선 치료
- 양성자 치료
- 기타 용도
제13장 CT 모의치료기 시장 : 최종 사용자별
- 서론
- 독립 방사선 치료 센터
- 병원
- 기타 최종 사용자
제14장 CT 모의치료기 시장 : 지역별
- 서론
- 북미
- 북미의 거시경제 전망
- 미국
- 캐나다
- 유럽
- 유럽의 거시경제 전망
- 독일
- 영국
- 이탈리아
- 스페인
- 기타 유럽
- 아시아태평양
- 아시아태평양의 거시경제 전망
- 일본
- 중국
- 인도
- 한국
- 호주
- 기타 아시아태평양
- 라틴아메리카
- 라틴아메리카의 거시경제 전망
- 브라질
- 멕시코
- 기타 라틴아메리카
- 중동 및 아프리카
- 중동 및 아프리카의 거시경제 전망
- GCC 국가
- 기타 중동 및 아프리카
제15장 경쟁 구도
- 서론
- 주요 진입기업의 전략/강점
- 수익 분석(2020-2024년)
- 시장 점유율 분석(2024년)
- 기업평가 및 재무 지표
- 브랜드/제품 비교
- 기업평가 매트릭스 : 주요 기업(2024년)
- 기업의 평가 매트릭스 : 스타트업/중소기업(2024년)
- 경쟁 시나리오
제16장 기업 프로파일
- 주요 기업
- SIEMENS HEALTHINEERS AG
- PHILIPS HEALTHCARE
- GE HEALTHCARE
- CANON MEDICAL SYSTEMS CORPORATION
- ELEKTA
- FUJIFILM HEALTHCARE
- ZIEHM IMAGING GMBH
- SHANGHAI UNITED IMAGING HEALTHCARE CO., LTD.
- NEUSOFT MEDICAL SYSTEMS CO., LTD.
- HITACHI HIGH-TECH CORPORATION
- SHINVA MEDICAL INSTRUMENT CO., LTD.
- ACCURAY INCORPORATED
- 기타 기업
- DMS HEALTH
- NORTH STAR IMAGING INC.
- FLUKE BIOMEDICAL
- PRIZMED IMAGING
- KONING HEALTH
- PLANMECA OY
- PINSENG HEALTHCARE
- SCANLAB
- ARINETA LTD.
- RYOEI
- BRAINLAB SE
- RADIOLOGY ONCOLOGY SYSTEMS
- BLOCK IMAGING, INC.
제17장 부록
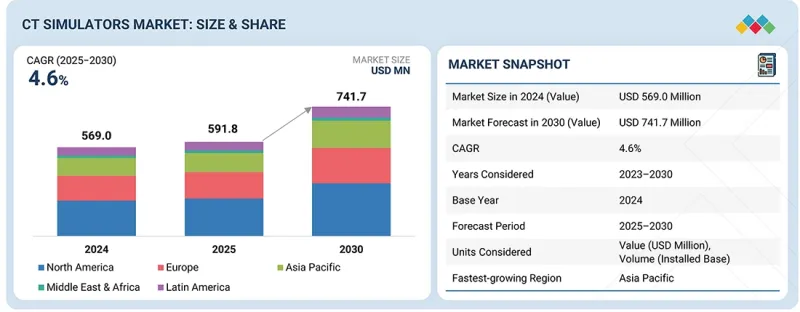
KTH 25.11.28The global CT simulators market is projected to reach USD 741.7 million by 2030, up from USD 598.1 million in 2025, at a CAGR of 4.6% during the forecast period.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD million) |
| Segments | Product Type, Technology, Application, End User, and Region |
| Regions covered | North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa |
The CT simulators market is experiencing strong growth driven by technological advancements, increasing disease rates, and strategic investments in healthcare infrastructure. These factors collectively enhance the efficiency, accuracy, and availability of CT simulation, resulting in improved patient outcomes in both diagnostic and therapeutic settings. Continuous innovation in CT simulation, including 3D/4D imaging, real-time capabilities, and portable solutions, has enhanced diagnostic accuracy and operational efficiency.
The integration of AI and machine learning further enhances image analysis and treatment planning, enabling automated anomaly detection, precise anatomical mapping, and personalized therapy, thereby improving patient outcomes and care quality.
"Multi-slice CT simulators dominated the market in 2024."
The multi-slice CT simulators segment contributes to the CT simulator market, driven by several key factors. Their superior imaging capabilities offer high-resolution visualization of anatomical structures, which is vital for accurate radiation therapy planning, ensuring precise tumor targeting while reducing exposure to surrounding healthy tissue. Advanced technology allows for faster scan times, increasing patient throughput and operational efficiency. Additionally, the growing prevalence of cancer and chronic diseases has boosted demand for precise diagnostic and treatment planning solutions. The increasing adoption of minimally invasive procedures, which rely on detailed imaging for accurate guidance, further strengthens the market position of multi-slice CT simulators.
"The independent radiotherapy segment held the largest share of the CT simulators market in 2024."
Independent radiotherapy centers hold the largest share of the CT simulators market due to their increasing number, technology focus, and high equipment utilization. According to the International Atomic Energy Agency (IAEA) and WHO's DIRAC database, over 60-65% of newly installed radiotherapy units worldwide are located in stand-alone or private cancer centers, especially across North America, Europe, and parts of the Asia Pacific. These centers heavily invest in dedicated imaging infrastructure, with each site typically needing at least one high-performance multi-slice CT simulator for treatment planning and quality assurance.
Independent radiotherapy centers mainly focus on cancer treatment workflows, where accurate imaging and simulation are essential for radiation therapy planning. Unlike hospitals, which can share diagnostic CT scanners among departments, radiotherapy centers need dedicated CT simulators optimized specifically for therapy planning-making adoption necessary rather than optional. These centers often manage a steady flow of oncology patients but do not have the complex departmental dependencies typical of hospitals. Therefore, they seek dedicated, high-throughput CT simulators that provide quick image acquisition, automated contouring, and seamless integration with Treatment Planning Systems (TPS) to reduce turnaround times.
"The Asia Pacific regional segment is expected to register the highest CAGR in the CT simulators market during the forecast period."
The Asia-Pacific region is showing the fastest growth in the CT simulator market, driven by the rising prevalence of chronic conditions such as cancer, cardiovascular diseases, and respiratory disorders. This trend is significantly increasing the demand for advanced diagnostic and treatment planning technologies, including CT simulators. Ongoing technological innovation-such as the integration of artificial intelligence (AI), advanced 3D/4D imaging techniques, and low-dose CT scanning-is improving both the performance and accessibility of CT simulators throughout the region. Large investments in healthcare infrastructure, especially in emerging economies like India and China, are supporting the quick adoption of cutting-edge medical technologies. Additionally, growing public awareness about the importance of early disease detection is boosting the use of advanced imaging tools in both urban and rural healthcare settings. Together, these factors establish the Asia-Pacific region as the main driver of CT simulator market growth during the forecast period.
A breakdown of the primary participants referred to for this report is provided below:
- By Company: Tier 1 (35%), Tier 2 (45%), and Tier 3 (20%)
- By Designation: C-level Executives (35%), Director-level Executives (25%), and Others (40%)
- By Region: North America (40%), Europe (30%), Asia Pacific (20%), Latin America (5%), and the Middle East & Africa (5%)
Prominent players in the CT simulators market include Siemens Healthineers (Germany), GE Healthcare (US), Philips Healthcare (Netherlands), Canon Medical Systems Corporation (Japan), Varian Medical Systems (US), Elekta (Switzerland), Fujifilm Corporation (US), Fluke Biomedical (US), Ziehm Imaging Gmbh (Germany), among others.
Research Coverage
- The report studies the CT simulators market based on product type, technology, application, end user, and region
- The report analyzes factors (such as drivers, restraints, opportunities, and challenges) affecting market growth
- The report evaluates the opportunities and challenges in the market for stakeholders and provides details of the competitive landscape for market leaders
- The report studies micro markets with respect to their growth trends, prospects, and contributions to the global CT simulators market
- The report forecasts the revenue of market segments with respect to five major regions
Key Benefits of Buying this Report
This report is valuable for both new and experienced players in the market, providing essential information to identify potential investment opportunities. It offers a comprehensive overview of both major and minor players, supporting effective risk analysis and informed investment decisions. The report includes precise segmentation by end users and geographic regions, giving detailed insights into niche market segments. Additionally, it highlights key trends, growth drivers, challenges, and opportunities, aiding strategic decision-making through a balanced approach analysis.
Through this report, readers get insightful views into the following parameters:
- Analysis of key drivers (Growing adoption of image-guided and adaptive radiotherapy (IGRT/ART), Global push toward hypofractionation and stereotactic body radiotherapy (SBRT), Rising cancer incidence and increasing diagnostic CT utilization), restraints (Recent advances in AI-driven CBCT-to-synthetic CT (sCT) conversions as replacement, Perceived overlap with diagnostic CT or PET/CT scans), opportunities (AI and PCCT unlocking high-performance simulation opportunities, Use of hybrid CT-linac and CT-on-rails solutions), challenges (High capital costs), relating to the growth of the CT simulators market.
- Product Development/Innovation: Detailed insights on upcoming technologies, research & development activities, and new product & service launches in the CT simulators market.
- Market Development: Comprehensive information about lucrative markets - the report analyses the CT simulators market across varied regions.
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the CT simulators market
- Competitive Assessment: A comprehensive analysis of market share, product offerings, and leading strategies of major players, such as Siemens Healthineers (Germany), GE Healthcare (US), Philips Healthcare (Netherlands), Canon Medical Systems Corporation (Japan), Varian Medical Systems (US), Elekta (Switzerland), Fujifilm Corporation (US), Fluke Biomedical (US), Ziehm Imaging Gmbh (Germany).
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.3.4 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
2 RESEARCH METHODOLOGY
- 2.1 RESEARCH DATA
- 2.1.1 SECONDARY RESEARCH
- 2.1.1.1 Key secondary sources
- 2.1.1.2 Key data from secondary sources
- 2.1.1.3 Objectives of secondary research
- 2.1.2 PRIMARY RESEARCH
- 2.1.2.1 Key primary sources
- 2.1.2.2 Key supply- and demand-side participants
- 2.1.2.3 Breakdown of primary interviews
- 2.1.2.4 Objectives of primary research
- 2.1.2.5 Key primary insights
- 2.1.1 SECONDARY RESEARCH
- 2.2 MARKET SIZE ESTIMATION
- 2.2.1 BOTTOM-UP APPROACH
- 2.2.1.1 Company revenue estimation
- 2.2.1.2 Customer-based market estimation
- 2.2.1.3 Primary interviews
- 2.2.2 TOP-DOWN APPROACH
- 2.2.1 BOTTOM-UP APPROACH
- 2.3 GROWTH RATE ASSUMPTIONS
- 2.4 DATA TRIANGULATION
- 2.5 STUDY ASSUMPTIONS
- 2.6 RESEARCH LIMITATIONS
- 2.7 RISK ANALYSIS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
- 4.1 CT SIMULATORS MARKET OVERVIEW
- 4.2 ASIA PACIFIC: GROWTH OPPORTUNITIES, BY END USER AND COUNTRY
- 4.3 CT SIMULATORS MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
5 MARKET OVERVIEW
- 5.1 INTRODUCTION
- 5.2 MARKET DYNAMICS
- 5.2.1 DRIVERS
- 5.2.1.1 Growing adoption of image-guided and adaptive radiotherapy (IGRT/ART)
- 5.2.1.2 Global push toward hypofractionation and stereotactic body radiotherapy (SBRT)
- 5.2.1.3 Rising cancer incidence and increasing diagnostic CT utilization
- 5.2.2 RESTRAINTS
- 5.2.2.1 Recent advances in AI-driven CBCT-to-synthetic CT (sCT) conversions as replacement
- 5.2.2.2 Perceived overlap with diagnostic CT or PET/CT scans
- 5.2.3 OPPORTUNITIES
- 5.2.3.1 AI and PCCT unlocking high-performance simulation opportunities
- 5.2.3.2 Use of hybrid CT-linac and CT-on-rails solutions
- 5.2.4 CHALLENGES
- 5.2.4.1 High capital costs
- 5.2.1 DRIVERS
- 5.3 UNMET NEEDS & WHITE SPACES
- 5.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 5.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
6 STRATEGIC DISRUPTION THROUGH TECHNOLOGY, PATENTS, AND AI ADOPTION
- 6.1 TECHNOLOGY ANALYSIS
- 6.1.1 KEY TECHNOLOGIES
- 6.1.1.1 Radiotherapy planning software/Value-based software
- 6.1.1.2 Flat-table couch & laser positioning
- 6.1.1.3 4D CT & motion management
- 6.1.2 COMPLEMENTARY TECHNOLOGIES
- 6.1.2.1 Linear accelerators (LINACs)
- 6.1.2.2 Dosimetry tools
- 6.1.2.3 Treatment verification systems
- 6.1.3 ADJACENT TECHNOLOGIES
- 6.1.3.1 MRI simulators
- 6.1.3.2 Cone-beam CT (CBCT) in LINACs
- 6.1.3.3 Photon and proton therapy delivery systems
- 6.1.1 KEY TECHNOLOGIES
- 6.2 PATENT ANALYSIS
- 6.2.1 INNOVATIONS AND PATENT REGISTRATIONS
- 6.3 FUTURE APPLICATIONS
- 6.4 IMPACT OF AI/GEN AI ON CT SIMULATORS MARKET
- 6.4.1 TOP USE CASES AND MARKET POTENTIAL
- 6.4.2 AI USE CASES
- 6.4.3 BEST PRACTICES IN CT SIMULATION
- 6.4.4 CASE STUDIES OF AI IMPLMENTATION
- 6.4.5 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS
- 6.4.6 CLIENT'S READINESS TO ADOPT GENERATIVE AI
- 6.5 SUCCESS STORIES & REAL-WORLD APPLICATIONS
7 SUSTAINABILITY & REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS & COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 REGULATORY FRAMEWORK
- 7.1.2.1 North America
- 7.1.2.1.1 US
- 7.1.2.1.2 Canada
- 7.1.2.2 Europe
- 7.1.2.2.1 UK
- 7.1.2.2.2 Germany
- 7.1.2.2.3 France
- 7.1.2.2.4 Italy
- 7.1.2.2.5 Spain
- 7.1.2.3 Asia Pacific
- 7.1.2.3.1 Japan
- 7.1.2.3.2 China
- 7.1.2.3.3 India
- 7.1.2.4 Latin America
- 7.1.2.4.1 Brazil
- 7.1.2.4.2 Mexico
- 7.1.2.5 Middle East & Africa
- 7.1.2.5.1 UAE
- 7.1.2.5.2 South Africa
- 7.1.2.1 North America
- 7.1.3 CERTIFICATIONS, LABELLING, AND ECO-STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 KEY STAKEHOLDERS & BUYING EVALUATION CRITERIA
- 8.1.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.1.2 KEY BUYING CRITERIA
- 8.2 DECISION-MAKING PROCESS
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM END USERS
- 8.4.1 UNMET NEEDS FROM END USERS
9 INDUSTRY TREND
- 9.1 PORTER'S FIVE FORCES ANALYSIS
- 9.1.1 THREAT OF NEW ENTRANTS
- 9.1.2 THREAT OF SUBSTITUTES
- 9.1.3 BARGAINING POWER OF SUPPLIERS
- 9.1.4 BARGAINING POWER OF BUYERS
- 9.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 9.2 MACROECONOMIC INDICATORS
- 9.2.1 INTRODUCTION
- 9.2.2 HEALTHCARE EXPENDITURE & INFRASTRUCTURE OUTLOOK
- 9.2.3 TRENDS IN GLOBAL CT INDUSTRY
- 9.3 VALUE CHAIN ANALYSIS
- 9.3.1 RESEARCH & DEVELOPMENT
- 9.3.2 RAW MATERIAL PROCUREMENT AND MANUFACTURING
- 9.3.3 DISTRIBUTION AND MARKETING & SALES
- 9.3.4 AFTER-SALES SERVICES
- 9.4 SUPPLY CHAIN ANALYSIS
- 9.4.1 PROMINENT COMPANIES
- 9.4.2 SMALL & MEDIUM-SIZED ENTERPRISES
- 9.4.3 END USERS
- 9.5 ECOSYSTEM ANALYSIS
- 9.5.1 ROLE IN ECOSYSTEM
- 9.6 PRICING ANALYSIS
- 9.6.1 AVERAGE SELLING PRICE TREND OF CT SIMULATORS, BY KEY PLAYER, 2022-2024
- 9.6.2 AVERAGE SELLING PRICE TREND OF CT SIMULATORS, BY REGION, 2022-2024
- 9.7 TRADE ANALYSIS
- 9.7.1 IMPORT DATA FOR HS CODE 9022.12
- 9.7.2 EXPORT DATA FOR HS CODE 9022.12
- 9.8 KEY CONFERENCES & EVENTS, 2025-2026
- 9.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- 9.10 INVESTMENT & FUNDING SCENARIO
- 9.11 CASE STUDY ANALYSIS
- 9.11.1 SEAMLESS WORKFLOW INTEGRATION BY SIEMENS HEALTHINEERS (VARIAN) TO STRENGTHEN CT SIMULATORS MARKET POSITION
- 9.11.2 INNOVATION BY GE HEALTHCARE TO ENHANCE CT SIMULATION EFFICIENCY AND PRECISION
- 9.11.3 PHILIPS BIG BORE CT TO EXPAND GLOBAL ADOPTION IN RADIOTHERAPY SIMULATION
- 9.12 IMPACT OF 2025 US TARIFF ON CT SIMULATORS MARKET
- 9.12.1 INTRODUCTION
- 9.12.2 KEY TARIFF RATES
- 9.12.3 PRICE IMPACT ANALYSIS
- 9.12.4 IMPACT ON COUNTRIES/REGIONS
- 9.12.4.1 North America
- 9.12.4.1.1 US
- 9.12.4.2 Europe
- 9.12.4.3 Asia Pacific
- 9.12.4.1 North America
- 9.12.5 IMPACT ON END-USE INDUSTRIES
10 CT SIMULATORS MARKET, BY TECHNOLOGY
- 10.1 INTRODUCTION
- 10.2 2D/3D CT SIMULATION TECHNOLOGY
- 10.2.1 CONVENTIONAL NEEDS AND SIZABLE DEMAND IN LOW-RESOURCE SETTINGS TO DRIVE MARKET DEMAND
- 10.3 3D/4D CT SIMULATION TECHNOLOGY
- 10.3.1 HIGH THROUGHPUT ABILITY AND EFFICIENCY TO PROPEL MARKET GROWTH
11 CT SIMULATORS MARKET, BY PRODUCT TYPE
- 11.1 INTRODUCTION
- 11.2 SINGLE-SLICE CT SIMULATORS
- 11.2.1 LOW-VOLUME APPLICATIONS AND COST-EFFECTIVENESS TO MAINTAIN STABLE DEMAND
- 11.3 MULTI-SLICE CT SIMULATORS
- 11.3.1 HIGH THROUGHPUT ABILITY AND EFFICIENCY TO FUEL MARKET DEMAND
12 CT SIMULATORS MARKET, BY APPLICATION
- 12.1 INTRODUCTION
- 12.2 IMAGE-GUIDED RADIATION THERAPY
- 12.2.1 HIGH-QUALITY CT SIMULATION TO NEED IGRT WORKFLOW FOR DRIVING DEMAND
- 12.3 3D CONFORMAL RADIATION THERAPY
- 12.3.1 CT SIMULATION TO PROVIDE VOLUMETRIC DATASETS FOR CONFORMAL BEAM SHAPING IN 3D CRT
- 12.4 BRACHYTHERAPY
- 12.4.1 OAR DELINEATION AND ACCURATE APPLICATOR VERIFICATION TO PROPEL DEMAND
- 12.5 ADAPTIVE RADIOTHERAPY/SURFACE-GUIDED RADIOTHERAPY
- 12.5.1 RISING ADOPTION OF CT SIMULATORS IN ADAPTIVE RADIOTHERAPY TO BOOST WORKFLOW PRECISION AND CLINICAL EFFICIENCY
- 12.6 PROTON THERAPY
- 12.6.1 CT SIMULATORS TO PROVIDE ESSENTIAL IMAGING SUPPORT FOR ACCURATE PROTON THERAPY PLANNING
- 12.7 OTHER APPLICATIONS
13 CT SIMULATORS MARKET, BY END USER
- 13.1 INTRODUCTION
- 13.2 INDEPENDENT RADIOTHERAPY CENTERS
- 13.2.1 GROWING INCIDENCE OF CANCER GLOBALLY TO PROPEL DEMAND FOR ADVANCED DIAGNOSTIC AND PLANNING TOOLS
- 13.3 HOSPITALS
- 13.3.1 RISING TREND OF INTEGRATED ONCOLOGY CARE SERVICES TO SUPPORT MARKET GROWTH
- 13.4 OTHER END USERS
14 CT SIMULATORS MARKET, BY REGION
- 14.1 INTRODUCTION
- 14.2 NORTH AMERICA
- 14.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 14.2.2 US
- 14.2.2.1 US to dominate North American CT simulators market during study period
- 14.2.3 CANADA
- 14.2.3.1 Increased demand for advanced imaging technologies to support market growth
- 14.3 EUROPE
- 14.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 14.3.2 GERMANY
- 14.3.2.1 Aging population with high burden of chronic diseases to augment market growth
- 14.3.3 UK
- 14.3.3.1 Need for faster cancer treatment and better image quality at lower radiation doses to support market growth
- 14.3.4 ITALY
- 14.3.4.1 Increasing investments in CT simulators to drive market
- 14.3.5 SPAIN
- 14.3.5.1 Increasing demand for integration of CT simulation solutions in radiation oncology department to propel market
- 14.3.6 REST OF EUROPE
- 14.4 ASIA PACIFIC
- 14.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 14.4.2 JAPAN
- 14.4.2.1 Increase in adoption of high-precision radiotherapy protocols to fuel market
- 14.4.3 CHINA
- 14.4.3.1 Expanding radiotherapy capacity across hospitals and cancer centers to aid market growth
- 14.4.4 INDIA
- 14.4.4.1 Improvements in healthcare infrastructure and rising incidence of cancer to boost market growth
- 14.4.5 SOUTH KOREA
- 14.4.5.1 Growing radiotherapy utilization to support market growth
- 14.4.6 AUSTRALIA
- 14.4.6.1 Expanding radiotherapy utilization to propel market growth
- 14.4.7 REST OF ASIA PACIFIC
- 14.5 LATIN AMERICA
- 14.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 14.5.2 BRAZIL
- 14.5.2.1 Collaborative programs and partnerships to expand access to radiotherapy technologies and boost market
- 14.5.3 MEXICO
- 14.5.3.1 Rising demand for radiotherapy planning to promote market growth
- 14.5.4 REST OF LATIN AMERICA
- 14.6 MIDDLE EAST & AFRICA
- 14.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 14.6.2 GCC COUNTRIES
- 14.6.2.1 Need for advanced cancer treatment to spur market growth
- 14.6.3 REST OF MIDDLE EAST & AFRICA
15 COMPETITIVE LANDSCAPE
- 15.1 INTRODUCTION
- 15.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 15.2.1 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN CT SIMULATORS MARKET
- 15.3 REVENUE ANALYSIS, 2020-2024
- 15.4 MARKET SHARE ANALYSIS, 2024
- 15.4.1 MARKET RANKING OF KEY PLAYERS
- 15.5 COMPANY VALUATION & FINANCIAL METRICS
- 15.5.1 FINANCIAL METRICS
- 15.5.2 COMPANY VALUATION
- 15.6 BRAND/PRODUCT COMPARISON
- 15.7 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2024
- 15.7.1 STARS
- 15.7.2 PERVASIVE PLAYERS
- 15.7.3 EMERGING LEADERS
- 15.7.4 PARTICIPANTS
- 15.7.5 COMPANY FOOTPRINT: KEY PLAYERS, 2024
- 15.7.5.1 Company footprint
- 15.7.5.2 Region footprint
- 15.7.5.3 Product type footprint
- 15.7.5.4 Technology footprint
- 15.7.5.5 Application footprint
- 15.7.5.6 End-user footprint
- 15.8 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2024
- 15.8.1 PROGRESSIVE COMPANIES
- 15.8.2 RESPONSIVE COMPANIES
- 15.8.3 DYNAMIC COMPANIES
- 15.8.4 STARTING BLOCKS
- 15.8.5 COMPETITIVE BENCHMARKING OF STARTUPS/SMES, 2024
- 15.8.5.1 Detailed list of key startups/SMEs
- 15.8.5.2 Competitive benchmarking of key startups/SMEs
- 15.9 COMPETITIVE SCENARIO
- 15.9.1 PRODUCT LAUNCHES
- 15.9.2 DEALS
- 15.9.3 EXPANSIONS
16 COMPANY PROFILES
- 16.1 KEY PLAYERS
- 16.1.1 SIEMENS HEALTHINEERS AG
- 16.1.1.1 Business overview
- 16.1.1.2 Products offered
- 16.1.1.3 Recent developments
- 16.1.1.3.1 Product launches
- 16.1.1.3.2 Deals
- 16.1.1.3.3 Expansions
- 16.1.1.3.4 Other developments
- 16.1.1.4 MnM view
- 16.1.1.4.1 Right to win
- 16.1.1.4.2 Strategic choices
- 16.1.1.4.3 Weaknesses & competitive threats
- 16.1.2 PHILIPS HEALTHCARE
- 16.1.2.1 Business overview
- 16.1.2.2 Products offered
- 16.1.2.3 Recent developments
- 16.1.2.3.1 Product launches
- 16.1.2.3.2 Expansions
- 16.1.2.4 MnM view
- 16.1.2.4.1 Right to win
- 16.1.2.4.2 Strategic choices
- 16.1.2.4.3 Weaknesses & competitive threats
- 16.1.3 GE HEALTHCARE
- 16.1.3.1 Business overview
- 16.1.3.2 Products offered
- 16.1.3.3 Recent developments
- 16.1.3.3.1 Product launches
- 16.1.3.3.2 Deals
- 16.1.3.3.3 Expansions
- 16.1.3.4 MnM view
- 16.1.3.4.1 Right to win
- 16.1.3.4.2 Strategic choices
- 16.1.3.4.3 Weaknesses & competitive threats
- 16.1.4 CANON MEDICAL SYSTEMS CORPORATION
- 16.1.4.1 Business overview
- 16.1.4.2 Products offered
- 16.1.4.3 Recent developments
- 16.1.4.3.1 Product launches
- 16.1.4.3.2 Deals
- 16.1.4.3.3 Expansions
- 16.1.4.4 MnM view
- 16.1.4.4.1 Right to win
- 16.1.4.4.2 Strategic choices
- 16.1.4.4.3 Weaknesses & competitive threats
- 16.1.5 ELEKTA
- 16.1.5.1 Business overview
- 16.1.5.2 Products offered
- 16.1.5.3 Recent developments
- 16.1.5.3.1 Product launches
- 16.1.5.3.2 Deals
- 16.1.5.3.3 Expansions
- 16.1.5.4 MnM view
- 16.1.5.4.1 Right to win
- 16.1.5.4.2 Strategic choices
- 16.1.5.4.3 Weaknesses & competitive threats
- 16.1.6 FUJIFILM HEALTHCARE
- 16.1.6.1 Business overview
- 16.1.6.2 Products offered
- 16.1.6.3 Recent developments
- 16.1.6.3.1 Deals
- 16.1.7 ZIEHM IMAGING GMBH
- 16.1.7.1 Business overview
- 16.1.7.2 Products offered
- 16.1.7.3 Recent developments
- 16.1.7.3.1 Product launches & approvals
- 16.1.7.3.2 Deals
- 16.1.8 SHANGHAI UNITED IMAGING HEALTHCARE CO., LTD.
- 16.1.8.1 Business overview
- 16.1.8.2 Products offered
- 16.1.8.3 Recent developments
- 16.1.8.3.1 Deals
- 16.1.9 NEUSOFT MEDICAL SYSTEMS CO., LTD.
- 16.1.9.1 Business overview
- 16.1.9.2 Products offered
- 16.1.9.3 Recent developments
- 16.1.9.3.1 Product approvals
- 16.1.9.3.2 Deals
- 16.1.10 HITACHI HIGH-TECH CORPORATION
- 16.1.10.1 Business overview
- 16.1.10.2 Products offered
- 16.1.10.3 Recent developments
- 16.1.10.3.1 Other developments
- 16.1.11 SHINVA MEDICAL INSTRUMENT CO., LTD.
- 16.1.11.1 Business overview
- 16.1.11.2 Products offered
- 16.1.12 ACCURAY INCORPORATED
- 16.1.12.1 Business overview
- 16.1.12.2 Products offered
- 16.1.1 SIEMENS HEALTHINEERS AG
- 16.2 OTHER PLAYERS
- 16.2.1 DMS HEALTH
- 16.2.2 NORTH STAR IMAGING INC.
- 16.2.3 FLUKE BIOMEDICAL
- 16.2.4 PRIZMED IMAGING
- 16.2.5 KONING HEALTH
- 16.2.6 PLANMECA OY
- 16.2.7 PINSENG HEALTHCARE
- 16.2.8 SCANLAB
- 16.2.9 ARINETA LTD.
- 16.2.10 RYOEI
- 16.2.11 BRAINLAB SE
- 16.2.12 RADIOLOGY ONCOLOGY SYSTEMS
- 16.2.13 BLOCK IMAGING, INC.
17 APPENDIX
- 17.1 DISCUSSION GUIDE
- 17.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 17.3 CUSTOMIZATION OPTIONS
- 17.4 RELATED REPORTS
- 17.5 AUTHOR DETAILS





