
|
시장보고서
상품코드
1984857
식품 병원체 안전 검사 기기 및 소모품 시장(-2031년) : 유형(시스템, 소모품&소모 자재), 검사 대상 식품, 사이트(사내 레버러토리, 외부 위탁 시설, 정부계 레버러토리), 기술, 지역별Food Pathogen Safety Testing Equipment & Supplies Market by Type (Systems, Consumables & Supplies ), Food Tested, by Site (In-House Laboratories, Outsourcing Facilities, Government Labs), Technology, and Region - Global Forecast to 2031 |
||||||
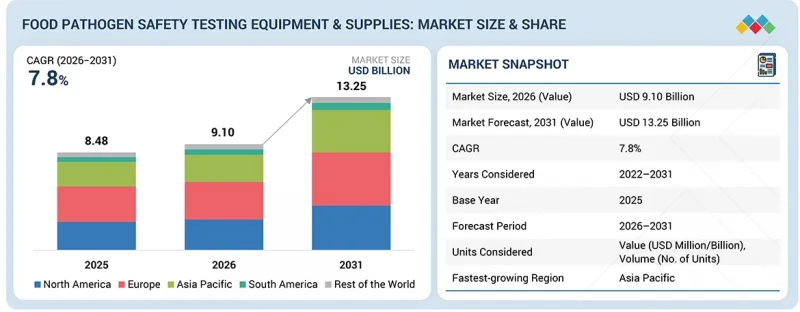
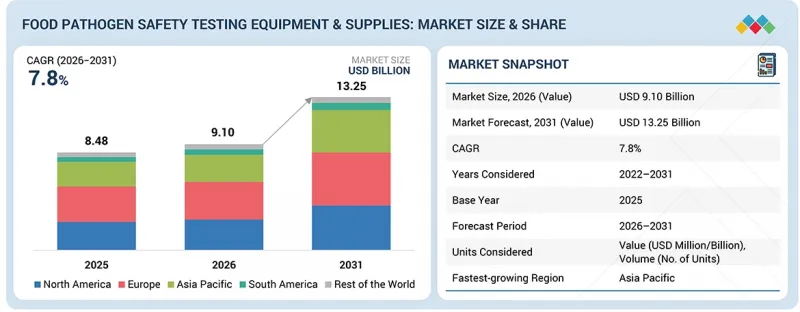
식품 병원체 안전 검사 기기 및 소모품 시장 규모는 2026년 91억 달러에서 2031년까지 CAGR 7.8%로 성장을 지속하여 132억 5,000만 달러에 이를 것으로 예측됩니다.
| 조사 범위 | |
|---|---|
| 조사 대상 기간 | 2021-2030년 |
| 기준연도 | 2024년 |
| 예측 기간 | 2025-2030년 |
| 단위 | 금액(달러) 및 킬로톤 |
| 부문 | 유형, 대상 식품, 기술, 지역 |
| 대상 지역 | 북미, 아시아태평양, 유럽, 중동 및 아프리카, 남미 |
식품 병원체 안전 검사 장비 및 소모품 시장은 지속적으로 확대되고 있으며, 이는 식품 산업, 제약 산업, 사료 산업에서 식품 안전과 품질에 대한 관심이 높아짐에 따라 식품 안전 및 품질에 대한 관심이 높아졌기 때문입니다. 식중독 발생률 증가와 규정 준수 요건의 강화로 인해 제조업체는 제품의 안전성을 유지하고 규정을 준수하기 위해 효과적인 병원체 검사 절차를 수립해야 합니다. 또한, 품질검사의 정확도 향상과 검사 결과의 신속화를 위해 사용되는 PCR법 기반 검사, 면역측정법, 자동화된 미생물 검출 및 검사 시스템 등 신속검사 방법의 개선도 시장 확대에 기여하고 있습니다. 또한 가공식품, 즉석식품, 포장식품의 인기가 높아짐에 따라 생산 및 공급망 전반에 걸쳐 신뢰할 수 있는 검사 및 소모품의 사용이 촉진되고 있습니다. 세계 식품 무역의 확대와 제품 추적성에 대한 관심이 높아짐에 따라 향후 몇 년 동안 현장 검사, 실험실 자동화, 식품 품질 관리의 전산화 시스템에 대한 투자 유입으로 식품 병원체 안전 검사 장비 및 소모품에 대한 지속적인 수요를 뒷받침할 것으로 예측됩니다.
제품 유형별로는 소모품 및 키트 부문이 가장 큰 점유율을 차지할 것으로 예측됩니다.
시장 점유율의 가장 큰 부분을 차지하는 것은 소모품 및 키트이며, 이는 주로 일상적인 병원체 검사 과정에서 반복적으로 사용되는 것에 기인합니다. 이들은 식품 생산 및 품질 관리 시설에서 이루어지는 모든 검사에 필수적인 시약, 검사 키트, 배지, 시료 준비 제품입니다. 식품 안전 기준을 준수하기 위한 일상적이고 신속한 검사에 대한 요구가 점점 더 높아짐에 따라 사내 및 제3자 검사 기관에서 고품질 소모품 소비에 대한 수요가 증가하고 있습니다. 또한, PCR 및 면역 측정법에 기반한 검사 기술의 사용이 증가함에 따라 병원체 검출 키트의 소비가 증가하고 있으며, 이는 이 부문 시장 지배력을 더욱 강화하는 요인으로 작용하고 있습니다.
검사 대상 식품 중 육류 및 가금류 카테고리가 가장 큰 비중을 차지할 것으로 예측됩니다.
육류 및 가금류 부문은 미생물 오염과 엄격한 규제 모니터링 요건에 가장 민감하게 영향을 받기 때문에 시장에서 가장 큰 점유율을 차지하고 있습니다. 이런 식품은 살모넬라균, 리스테리아균, 대장균의 발생 위험이 높습니다. 따라서 가공, 포장, 유통의 각 공정에서 식품을 정기적으로 검사하는 것이 중요합니다. 전 세계적으로 육가공 및 포장육 제품의 소비가 증가함에 따라 최적의 병원체 검사 솔루션에 대한 수요가 더욱 증가하고 있습니다. 이에 따라 식품 제조 기업들은 제품의 안전성을 보장하고 식품 안전 기준을 준수하기 위해 첨단 검사 장비와 소모품에 대한 투자를 확대하고 있습니다.
사이트별로는 외부 위탁 시설(서비스 랩)이 시장을 주도할 전망
가장 큰 시장 부문은 제3자 기관 및 위탁 검사 기관을 통한 아웃소싱 시설입니다. 검사 인프라와 기술력 비용이 높기 때문에 많은 식품 제조업체는 병원체 검사 서비스를 전문 검사 기관에 위탁하고 있습니다. 검사기관은 높은 수준의 검사, 신속한 결과 제공, 규정 준수 이행에 대한 지원을 제공합니다. 식품 안전에 대한 요구사항이 증가하고 정밀한 미생물 검사에 대한 수요가 증가함에 따라 기업들은 정기적인 품질 관리 및 검증 절차를 공인된 외부 검사 기관에 위탁하고 있습니다.
지역별로 보면 유럽이 가장 큰 시장으로
유럽은 세계 시장 점유율에서 가장 큰 비중을 차지하고 있으며, 이는 식품 가공 산업 전반의 엄격한 식품 안전 기준과 종합적인 품질 보증 시스템 덕분입니다. 식품 안전과 추적성을 확립하기 위한 규제 검사의 도입으로 병원체 검사 솔루션의 사용이 촉진되고 있으며, 이 지역에서는 필수 검사 요건이 되었습니다. 또한, 가공식품 제조업체의 높은 집중도와 식품 품질 및 식품 안전에 대한 소비자의 인식이 높아진 것도 시장 성장에 기여하고 있습니다. 또한, 실험실 자동화 및 첨단 미생물 검사 기술에 대한 투자도 이 지역에서 이루어지고 있으며, 이는 시장에서의 선도적인 위치로 이어지고 있습니다.
세계의 식품 병원체 안전 검사 기기 및 소모품 시장을 조사했으며, 시장 개요, 시장 성장에 영향을 미치는 각종 영향요인 분석, 기술 및 특허 동향, 법 및 규제 환경, 사례 분석, 시장 규모 추이 및 예측, 각종 부문별/지역별/주요 국가별 상세 분석, 경쟁 구도, 주요 기업 개요 등의 정보를 정리하여 전해드립니다. 정리되어 있습니다.
자주 묻는 질문
목차
제1장 서론
제2장 주요 요약
제3장 프리미엄 인사이트
제4장 시장 개요
제5장 업계 동향
제6장 기술 진보, AI의 영향, 특허, 혁신, 향후 용도
제7장 지속가능성과 규제 상황
제8장 고객 현황과 구매 행동
제9장 식품 병원체 안전 검사 기기 및 소모품 시장 : 유형별
제10장 식품 병원체 안전 검사 기기 및 소모품 시장 : 검사 대상 식품별
제11장 식품 병원체 안전 검사 기기 및 소모품 시장 : 사이트별
제12장 식품 병원체 안전 검사 기기 및 소모품 시장 : 기술별(정성적)
제13장 식품 병원체 안전 검사 기기 및 소모품 시장 : 지역별
제14장 경쟁 구도
제15장 기업 개요
제16장 조사 방법
제17장 인접 시장 및 관련 시장
제18장 부록
LSH 26.04.13The food pathogen safety testing equipment & supplies market is estimated to be USD 9.10 billion in 2026 and is projected to reach USD 13.25 billion by 2031, at a CAGR of 7.8%.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2021-2030 |
| Base Year | 2024 |
| Forecast Period | 2025-2030 |
| Units Considered | Value (USD Million/Billion) and Volume (Kiloton) |
| Segments | Type, Food Tested, Technology and Region |
| Regions covered | North America, Asia Pacific, Europe, the Middle East & Africa, and South America |
The market is continuing to rise, and this is a result of the rising interest in food safety and quality in the food industry, pharmaceutical industry, and also in the animal feed industry. Increased rate of food-borne diseases and the high compliance demands are compelling manufacturers to ensure that they establish effective pathogen testing procedures to preserve product safety as well as ensure that their products comply with the provisions. The market is also expanding due to the improvement of rapid testing methods like the PCR-based testing, immunoassays, and the automated microbial detection and testing system, which are used to enhance quality testing and turnaround time. Moreover, the growing popularity of processed food products, ready-to-eat and packaged food products, is prompting the use of tests and consumables that can be trusted across the production and supply chain. As the food trade worldwide continues to grow and increasing attention is given to the traceability of products, it can be forecasted that the ongoing demand for food pathogen safety testing equipment and supplies will be supported by the influx of investments in on-site testing, laboratory automation, and computerized systems of food quality control over the coming years.
In type, the segments of consumables and kits are projected to take the biggest share of the market in the food pathogen safety testing equipment and supplies.
The highest share of the market is comprised of consumables and kits, which are principally attributed to the recurring nature of their application in routine pathogen testing processes. These will be the reagents, test kits, culture media, and sample preparation products that are mandatory in every test performed in food production and quality control plants. The ever-increasing requirements of routine and quick testing in order to comply with food safety standards have raised the pressure on the consumption of good-quality consumables within the in-house and third-party laboratories. Also, the increasing usage of PCR-based and immunoassay-based testing technologies is contributing to the consumption of pathogen detection kits, which has a similar effect on the dominance of the segment in the market.
The meat & poultry category is determined to take the highest portion of the market among foods tested.
The meat & poultry segment is the most vulnerable to microbial contamination and high regulatory monitoring requirements; therefore, it has the highest share in the market. Salmonella, Listeria, and E. coli are more likely to happen in this type of food. Thus, it is important to regularly test food products during processing, packaging, and distribution processes. The rise in the consumption of processed and packaged meat products around the world has further augmented the demand to identify the best pathogen testing solutions. Consequently, food manufacturing companies are putting more money into sophisticated testing tools and consumables in order to secure the safety of the products and have them in line with food safety standards.
According to site, outsourcing facilities (service laboratories) are projected to take over the market.
The largest site segment is outsourcing facilities through third-party and contract testing laboratories. The high cost of testing infrastructure and technical skills makes many food manufacturers outsource pathogen testing services to special laboratories. Laboratories provide a high level of testing, quicker turnaround, and assistance when it comes to fulfilling regulatory compliance. The rising number of food safety requirements, as well as the demand to perform precise microbial testing, prompts businesses to trust certified external laboratories to perform regular quality control and verification procedures.
In terms of region, Europe has been estimated to be the largest market in the food pathogen safety testing equipment and supplies.
The Europe region is the greatest contributor to global market share, which is a result of stringent food safety standards and comprehensive quality assurance systems in the entire food processing industry. The introduction of regulatory tests to establish food safety and traceability has prompted the use of pathogen test solutions, which have become a mandatory testing requirement in the region. Also, the high concentration of processed food producers and increased awareness of consumers about the quality of food and food safety have also contributed to market growth. The laboratory automation and sophisticated microbial testing technologies are also making investments in the region, leading to the top position in the market.
In-depth interviews have been conducted with chief executive officers (CEOs), directors, and other executives from various key organizations operating in the food pathogen safety testing equipment & supplies market.
- By Company Type: Tier 1 - 30%, Tier 2 - 25%, and Tier 3 - 45%
- By Designation: Directors - 30%, Managers - 25%, Others - 45%
- By Region: North America - 20%, Europe - 30%, Asia Pacific - 40%, and Rest of the World - 10%
Prominent companies in the market include Thermo Fisher Scientific Inc. (US), Bio-Rad Laboratories, Inc. (US), Merck KGaA (Germany), Neogen Corporation (US), bioMerieux (France), Agilent Technologies, Inc. (US), QIAGEN (Germany), Shimadzu Corporation (Japan), Bruker (US), PerkinElmer Inc. (US), Hygiena, LLC (US), Solabia Group (France), Roka Bio Science (US), Promega Corporation (US), Romer Labs Division Holding (Austria), and others.
Research Coverage
This research report categorizes the food pathogen safety testing equipment & supplies market by type (systems [hybridization-based, chromatography-based, spectrometry-based, immunoassay-based], consumables & supplies [test kits, microbial culture media, reagents & other consumables]), food tested (meat & poultry, fish & seafood, processed food, fruits & vegetables, dairy products, cereals & grains, other food products), by site (in-house laboratories [factory labs], outsourcing facilities [service labs], government labs), technology [qualitative], and region (North America, Europe, Asia Pacific, South America, and Rest of the World).
The report's scope encompasses detailed information on the major factors, including drivers, restraints, challenges, and opportunities, that influence the growth of the food pathogen safety testing equipment & supplies industry. A thorough analysis of the key industry players has been done to provide insights into their business, services, key strategies, contracts, partnerships, agreements, product launches, mergers & acquisitions, and recent developments associated with the food pathogen safety testing equipment & supplies market. This report provides a competitive analysis of emerging startups in the food pathogen safety testing equipment & supplies market ecosystem. Furthermore, the study covers industry-specific trends, including technology analysis, ecosystem & market mapping, and patent & regulatory landscape, among others.
Reasons to Buy This Report
The report provides market leaders/new entrants with information on the closest approximations of revenue numbers for the overall food pathogen safety testing equipment & supplies market and its subsegments. It will help stakeholders understand the competitive landscape and gain more insights to better position their businesses and plan suitable go-to-market strategies. The report also helps stakeholders understand the pulse of the market, providing them with information on key market drivers, restraints, challenges, and opportunities.
The report provides insights into the following pointers:
- Analysis of key drivers (expansion of genomic surveillance and traceability), restraints (high capital and recurring cost), opportunities (expansion of portable and on-site rapid testing), and challenges (regulatory fragmentation and acceptance timelines) influencing the growth of the food pathogen safety testing equipment & supplies market
- Product Development/Innovation: Detailed insights into research & development activities and new product launches in the food pathogen safety testing equipment & supplies market
- Market Development: Comprehensive information about lucrative markets-analysis of food pathogen safety testing equipment & supplies across varied regions
- Market Diversification: Exhaustive information about new product sources, untapped geographies, recent developments, and investments in the food pathogen safety testing equipment & supplies market
- Competitive Assessment: In-depth assessment of market shares, growth strategies, product offerings, brand/product comparison, and product footprints of leading players such as Thermo Fisher Scientific Inc. (US), Bio-Rad Laboratories, Inc. (US), Merck KGaA (Germany), Neogen Corporation (US), bioMerieux (France), Agilent Technologies, Inc. (US), QIAGEN (Germany), Shimadzu Corporation (Japan), Bruker (US), PerkinElmer Inc. (US), Hygiena, LLC (US), Solabia Group (France), Roka Bio Science (US), Promega Corporation (US), Romer Labs Division Holding (Austria) and others in the food pathogen safety testing equipment & supplies market
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE AND SEGMENTATION
- 1.3.1 MARKETS COVERED AND REGIONAL SCOPE
- 1.3.2 INCLUSIONS AND EXCLUSIONS
- 1.4 YEARS CONSIDERED
- 1.5 UNITS CONSIDERED
- 1.5.1 CURRENCY UNIT
- 1.5.2 VOLUME UNIT
- 1.6 STAKEHOLDERS
- 1.7 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 MARKET HIGHLIGHTS AND KEY INSIGHTS
- 2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS IN FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 2.4 HIGH GROWTH SEGMENTS
- 2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 ATTRACTIVE OPPORTUNITIES FOR PLAYERS IN FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 3.2 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY TYPE AND REGION
- 3.3 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY SITE
- 3.4 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY TYPES OF FOOD TESTED
- 3.5 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY SYSTEMS
- 3.6 FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET, BY CONSUMABLES & SUPPLIES
- 3.7 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY COUNTRY
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Persistent burden of foodborne illness
- 4.2.1.2 Regulatory tightening and preventive controls
- 4.2.1.3 Expansion of genomic surveillance and traceability
- 4.2.1.4 Faster testing and automation needs
- 4.2.2 RESTRAINTS
- 4.2.2.1 High capital and recurring costs
- 4.2.2.2 Workforce and technical capacity gaps
- 4.2.2.3 Complex validation and multi-jurisdictional compliance
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Expansion of portable and on-site rapid testing
- 4.2.3.2 Digital integration and smart laboratory ecosystems
- 4.2.3.3 Reagent-as-a-service and flexible commercial models
- 4.2.4 CHALLENGES
- 4.2.4.1 Regulatory fragmentation and acceptance timelines
- 4.2.4.2 Cybersecurity and data integrity risks
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS AND WHITE SPACES
- 4.3.1 UNMET NEEDS IN FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 4.3.1.1 Rapid on-site confirmatory testing
- 4.3.1.2 Cost-effective validated solutions for emerging markets
- 4.3.1.3 Laboratory digitization and cybersecurity-hardened solutions
- 4.3.2 WHITE SPACE OPPORTUNITIES
- 4.3.2.1 Viability-aware rapid testing platforms
- 4.3.2.2 Flexible commercial deployment models
- 4.3.2.3 Genomic surveillance-as-a-service platforms
- 4.3.2.4 Digitally secure smart laboratory infrastructure
- 4.3.1 UNMET NEEDS IN FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 4.4 INTERCONNECTED MARKETS AND CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 INTENSITY OF COMPETITIVE RIVALRY
- 5.1.2 BARGAINING POWER OF SUPPLIERS
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 THREAT OF SUBSTITUTES
- 5.1.5 THREAT OF NEW ENTRANTS
- 5.2 MACROECONOMICS OUTLOOK
- 5.2.1 RISING GLOBAL POPULATION AND INCREASING FOOD DEMAND
- 5.2.2 INDUSTRIALIZATION OF FOOD PROCESSING IN EMERGING ECONOMIES
- 5.2.3 RISING PUBLIC HEALTH EXPENDITURE AND FOOD SAFETY INVESTMENTS
- 5.3 SUPPLY CHAIN ANALYSIS (MICROBIOLOGY AGAR CULTURE PRODUCTS)
- 5.3.1 INPUT AND RAW MATERIAL SOURCING
- 5.3.2 CULTIVATION AND SEAWEED PROCESSING
- 5.3.3 AGAR EXTRACTION AND REFINEMENT
- 5.3.4 MEDIA FORMULATION AND MANUFACTURING
- 5.3.5 QUALITY TESTING AND CERTIFICATION
- 5.3.6 PACKAGING & LABELING
- 5.3.7 WAREHOUSING & DISTRIBUTION
- 5.3.8 END-USE APPLICATION
- 5.4 VALUE CHAIN ANALYSIS (FOOD PATHOGEN TESTING KITS)
- 5.4.1 RAW MATERIAL PROCUREMENT
- 5.4.2 KIT FORMULATION & MANUFACTURING
- 5.4.3 QUALITY ASSURANCE & REGULATORY CERTIFICATION
- 5.4.4 SALES, MARKETING & DISTRIBUTION
- 5.5 ECOSYSTEM ANALYSIS
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE OF KEY PLAYERS, BY SYSTEM
- 5.6.2 AVERAGE SELLING PRICE, BY SYSTEMS
- 5.6.3 AVERAGE SELLING PRICE OF TEST KITS, BY REGION
- 5.6.4 AVERAGE SELLING PRICE OF MICROBIAL CULTURE MEDIA, BY REGION
- 5.6.5 AVERAGE SELLING PRICE OF REAGENTS & CONSUMABLES, BY REGION
- 5.7 TRADE ANALYSIS
- 5.7.1 IMPORT SCENARIO (HS CODE 902720)
- 5.7.2 EXPORT SCENARIO (HS CODE 902720)
- 5.8 KEY CONFERENCES AND EVENTS, 2026-2027
- 5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- 5.10 INVESTMENT AND FUNDING SCENARIO
- 5.11 CASE STUDY ANALYSIS
- 5.11.1 BCN RESEARCH LABORATORIES PARTNERED WITH BIOMERIEUX TO INTRODUCE GENE-UP SYSTEMS FOR FOOD SAFETY TESTING
- 5.11.2 EUROFINS COLLABORATED WITH RHEONIX TO INTRODUCE RHEONIX LISTERIA PATTERNALERT ASSAY
- 5.12 IMPACT OF 2025 US TARIFF - FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 5.12.1 INTRODUCTION
- 5.12.2 KEY TARIFF RATES
- 5.12.3 PRICE IMPACT ANALYSIS
- 5.12.4 IMPACT ON COUNTRIES/REGIONS
- 5.12.4.1 US
- 5.12.4.2 Europe
- 5.12.4.3 Asia Pacific
- 5.12.5 IMPACT ON END-USE INDUSTRIES
6 TECHNOLOGY ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 KEY TECHNOLOGIES
- 6.1.1 PCR-BASED MOLECULAR DIAGNOSTICS
- 6.1.2 MICROARRAY-BASED DETECTION PLATFORMS
- 6.1.3 RAPID ON-SITE DETECTION SYSTEMS
- 6.2 COMPLEMENTARY TECHNOLOGIES
- 6.2.1 AUTOMATED MICROBIAL DETECTION SYSTEMS
- 6.2.2 CHROMATOGRAPHY-BASED TESTING SOLUTIONS
- 6.3 ADJACENT TECHNOLOGIES
- 6.3.1 BIOSENSOR-BASED DETECTION TECHNOLOGIES
- 6.3.2 AI-ENABLED FOOD SAFETY MONITORING PLATFORMS
- 6.4 PATENT ANALYSIS
- 6.4.1 INTRODUCTION
- 6.4.2 LIST OF MAJOR PATENTS, JANUARY 2017-DECEMBER 2025
- 6.5 FUTURE APPLICATIONS
- 6.5.1 AI-INTEGRATED PATHOGEN DETECTION PLATFORMS
- 6.5.2 PORTABLE ON-SITE FOOD TESTING DEVICES
- 6.5.3 LAB-ON-CHIP RAPID DETECTION SYSTEMS
- 6.5.4 MICROFLUIDIC BIOSENSOR-BASED DETECTION
- 6.5.5 REAL-TIME FOOD QUALITY MONITORING SENSORS
- 6.6 IMPACT OF AI/GEN AI ON FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES INDUSTRY
- 6.6.1 TOP USE CASES AND MARKET POTENTIAL
- 6.6.2 BEST PRACTICES IN BIOTECHNOLOGY PROCESSING AND CULTIVATION
- 6.6.3 CASE STUDIES OF AI IMPLEMENTATION IN FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 6.6.4 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS
- 6.6.5 READINESS TO ADOPT GENERATIVE AI IN FOOD PATHOGEN SAFETY TESTING EQUIPMENT & SUPPLIES MARKET
- 6.7 SUCCESS STORIES AND REAL-WORLD APPLICATIONS
- 6.7.1 CLEAR LABS: AUTOMATED NGS-BASED PATHOGEN DETECTION FOR FOOD SAFETY
- 6.7.2 NEMIS TECHNOLOGIES: ON-SITE RAPID PATHOGEN DETECTION FOR FOOD PROCESSING FACILITIES
- 6.7.3 PATHOGENDX: MICROARRAY-BASED MULTI-PATHOGEN DETECTION TECHNOLOGY
7 SUSTAINABILITY AND REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS AND COMPLIANCE
- 7.1.1 REGULATORY FRAMEWORK
- 7.1.1.1 Global Food Safety Initiative (GFSI)
- 7.1.1.2 North America
- 7.1.1.2.1 US
- 7.1.1.2.1.1 Federal Legislation
- 7.1.1.2.1.2 State Legislation
- 7.1.1.2.1.3 Food Safety in Retail Food
- 7.1.1.2.1.4 Food Safety in Trade
- 7.1.1.2.1.5 HACCP Regulation
- 7.1.1.2.1.6 US Regulation for Foodborne Pathogens in Poultry
- 7.1.1.2.1.7 Food Safety Regulation for Fruit and Vegetable Growers
- 7.1.1.2.2 Canada
- 7.1.1.2.3 Mexico
- 7.1.1.2.1 US
- 7.1.1.3 Europe
- 7.1.1.3.1 European Union
- 7.1.1.3.1.1 Microbiological Criteria Regulation
- 7.1.1.3.2 Germany
- 7.1.1.3.3 UK
- 7.1.1.3.4 France
- 7.1.1.3.5 Italy
- 7.1.1.3.6 Poland
- 7.1.1.3.1 European Union
- 7.1.1.4 Asia Pacific
- 7.1.1.4.1 China
- 7.1.1.4.2 Japan
- 7.1.1.4.3 India
- 7.1.1.4.3.1 Food Safety Standards Amendment Regulation, 2012
- 7.1.1.4.3.2 Food Safety Standards Amendment Regulation, 2011
- 7.1.1.4.3.3 Food Safety and Standards Act, 2006
- 7.1.1.4.4 Australia and New Zealand
- 7.1.1.4.4.1 Food Standards Australia and New Zealand
- 7.1.1.5 South America
- 7.1.1.5.1 Brazil
- 7.1.1.5.1.1 Ministry of Agriculture, Livestock, and Food Supply (MAPA)
- 7.1.1.5.1.2 Ministry of Health (MS)
- 7.1.1.5.2 Argentina
- 7.1.1.5.1 Brazil
- 7.1.1.6 Rest of the World
- 7.1.1.6.1 South Africa
- 7.1.1.6.1.1 International vs. Local Standards & Legislation
- 7.1.1.6.1.2 Private Standards in South Africa and Requirements for Product Testing
- 7.1.1.6.1 South Africa
- 7.1.2 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.3 INDUSTRY STANDARDS
- 7.1.4 LABELING REQUIREMENTS AND CLAIMS
- 7.1.5 ANTICIPATED REGULATORY CHANGES IN NEXT 5-10 YEARS
- 7.1.5.1 Stricter validation requirements for microbiological testing methods
- 7.1.5.2 Expansion of rapid testing approval pathways
- 7.1.5.3 Mandatory digital traceability in food safety testing workflows
- 7.1.5.4 Strengthening of environmental monitoring and hygiene testing regulations
- 7.1.1 REGULATORY FRAMEWORK
- 7.2 SUSTAINABILITY INITIATIVES
- 7.2.1 ENERGY-EFFICIENT LABORATORY SYSTEMS
- 7.2.2 SUSTAINABLE REAGENTS AND WASTE MANAGEMENT
- 7.3 SUSTAINABILITY IMPACT AND REGULATORY POLICY INITIATIVES
- 7.4 CERTIFICATIONS, LABELING, ECO-STANDARDS
8 CUSTOMER LANDSCAPE AND BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS AND BUYING EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 BUYING CRITERIA
- 8.3 ADOPTION BARRIERS AND INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.5 MARKET PROFITABILITY
- 8.5.1 REVENUE POTENTIAL
- 8.5.2 COST DYNAMICS
- 8.5.3 MARGIN OPPORTUNITIES, BY SOURCE
9 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY TYPE
- 9.1 INTRODUCTION
- 9.2 SYSTEMS
- 9.2.1 HYBRIDIZATION-BASED
- 9.2.1.1 Need for high analytical precision in food testing to drive adoption of hybridization-based systems
- 9.2.1.2 Polymerase chain reaction (PCR)
- 9.2.1.3 Gene amplifiers
- 9.2.2 CHROMATOGRAPHY-BASED
- 9.2.2.1 Focus on detecting chemical contaminants in food to encourage use of chromatography-based techniques
- 9.2.2.2 High-performance liquid chromatography (HPLC)
- 9.2.2.3 Liquid chromatography
- 9.2.2.4 Gas chromatography
- 9.2.2.5 Other chromatography-based systems
- 9.2.3 SPECTROMETRY-BASED
- 9.2.3.1 Growing demand to gain analytical approach to food pathogen testing to drive market
- 9.2.3.2 UV-visible spectroscopy
- 9.2.3.3 Infrared (IR) spectroscopy
- 9.2.3.4 Mass spectrometry (MS)
- 9.2.4 IMMUNOASSAY-BASED
- 9.2.4.1 Focus on building ability to test multiple samples to drive use of immunoassay-based systems
- 9.2.1 HYBRIDIZATION-BASED
- 9.3 CONSUMABLES & SUPPLIES
- 9.3.1 TEST KITS
- 9.3.1.1 Need for user-friendly and efficient food testing solutions to boost growth
- 9.3.2 MICROBIAL CULTURE MEDIA
- 9.3.2.1 Growing demand for advanced technologies to drive adoption of food safety testing solutions
- 9.3.3 REAGENTS & OTHER CONSUMABLES
- 9.3.3.1 Need for operational efficiency and ease of use in food safety testing environments
- 9.3.1 TEST KITS
10 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY FOOD TESTED
- 10.1 INTRODUCTION
- 10.2 MEAT & POULTRY
- 10.2.1 RISING GLOBAL MEAT AND POULTRY CONSUMPTION TO SPUR DEMAND
- 10.3 FISH & SEAFOOD
- 10.3.1 GROWING RISK OF CONTAMINATION OF FISH AND SEAFOOD DUE TO SUPPLY CHAIN COMPLEXITIES TO DRIVE MARKET
- 10.4 DAIRY PRODUCTS
- 10.4.1 MOUNTING CASES OF LISTERIOSIS AND OTHER PATHOGEN-RELATED ILLNESSES IN DAIRY PRODUCTS TO STIMULATE MARKET DEMAND
- 10.5 PROCESSED FOOD
- 10.5.1 PROCESSED FOODS' SUSCEPTIBILITY TO PATHOGEN CONTAMINATION TO DRIVE DEMAND FOR SAFETY TESTING EQUIPMENT
- 10.6 FRUITS & VEGETABLES
- 10.6.1 MULTIPLE OUTBREAKS ASSOCIATED WITH PATHOGENS IN FRUITS AND VEGETABLES TO FUEL MARKET GROWTH
- 10.7 CEREALS & GRAINS
- 10.7.1 GROWING RISK OF CROSS-CONTAMINATION DUE TO WIDE APPLICATION OF CEREALS AND GRAINS IN FOOD INDUSTRY TO BOOST MARKET
- 10.8 OTHER FOOD PRODUCTS
11 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY SITE
- 11.1 INTRODUCTION
- 11.2 IN-HOUSE LABORATORIES (FACTORY LABS)
- 11.2.1 NEED TO MAINTAIN BRAND REPUTATION AND CONSUMER TRUST TO DRIVE USE OF IN-HOUSE LABORATORIES
- 11.3 OUTSOURCING FACILITIES (SERVICE LABS)
- 11.3.1 NEED TO ACCESS SPECIALIZED EXPERTISE AND ADVANCED TECHNOLOGIES TO DRIVE DEMAND FOR OUTSOURCING LABORATORIES
- 11.4 GOVERNMENT LABS
- 11.4.1 INDEPENDENT AND IMPARTIAL TESTING PROVIDED BY GOVERNMENT LABS TO FUEL MARKET GROWTH
12 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY TECHNOLOGY (QUALITATIVE)
- 12.1 INTRODUCTION
- 12.2 MOLECULAR METHODS
- 12.3 IMMUNOLOGICAL METHODS
- 12.4 CULTURAL AND TRADITIONAL METHODS
- 12.5 SENSOR AND BIOSENSOR TECHNOLOGIES
- 12.6 EMERGING RAPID AND POINT OF CARE TECHNOLOGIES
13 FOOD PATHOGEN SAFETY TESTING EQUIPMENT AND SUPPLIES MARKET, BY REGION
- 13.1 INTRODUCTION
- 13.2 NORTH AMERICA
- 13.2.1 US
- 13.2.1.1 Stringent food safety regulations to stimulate market
- 13.2.2 CANADA
- 13.2.2.1 Government initiatives aimed at reducing pathogens in meat and poultry to boost growth
- 13.2.3 MEXICO
- 13.2.3.1 Growing food exports to US and need for stringent safety measures to boost market
- 13.2.1 US
- 13.3 EUROPE
- 13.3.1 GERMANY
- 13.3.1.1 Rising concerns over foodborne illnesses to drive market
- 13.3.2 FRANCE
- 13.3.2.1 Prevalence of multiple product recalls in country to stimulate growth
- 13.3.3 UK
- 13.3.3.1 Government funding and support initiatives for tracking foodborne pathogens to propel market
- 13.3.4 ITALY
- 13.3.4.1 Thriving tourism and hospitality industry to boost growth
- 13.3.5 SPAIN
- 13.3.5.1 Ongoing struggle with listeriosis and other foodborne illnesses to spur growth
- 13.3.6 POLAND
- 13.3.6.1 Growing dairy industry and exports to Ukraine to drive growth
- 13.3.7 REST OF EUROPE
- 13.3.1 GERMANY
- 13.4 ASIA PACIFIC
- 13.4.1 CHINA
- 13.4.1.1 Government regulations and certifications for food safety to propel market
- 13.4.2 JAPAN
- 13.4.2.1 Rising incidences of food poisoning and increased food safety tests to drive market
- 13.4.3 INDIA
- 13.4.3.1 Growing food processing market to boost demand for food pathogen testing equipment
- 13.4.4 AUSTRALIA & NEW ZEALAND
- 13.4.4.1 Fear of outbreaks of Salmonella and E. coli to boost demand for food pathogen testing equipment
- 13.4.5 SOUTHEAST ASIA
- 13.4.5.1 Increasing awareness regarding safety and quality of food to encourage adoption of food pathogen testing equipment
- 13.4.6 REST OF ASIA PACIFIC
- 13.4.1 CHINA
- 13.5 SOUTH AMERICA
- 13.5.1 BRAZIL
- 13.5.1.1 Booming food processing industry to spur market
- 13.5.2 ARGENTINA
- 13.5.2.1 Awareness among consumers regarding mounting cases of foodborne illnesses to contribute to market growth
- 13.5.3 REST OF SOUTH AMERICA
- 13.5.1 BRAZIL
- 13.6 REST OF THE WORLD
- 13.6.1 MIDDLE EAST
- 13.6.1.1 Heavy reliance on imported foods to drive demand for food pathogen safety testing equipment
- 13.6.2 AFRICA
- 13.6.2.1 Increasing international collaborations dedicated to elevating food safety standards to drive demand
- 13.6.1 MIDDLE EAST
14 COMPETITIVE LANDSCAPE
- 14.1 OVERVIEW
- 14.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 14.3 REVENUE ANALYSIS, 2022-2024
- 14.4 MARKET SHARE ANALYSIS, 2025
- 14.5 COMPANY VALUATION AND FINANCIAL METRICS
- 14.5.1 COMPANY VALUATION
- 14.5.2 EV/EBITDA
- 14.6 BRAND COMPARISON ANALYSIS
- 14.7 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 14.7.1 STARS
- 14.7.2 EMERGING LEADERS
- 14.7.3 PERVASIVE PLAYERS
- 14.7.4 PARTICIPANTS
- 14.7.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 14.8 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 14.8.1 PROGRESSIVE COMPANIES
- 14.8.2 RESPONSIVE COMPANIES
- 14.8.3 DYNAMIC COMPANIES
- 14.8.4 STARTING BLOCKS
- 14.8.5 COMPETITIVE BENCHMARKING: KEY STARTUPS/SMES, 2025
- 14.9 COMPETITIVE SCENARIO AND TRENDS
- 14.9.1 PRODUCT LAUNCHES
- 14.9.2 DEALS
- 14.9.3 EXPANSIONS
- 14.9.4 OTHER DEVELOPMENTS
15 COMPANY PROFILES
- 15.1 KEY PLAYERS
- 15.1.1 THERMO FISHER SCIENTIFIC INC.
- 15.1.1.1 Business overview
- 15.1.1.2 Products/Solutions/Services offered
- 15.1.1.3 Recent developments
- 15.1.1.3.1 Deals
- 15.1.1.3.2 Expansions
- 15.1.1.3.3 Other developments
- 15.1.1.4 MnM view
- 15.1.1.4.1 Key strengths
- 15.1.1.4.2 Strategic choices made
- 15.1.1.4.3 Weaknesses and competitive threats
- 15.1.2 BIO-RAD LABORATORIES, INC.
- 15.1.2.1 Business overview
- 15.1.2.2 Products/Solutions/Services offered
- 15.1.2.3 Recent developments
- 15.1.2.3.1 Product launches
- 15.1.2.4 MnM view
- 15.1.2.4.1 Key strengths
- 15.1.2.4.2 Strategic choices
- 15.1.2.4.3 Weaknesses and competitive threats
- 15.1.3 MERCK KGAA
- 15.1.3.1 Business overview
- 15.1.3.2 Products/Solutions/Services offered
- 15.1.3.3 Recent developments
- 15.1.3.3.1 Expansions
- 15.1.3.4 MnM view
- 15.1.3.4.1 Key strengths
- 15.1.3.4.2 Strategic choices
- 15.1.3.4.3 Weaknesses and competitive threats
- 15.1.4 NEOGEN CORPORATION
- 15.1.4.1 Business overview
- 15.1.4.2 Products/Solutions/Services offered
- 15.1.4.3 Recent developments
- 15.1.4.3.1 Product launches
- 15.1.4.3.2 Deals
- 15.1.4.4 MnM view
- 15.1.4.4.1 Key strengths
- 15.1.4.4.2 Strategic choices
- 15.1.4.4.3 Weaknesses and competitive threats
- 15.1.5 SHIMADZU CORPORATION
- 15.1.5.1 Business overview
- 15.1.5.2 Products/Solutions/Services offered
- 15.1.5.3 Recent developments
- 15.1.5.3.1 Product launches
- 15.1.5.4 MnM view
- 15.1.5.4.1 Key strengths
- 15.1.5.4.2 Strategic choices made
- 15.1.5.4.3 Weaknesses and competitive threats
- 15.1.6 BIOMERIEUX
- 15.1.6.1 Business overview
- 15.1.6.2 Products/Solutions/Services offered
- 15.1.6.3 Recent developments
- 15.1.6.3.1 Product launches
- 15.1.6.3.2 Deals
- 15.1.6.4 MnM view
- 15.1.7 AGILENT TECHNOLOGIES, INC.
- 15.1.7.1 Business overview
- 15.1.7.2 Products/Solutions/Services offered
- 15.1.7.3 Recent developments
- 15.1.7.3.1 Expansions
- 15.1.7.4 MnM view
- 15.1.8 QIAGEN
- 15.1.8.1 Business overview
- 15.1.8.2 Products/Solutions/Services offered
- 15.1.8.3 Recent developments
- 15.1.8.3.1 Product launches
- 15.1.8.3.2 Deals
- 15.1.8.4 MnM view
- 15.1.9 BRUKER
- 15.1.9.1 Business overview
- 15.1.9.2 Products/Solutions/Services offered
- 15.1.9.3 Recent developments
- 15.1.9.3.1 Deals
- 15.1.9.4 MnM view
- 15.1.10 PERKINELMER INC.
- 15.1.10.1 Business overview
- 15.1.10.2 Products/Solutions/Services offered
- 15.1.10.3 Recent developments
- 15.1.10.3.1 Product launches
- 15.1.10.3.2 Expansions
- 15.1.10.4 MnM view
- 15.1.11 HYGIENA, LLC
- 15.1.11.1 Business overview
- 15.1.11.2 Products/Solutions/Services offered
- 15.1.11.3 Recent developments
- 15.1.11.3.1 Product launches
- 15.1.11.3.2 Deals
- 15.1.11.4 MnM view
- 15.1.12 SOLABIA GROUP (BIOKAR)
- 15.1.12.1 Business overview
- 15.1.12.2 Products/Solutions/Services offered
- 15.1.12.3 Recent developments
- 15.1.12.3.1 Deals
- 15.1.12.4 MnM view
- 15.1.13 ROKA BIO SCIENCE
- 15.1.13.1 Business overview
- 15.1.13.2 Products/Solutions/Services offered
- 15.1.13.3 Recent developments
- 15.1.13.4 MnM view
- 15.1.14 PROMEGA CORPORATION
- 15.1.14.1 Business overview
- 15.1.14.2 Products/Solutions/Services offered
- 15.1.14.3 Recent developments
- 15.1.14.3.1 Deals
- 15.1.14.4 MnM view
- 15.1.15 ROMER LABS DIVISION HOLDING
- 15.1.15.1 Business overview
- 15.1.15.2 Products/Solutions/Services offered
- 15.1.15.3 Recent developments
- 15.1.15.4 MnM view
- 15.1.1 THERMO FISHER SCIENTIFIC INC.
- 15.2 STARTUPS/SMES
- 15.2.1 CHARM SCIENCES
- 15.2.1.1 Business overview
- 15.2.1.2 Products/Solutions/Services offered
- 15.2.1.3 Recent developments
- 15.2.1.3.1 Other developments
- 15.2.1.4 MnM view
- 15.2.2 MICROBIOLOGICS, INC.
- 15.2.2.1 Business overview
- 15.2.2.2 Products/Solutions/Services offered
- 15.2.2.3 Recent developments
- 15.2.2.3.1 Deals
- 15.2.2.4 MnM view
- 15.2.3 R-BIOPHARM
- 15.2.3.1 Business overview
- 15.2.3.2 Products/Solutions/Services offered
- 15.2.3.3 Recent developments
- 15.2.3.3.1 Deals
- 15.2.3.4 MnM view
- 15.2.4 CONDALAB
- 15.2.4.1 Business overview
- 15.2.4.2 Products/Solutions/Services offered
- 15.2.4.3 Recent developments
- 15.2.4.4 MnM view
- 15.2.5 CHROMAGAR
- 15.2.5.1 Business overview
- 15.2.5.2 Products/Solutions/Services offered
- 15.2.5.3 Recent developments
- 15.2.5.4 MnM view
- 15.2.6 GOLD STANDARD DIAGNOSTICS
- 15.2.7 CLEAR LABS, INC.
- 15.2.8 RING BIOTECHNOLOGY CO LTD.
- 15.2.9 NEMIS TECHNOLOGIES AG
- 15.2.10 PATHOGENDX CORPORATION
- 15.2.1 CHARM SCIENCES
16 RESEARCH METHODOLOGY
- 16.1 RESEARCH DATA
- 16.1.1 SECONDARY DATA
- 16.1.1.1 Key data from secondary sources
- 16.1.2 PRIMARY DATA
- 16.1.2.1 Key data from primary sources
- 16.1.2.2 Breakdown of primary profiles
- 16.1.2.3 Key insights from industry experts
- 16.1.1 SECONDARY DATA
- 16.2 MARKET SIZE ESTIMATION
- 16.2.1 TOP-DOWN APPROACH
- 16.2.2 SUPPLY-SIDE ANALYSIS
- 16.2.3 BOTTOM-UP APPROACH (DEMAND SIDE)
- 16.3 DATA TRIANGULATION
- 16.4 RESEARCH ASSUMPTIONS
- 16.5 RESEARCH LIMITATIONS AND RISK ASSESSMENT
17 ADJACENT AND RELATED MARKETS
- 17.1 INTRODUCTION
- 17.2 LIMITATIONS
- 17.3 FOOD SAFETY TESTING MARKET
- 17.3.1 MARKET DEFINITION
- 17.3.2 MARKET OVERVIEW
- 17.4 FOOD DIAGNOSTICS MARKET
- 17.4.1 MARKET DEFINITION
- 17.4.2 MARKET OVERVIEW
18 APPENDIX
- 18.1 DISCUSSION GUIDE
- 18.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 18.3 CUSTOMIZATION OPTIONS
- 18.4 RELATED REPORTS
- 18.5 AUTHOR DETAILS





