
|
시장보고서
상품코드
1984858
멸균 장비 시장 : 제품별, 기술별, 지역별 - 예측(-2031년)Sterilization Equipment Market by Product (Instruments, Accessories, Service), Technology (Heat, Low-temperature, Radiation ), Region - Global Forecast to 2031 |
||||||
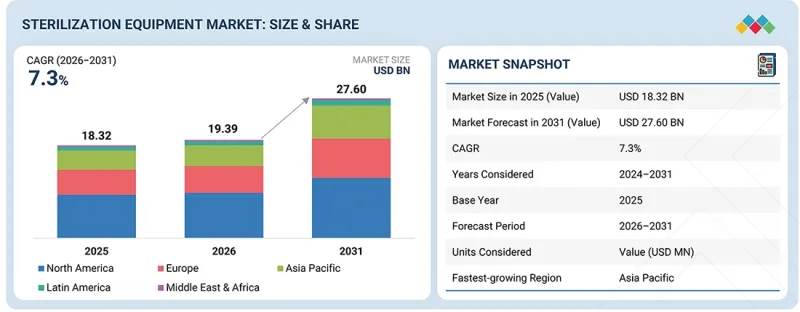
세계의 멸균 장비 시장 규모는 2026년에 약 193억 9,000만 달러, 2031년까지 276억 달러에 이를 것으로 예측되어 CAGR로 7.3%의 성장이 전망되고 있습니다.
| 조사 범위 | |
|---|---|
| 조사 대상 기간 | 2024-2031년 |
| 기준연도 | 2025년 |
| 예측 기간 | 2025-2031년 |
| 단위 | 10억 달러 |
| 부문 | 제품 및 서비스, 기술, 최종사용자 |
| 대상 지역 | 북미, 유럽, 아시아태평양, 라틴아메리카, 중동 및 아프리카 |
의료시설이 감염 대책과 인프라 개발을 우선시하는 가운데, 시장은 꾸준한 성장세를 보이고 있습니다. 또한 병원, 제약회사, 의료기기 제조업체의 멸균 서비스에 대한 수요도 증가하고 있습니다. 의료기기의 복잡성 증가와 함께 멸균 공정에 대한 규제 요건이 강화됨에 따라 전 세계적으로 멸균 장비에 대한 수요가 급증하고 있습니다.
"멸균 소모품 및 액세서리 부문은 예측 기간 동안 가장 높은 CAGR로 성장할 것으로 예측됩니다. "
이 부문의 성장은 주로 모든 멸균 사이클에 필요한 인디케이터, 파우치, 랩과 같은 멸균 소모품에 대한 지속적인 수요에 의해 촉진되고 있습니다. 수술 건수 증가, 입원 환자 수 증가, 감염 관리 및 규제 준수에 대한 요구가 증가함에 따라 의료시설과 생명과학 산업에서 더 많은 멸균 소모품 및 액세서리가 사용되고 있습니다.
"열/고온 살균 부문이 2025년 멸균 장비 시장에서 가장 큰 점유율을 차지했습니다. "
병원, 연구소, 제약 제조 시설에서 증기 멸균이 일반적으로 사용되기 때문에 이 부문이 시장을 독점하고 있습니다. 의료 분야에서는 열을 이용한 멸균 방법이 모든 유형의 미생물로부터 수술기구 및 의료기기를 효과적으로 세척하면서 저비용으로 신뢰할 수 있는 결과를 얻을 수 있기 때문에 선호되고 있습니다. 수술 건수 증가와 엄격한 감염 관리 정책의 시행으로 의료 현장에서는 고온 멸균 기술에 대한 수요가 지속적으로 증가하고 있습니다.
"지역별로는 아시아태평양이 멸균기기 시장에서 가장 높은 성장률을 나타낼 것으로 예측됩니다. "
개발도상국의 급속한 경제 성장, 의료비 증가, 지역 내 의료시설 확충 등 여러 요인으로 인해 이 지역이 가장 높은 성장률을 보이고 있습니다. 또한, 급성/만성 질환의 유병률 증가, 수술 건수 증가, 제약 및 의료기기 제조 산업의 확장은 아시아태평양의 멸균 장비에 대한 수요를 촉진하고 있습니다.
세계의 멸균기기 시장에 대해 조사 분석했으며, 주요 촉진요인 및 저해요인, 제품 개발 및 혁신, 경쟁 구도에 대해 조사 분석하여 전해드립니다.
자주 묻는 질문
목차
제1장 서론
제2장 주요 요약
제3장 중요 지견
제4장 시장 개요
제5장 업계 동향
제6장 기술 진보, AI의 영향, 특허, 혁신, 향후 용도
제7장 규제 상황과 지속가능성 이니셔티브
제8장 고객 현황과 구매 행동
제9장 멸균 장비 시장 : 제품 및 서비스별
제10장 멸균 장비 시장 : 기술별
제11장 멸균 장비 시장 : 최종사용자별
제12장 멸균 장비 시장, 수량 분석
제13장 멸균 장비 시장 : 지역별
제14장 경쟁 구도
제15장 기업 개요
제16장 조사 방법
제17장 부록
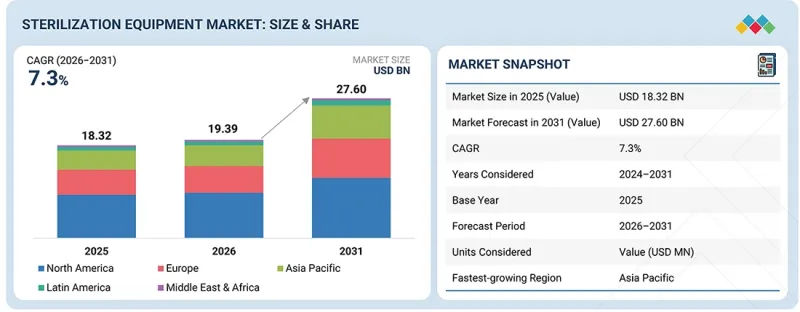
LSH 26.04.13The sterilization equipment market was valued at approximately USD 19.39 billion in 2026 and is projected to reach USD 27.60 billion by 2031, growing at a CAGR of 7.3%.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2025-2031 |
| Units Considered | Value (USD billion) |
| Segments | Product & Services, Technology, and End User |
| Regions covered | North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa |
The market is experiencing steady growth as healthcare facilities prioritize infection control measures and infrastructure development. Additionally, hospitals, pharmaceutical companies, and medical device manufacturers are increasingly seeking sterilization services. The growing complexity of medical devices, coupled with stringent regulatory requirements for sterilization processes, has driven a worldwide surge in demand for sterilization equipment.
"Sterilization consumables & accessories segment is expected to grow at the highest CAGR during the forecast period."
The growth of this segment is mainly driven by recurring demand for sterilization consumables, such as indicators, pouches, and wraps, which are required for every sterilization cycle. The increase in surgical procedures, along with rising hospital admissions and the growing need for infection control and regulatory compliance, has led healthcare facilities and the life sciences industry to use more sterilization consumables and accessories.
"Heat/high-temperature sterilization segment held the largest share of the sterilization equipment market in 2025."
The segment has dominated the market because hospitals, laboratories, and pharmaceutical manufacturing facilities commonly use steam sterilization. The medical field prefers heat-based sterilization methods because they deliver reliable results at a lower cost while effectively cleaning surgical instruments and medical equipment of all types of microorganisms. The healthcare environment continues to demand high-temperature sterilization technologies due to both the increasing number of surgical procedures and the implementation of strict infection control policies.
"Asia Pacific will experience the fastest growth rate in the sterilization equipment market, by region."
The region is experiencing the fastest growth rate due to several key factors, including rapid economic growth in developing nations, rising healthcare spending, and the development of healthcare facilities throughout the area. Additionally, the rising prevalence of acute and chronic diseases, the growing number of surgeries, and the expanding presence of pharmaceutical & medical device manufacturing industries are driving demand for sterilization equipment in the Asia Pacific region.
The breakdown of the profiles of primary participants in the sterilization equipment market is provided below:
- By Company Type: Tier 1 - 26%, Tier 2 - 44%, and Tier 3 - 30%
- By Designation: C-level - 30%, D-level - 34%, and Others - 36%
- By Region: North America - 51%, Europe - 21%, Asia Pacific - 18%, Latin America - 6%, and Middle East & Africa - 4%
The key players in the sterilization equipment market are STERIS (US), Getinge AB (Sweden), Fortive (US), Solventum (US), Sotera Health (US), Cardinal Health (US), Stryker Corporation (US), Merck KGaA (Germany), MMM Group (Germany), ANTONIO MATACHANA, S.A. (Spain), Miele Group (Germany), Tuttnauer (Netherlands), Andersen Sterilizers (US), Noxilizer, Inc. (US), DE LAMA S.P.A. (Italy), C.B.M. S.r.l. Medical Equipment (Italy), E-BEAM Services Inc. (US), Life Science Outsourcing, Inc. (US), Systec GmbH & Co. KG (Germany), Continental Equipment Company (US), Midwest Sterilization Corp. (US), BGS Beta-Gamma-Service GmbH & Co. KG (Germany), ACMAS Technologies (P) Ltd. (India), Celitron Medical Technologies Kft (Hungary), and Prince Sterilization Services, LLC (US).
Research Coverage:
This research report categorizes the sterilization equipment market by product & service (sterilization instruments, sterilization services, and sterilization consumables & accessories), technology (heat/high-temperature sterilization, low-temperature sterilization, and ionizing radiation sterilization), end user (hospitals & clinics, medical device companies, pharmaceutical companies, food & beverages companies, and other end users), and region (North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa). The scope of the report covers detailed information regarding the major factors, such as drivers, restraints, opportunities, and challenges influencing the growth of the sterilization equipment market. A detailed analysis of key industry players has been conducted to provide insights into their business overviews, solutions, key strategies, such as partnerships, collaborations, expansions, agreements, new product launches, and recent developments in the sterilization equipment market. A competitive analysis of upcoming startups in the sterilization equipment market ecosystem is covered in this report.
Reasons to buy this report:
The report will help market leaders/new entrants in this market by providing information on the closest approximations of revenue for the overall sterilization equipment market and its subsegments. This report will help stakeholders understand the competitive landscape and gain deeper insights to better position their businesses and plan suitable go-to-market strategies. The report also helps stakeholders understand the pulse of the market and provides them with information on key market drivers, restraints, opportunities, and challenges.
The report provides insights into the following pointers:
- Analysis of key drivers (rising prevalence of HAIs, increasing number of surgical procedures, growing geriatric population and the increasing incidence of chronic diseases, increasing emphasis on sterilization & disinfection of food, increasing outsourcing of sterilization services by pharmaceutical companies and hospitals, advancements in sterilization equipment technology), opportunities (increasing number of medical device and pharmaceutical companies in emerging economies, rising use of e-beam sterilization, reintegration of ethylene oxide sterilization, rising focus on infection prevention in non-healthcare sectors), restraints (sterilization of advanced medical instruments, adverse effects of chemical disinfectants), and challenges (end-user non-compliance with sterilization standards, increasing adoption of single-use medical devices) influencing the growth of the sterilization equipment market.
- Product Development/Innovation: Detailed insights on upcoming technologies, research & development activities, and new product launches in the sterilization equipment market.
- Market Development: Comprehensive information about lucrative markets-the report analyzes the sterilization equipment market across varied regions.
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the sterilization equipment market.
Competitive Assessment: In-depth assessment of market shares, growth strategies, and product offerings of leading players like STERIS (US), Getinge AB (Sweden), Fortive (US), Solventum (US), and Sotera Health (US).
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKETS COVERED AND REGIONAL SCOPE
- 1.3.2 REGIONAL SCOPE
- 1.3.3 INCLUSIONS AND EXCLUSIONS
- 1.3.4 YEARS CONSIDERED
- 1.3.5 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS AND MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: SHARE INSIGHTS AND STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS SHAPING MARKET
- 2.4 FASTEST-GROWTH SEGMENTS AND EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 STERILIZATION EQUIPMENT MARKET OVERVIEW
- 3.2 STERILIZATION EQUIPMENT MARKET, BY PRODUCT & SERVICE, 2026 VS. 2031
- 3.3 STERILIZATION EQUIPMENT MARKET, BY TECHNOLOGY, 2026 VS. 2031
- 3.4 STERILIZATION EQUIPMENT MARKET, BY END USER, 2026 VS. 2031
- 3.5 STERILIZATION EQUIPMENT MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Rising prevalence of HAIs
- 4.2.1.2 Increasing number of surgical procedures
- 4.2.1.3 Growing geriatric population and increasing incidence of chronic diseases
- 4.2.1.4 Increasing emphasis on sterilization & disinfection of food
- 4.2.1.5 Increasing outsourcing of sterilization services by pharmaceutical companies and hospitals
- 4.2.1.6 Advancements in sterilization equipment technology
- 4.2.2 RESTRAINTS
- 4.2.2.1 Sterilization of advanced medical instruments
- 4.2.2.2 Adverse effects of chemical disinfectants
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Increasing number of medical device and pharmaceutical companies in emerging economies
- 4.2.3.2 Rising use of E-beam sterilization
- 4.2.3.3 Reintegration of ethylene oxide sterilization
- 4.2.3.4 Rising focus on infection prevention in non-healthcare sectors
- 4.2.4 CHALLENGES
- 4.2.4.1 End-user non-compliance with sterilization standards
- 4.2.4.2 Increasing adoption of single-use medical devices
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS AND WHITE SPACES
- 4.3.1 UNMET NEEDS IN STERILIZATION EQUIPMENT MARKET
- 4.3.2 WHITE SPACE OPPORTUNITIES
- 4.4 INTERCONNECTED MARKETS AND CROSS-SECTOR OPPORTUNITIES
- 4.4.1 INTERCONNECTED MARKETS
- 4.4.2 CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER 1/2/3 PLAYERS
- 4.5.1 KEY MOVES AND STRATEGIC FOCUS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 BARGAINING POWER OF SUPPLIERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMIC INDICATORS
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS AND FORECAST
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.4 VALUE CHAIN ANALYSIS
- 5.5 ECOSYSTEM ANALYSIS
- 5.5.1 STERILIZATION EQUIPMENT MARKET: ROLE OF COMPANIES IN ECOSYSTEM
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE OF STERILIZATION EQUIPMENT, BY PRODUCT, 2024-2026
- 5.6.2 AVERAGE SELLING PRICE OF STERILIZATION EQUIPMENT, BY KEY PLAYER, 2024-2026
- 5.6.3 AVERAGE SELLING PRICE OF STERILIZATION EQUIPMENT PRODUCTS, BY REGION, 2024-2026
- 5.7 TRADE ANALYSIS
- 5.7.1 IMPORT SCENARIO (HS CODE 841920)
- 5.7.2 EXPORT SCENARIO (HS CODE 841920)
- 5.8 KEY CONFERENCES & EVENTS, 2026-2027
- 5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- 5.10 INVESTMENT & FUNDING SCENARIO
- 5.11 CASE STUDY ANALYSIS
- 5.11.1 CASE STUDY 1: STERILIZATION OF 20 BILLION MEDICAL DEVICES BY ETHYLENE OXIDE (ETO): CONSEQUENCES OF ETO CLOSURES AND ALTERNATIVE STERILIZATION TECHNOLOGIES/SOLUTIONS
- 5.11.2 CASE STUDY 2: VAPORIZED HYDROGEN PEROXIDE (VHP) BIODECONTAMINATION: SUPERIOR PROCESS
- 5.11.3 CASE STUDY 3: IMPACT OF ENHANCED CLEANING ON BACTERIAL CONTAMINATION OF HOSPITAL ENVIRONMENTAL SURFACES: CLINICAL TRIAL IN CRITICAL CARE UNIT, EGYPT
- 5.12 IMPACT OF 2025 US TARIFF ON STERILIZATION EQUIPMENT MARKET
- 5.12.1 INTRODUCTION
- 5.12.2 KEY TARIFF RATES
- 5.12.3 PRICE IMPACT ANALYSIS
- 5.12.4 IMPACT ON REGION
- 5.12.4.1 North America
- 5.12.4.2 Europe
- 5.12.4.3 Asia Pacific
- 5.12.5 IMPACT ON END-USE INDUSTRIES
- 5.12.5.1 Hospitals and clinics
- 5.12.5.2 Medical device companies
- 5.12.5.3 Pharmaceutical and life science industries
- 5.12.5.4 Food industry
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 KEY EMERGING TECHNOLOGIES
- 6.1.1 ULTRA-HIGH-PRESSURE STERILIZATION
- 6.1.2 NITROGEN DIOXIDE (NO2) STERILIZATION
- 6.2 COMPLEMENTARY TECHNOLOGIES
- 6.2.1 SUPERCRITICAL CO2 TECHNOLOGY
- 6.2.2 PULSED ELECTRIC FIELD (PEF) STERILIZATION
- 6.2.3 PLASMA STERILIZATION
- 6.3 TECHNOLOGY/PRODUCT ROADMAP
- 6.4 PATENT ANALYSIS
- 6.4.1 LIST OF MAJOR PATENTS
- 6.5 FUTURE APPLICATIONS
- 6.6 IMPACT OF AI/GENERATIVE AI ON STERILIZATION EQUIPMENT MARKET
- 6.6.1 INTRODUCTION
- 6.6.2 TOP USE CASES AND MARKET POTENTIAL
- 6.6.3 KEY COMPANIES IMPLEMENTING AI
- 6.6.4 FUTURE OF AI
7 REGULATORY LANDSCAPE AND SUSTAINABILITY INITIATIVES
- 7.1 REGIONAL REGULATIONS AND COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.2 SUSTAINABILITY INITIATIVES
- 7.2.1 ENVIRONMENTAL IMPACT AND ECO-FRIENDLY INITIATIVES IN STERILIZATION EQUIPMENT
- 7.2.1.1 Eco-friendly Initiatives
- 7.2.1 ENVIRONMENTAL IMPACT AND ECO-FRIENDLY INITIATIVES IN STERILIZATION EQUIPMENT
- 7.3 SUSTAINABILITY IMPACT AND REGULATORY POLICY INITIATIVES
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS AND BUYING EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 BUYING CRITERIA
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS IN VARIOUS END-USE INDUSTRIES
- 8.5 MARKET PROFITABILITY
- 8.5.1 REVENUE POTENTIAL
- 8.5.2 COST DYNAMICS
- 8.5.3 MARGIN OPPORTUNITIES IN KEY APPLICATIONS
9 STERILIZATION EQUIPMENT MARKET, BY PRODUCT & SERVICE
- 9.1 INTRODUCTION
- 9.2 STERILIZATION INSTRUMENTS
- 9.2.1 RISING DEMAND FOR PATIENT SAFETY IN HEALTHCARE SETTINGS TO DRIVE MARKET
- 9.3 STERILIZATION SERVICES
- 9.3.1 ON-SITE STERILIZATION
- 9.3.1.1 Low operational costs and lesser turnaround time to boost demand
- 9.3.2 OFF-SITE STERILIZATION
- 9.3.2.1 Rising trend for outsourcing sterilization services to drive market
- 9.3.1 ON-SITE STERILIZATION
- 9.4 STERILIZATION CONSUMABLES & ACCESSORIES
- 9.4.1 STERILANTS
- 9.4.1.1 Rising adoption of advanced medical devices to drive demand for sterilants
- 9.4.2 STERILIZATION CONTAINERS
- 9.4.2.1 Increasing focus on safe instrument transport and reusability to propel demand
- 9.4.3 INDICATORS
- 9.4.3.1 Chemical indicators
- 9.4.3.1.1 Low cost of chemical indicators to aid adoption
- 9.4.3.2 Biological indicators
- 9.4.3.2.1 Efficacy of biological indicators to drive demand
- 9.4.3.1 Chemical indicators
- 9.4.4 POUCHES & WRAPS
- 9.4.4.1 Technological advancements in sterilization pouches & wraps to support growth
- 9.4.5 OTHER STERILIZATION CONSUMABLES & ACCESSORIES
- 9.4.1 STERILANTS
10 STERILIZATION EQUIPMENT MARKET, BY TECHNOLOGY
- 10.1 INTRODUCTION
- 10.2 HEAT/HIGH-TEMPERATURE STERILIZATION
- 10.2.1 STRINGENT GOVERNMENT REGULATIONS ON BIOMEDICAL WASTE MANAGEMENT TO DRIVE MARKET
- 10.2.2 MOIST HEAT/STEAM STERILIZATION
- 10.2.2.1 Moist heat/steam sterilization instruments
- 10.2.2.2 Moist heat/steam sterilization consumables & accessories
- 10.2.2.3 Moist heat/steam sterilization services
- 10.2.3 DRY HEAT STERILIZATION
- 10.2.3.1 Dry heat sterilization instruments
- 10.2.3.2 Dry heat sterilization consumables & accessories
- 10.2.3.3 Dry heat sterilization services
- 10.3 LOW-TEMPERATURE STERILIZATION
- 10.3.1 ABILITY TO STERILIZE HEAT-SENSITIVE MEDICAL DEVICES TO BOOST MARKET
- 10.3.2 HYDROGEN PEROXIDE STERILIZATION
- 10.3.2.1 Hydrogen peroxide sterilization instruments
- 10.3.2.2 Hydrogen peroxide sterilization consumables & accessories
- 10.3.2.3 Hydrogen peroxide sterilization services
- 10.3.3 ETHYLENE OXIDE STERILIZATION
- 10.3.3.1 Ethylene oxide sterilization instruments
- 10.3.3.2 Ethylene oxide sterilization consumables & accessories
- 10.3.3.3 Ethylene oxide sterilization services
- 10.3.4 OZONE-BASED STERILIZATION
- 10.3.4.1 Ozone-based sterilization instruments
- 10.3.4.2 Ozone-based sterilization consumables & accessories
- 10.3.4.3 Ozone-based sterilization services
- 10.3.5 FORMALDEHYDE STERILIZATION
- 10.3.5.1 Formaldehyde sterilization instruments
- 10.3.5.2 Formaldehyde sterilization consumables & accessories
- 10.3.5.3 Formaldehyde sterilization services
- 10.3.6 OTHER LOW-TEMPERATURE STERILIZATION
- 10.3.6.1 Other low-temperature sterilization instruments
- 10.3.6.2 Other low-temperature sterilization consumables & accessories
- 10.3.6.3 Other low-temperature sterilization services
- 10.4 IONIZING RADIATION STERILIZATION
- 10.4.1 SAFETY, RELIABILITY, AND HIGH EFFECTIVENESS OF IONIZING RADIATION STERILIZATION TO DRIVE ADOPTION
- 10.4.2 E-BEAM RADIATION STERILIZATION
- 10.4.2.1 E-beam radiation sterilization instruments
- 10.4.2.2 E-beam radiation sterilization services
- 10.4.3 GAMMA RADIATION STERILIZATION
- 10.4.3.1 Gamma radiation sterilization instruments
- 10.4.3.2 Gamma radiation sterilization services
- 10.4.4 X-RAY STERILIZATION
- 10.4.4.1 X-ray sterilization instruments
- 10.4.4.2 X-ray sterilization services
11 STERILIZATION EQUIPMENT MARKET, BY END USER
- 11.1 INTRODUCTION
- 11.2 HOSPITALS & CLINICS
- 11.2.1 STRINGENT CLEANLINESS PROTOCOLS FOR RESTRICTION OF HAIS TO PROPEL MARKET
- 11.3 MEDICAL DEVICE COMPANIES
- 11.3.1 RISING TECHNOLOGICAL INNOVATIONS IN STERILIZATION EQUIPMENT PRODUCTS TO DRIVE MARKET
- 11.4 PHARMACEUTICAL COMPANIES
- 11.4.1 GROWING NEED FOR SAFETY ASSURANCE AND REGULATORY APPROVALS TO BOOST MARKET
- 11.5 FOOD & BEVERAGE COMPANIES
- 11.5.1 RISING INCIDENCE OF FOODBORNE DISEASES TO SUPPORT MARKET GROWTH
- 11.6 OTHER END USERS
12 STERILIZATION EQUIPMENT MARKET, VOLUME ANALYSIS
- 12.1 INTRODUCTION
- 12.1.1 NORTH AMERICA: NUMBER OF TERMINAL STERILIZATION CYCLES CONDUCTED IN HOSPITALS, BY COUNTRY, 2024 -2031 (MILLION)
13 STERILIZATION EQUIPMENT MARKET, BY REGION
- 13.1 INTRODUCTION
- 13.2 NORTH AMERICA
- 13.2.1 US
- 13.2.1.1 Presence of well-established pharmaceutical and medical device companies to drive growth
- 13.2.2 CANADA
- 13.2.2.1 Growing number of surgical procedures to propel market
- 13.2.1 US
- 13.3 EUROPE
- 13.3.1 GERMANY
- 13.3.1.1 Stringent regulations for food, pharmaceutical, and medical device industries to fuel growth
- 13.3.2 UK
- 13.3.2.1 Growing patient population and rising surgical volume to spur growth
- 13.3.3 FRANCE
- 13.3.3.1 Highly advanced and well-established healthcare system to drive demand for sterilization equipment
- 13.3.4 ITALY
- 13.3.4.1 Growing geriatric population and improved life expectancy to support market growth
- 13.3.5 SPAIN
- 13.3.5.1 Government efforts to improve health infrastructure to fuel demand
- 13.3.6 REST OF EUROPE
- 13.3.1 GERMANY
- 13.4 ASIA PACIFIC
- 13.4.1 JAPAN
- 13.4.1.1 Growing geriatric population and increasing incidence of chronic diseases to drive market
- 13.4.2 CHINA
- 13.4.2.1 Rising number of hospitals and increasing healthcare expenditure to boost adoption
- 13.4.3 INDIA
- 13.4.3.1 Infrastructural improvements to support growth
- 13.4.4 AUSTRALIA
- 13.4.4.1 Increasing demand for healthcare services to support market growth
- 13.4.5 SOUTH KOREA
- 13.4.5.1 Increasing government efforts to broaden healthcare coverage to drive market
- 13.4.6 REST OF ASIA PACIFIC
- 13.4.1 JAPAN
- 13.5 LATIN AMERICA
- 13.5.1 BRAZIL
- 13.5.1.1 Rising number of cosmetic surgeries to boost adoption of sterilization equipment
- 13.5.2 MEXICO
- 13.5.2.1 Growing medical tourism industry to support market growth
- 13.5.3 REST OF LATIN AMERICA
- 13.5.1 BRAZIL
- 13.6 MIDDLE EAST & AFRICA
- 13.6.1 SAUDI ARABIA
- 13.6.1.1 Increasing expenditure on healthcare infrastructure and services to drive market
- 13.6.2 UAE
- 13.6.2.1 Government initiatives and increasing public-private partnerships to support market growth
- 13.6.3 REST OF MIDDLE EAST & AFRICA
- 13.6.1 SAUDI ARABIA
14 COMPETITIVE LANDSCAPE
- 14.1 INTRODUCTION
- 14.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 14.2.1 OVERVIEW OF STRATEGIES DEPLOYED BY PLAYERS IN STERILIZATION EQUIPMENT MARKET
- 14.3 REVENUE ANALYSIS, 2021-2025
- 14.4 MARKET SHARE ANALYSIS, 2025
- 14.4.1 GLOBAL MARKET SHARE ANALYSIS, 2025
- 14.4.2 US MARKET SHARE ANALYSIS, 2025
- 14.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 14.5.1 STARS
- 14.5.2 EMERGING LEADERS
- 14.5.3 PERVASIVE PLAYERS
- 14.5.4 PARTICIPANTS
- 14.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 14.5.5.1 Company footprint
- 14.5.5.2 Region footprint
- 14.5.5.3 Product & service footprint
- 14.5.5.4 Technology footprint
- 14.5.5.5 End-user footprint
- 14.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 14.6.1 PROGRESSIVE COMPANIES
- 14.6.2 RESPONSIVE COMPANIES
- 14.6.3 DYNAMIC COMPANIES
- 14.6.4 STARTING BLOCKS
- 14.6.5 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 14.6.5.1 Detailed list of key startups/SMEs
- 14.6.5.2 Competitive benchmarking of startups/SMEs (1/2)
- 14.6.5.3 Competitive benchmarking of startups/SMEs (2/2)
- 14.7 COMPANY VALUATION & FINANCIAL METRICS
- 14.7.1 FINANCIAL METRICS
- 14.7.2 COMPANY VALUATION
- 14.8 BRAND/PRODUCT COMPARISON
- 14.9 COMPETITIVE SCENARIO
- 14.9.1 PRODUCT LAUNCHES & APPROVALS
- 14.9.2 DEALS
- 14.9.3 EXPANSIONS
15 COMPANY PROFILES
- 15.1 KEY PLAYERS
- 15.1.1 STERIS
- 15.1.1.1 Business overview
- 15.1.1.2 Products & services offered
- 15.1.1.3 Recent developments
- 15.1.1.3.1 Product launches & approvals
- 15.1.1.3.2 Deals
- 15.1.1.3.3 Expansions
- 15.1.1.4 MnM view
- 15.1.1.4.1 Key strengths
- 15.1.1.4.2 Strategic choices
- 15.1.1.4.3 Weaknesses & competitive threats
- 15.1.2 SOTERA HEALTH
- 15.1.2.1 Business overview
- 15.1.2.2 Products & services offered
- 15.1.2.3 Recent developments
- 15.1.2.3.1 Expansions
- 15.1.2.4 MnM view
- 15.1.2.4.1 Key strengths
- 15.1.2.4.2 Strategic choices
- 15.1.2.4.3 Weaknesses & competitive threats
- 15.1.3 FORTIVE
- 15.1.3.1 Business overview
- 15.1.3.2 Products & services offered
- 15.1.3.3 Recent developments
- 15.1.3.3.1 Product launches
- 15.1.3.4 MnM view
- 15.1.3.4.1 Key strengths
- 15.1.3.4.2 Strategic choices
- 15.1.3.4.3 Weaknesses & competitive threats
- 15.1.4 GETINGE AB
- 15.1.4.1 Business overview
- 15.1.4.2 Products & services offered
- 15.1.4.3 Recent developments
- 15.1.4.3.1 Product launches
- 15.1.4.3.2 Deals
- 15.1.4.3.3 Expansions
- 15.1.4.4 MnM view
- 15.1.4.4.1 Key strengths
- 15.1.4.4.2 Strategic choices
- 15.1.4.4.3 Weaknesses and competitive threats
- 15.1.5 SOLVENTUM
- 15.1.5.1 Business overview
- 15.1.5.2 Products & services offered
- 15.1.5.3 Recent developments
- 15.1.5.3.1 Product launches
- 15.1.5.3.2 Other developments
- 15.1.5.4 MnM view
- 15.1.5.4.1 Key strengths
- 15.1.5.4.2 Strategic choices
- 15.1.5.4.3 Weaknesses & competitive threats
- 15.1.6 CARDINAL HEALTH
- 15.1.6.1 Business overview
- 15.1.6.2 Products & services offered
- 15.1.6.3 Recent developments
- 15.1.6.3.1 Expansions
- 15.1.7 STRYKER CORPORATION
- 15.1.7.1 Business overview
- 15.1.7.2 Products & services offered
- 15.1.8 MERCK KGAA
- 15.1.8.1 Business overview
- 15.1.8.2 Products & services offered
- 15.1.9 MMM GROUP
- 15.1.9.1 Business overview
- 15.1.9.2 Products & services offered
- 15.1.10 ANTONIO MATACHANA, S.A.
- 15.1.10.1 Business overview
- 15.1.10.2 Products & services offered
- 15.1.1 STERIS
- 15.2 OTHER PLAYERS
- 15.2.1 MIELE GROUP (JOINT VENTURE WITH METALL ZUG)
- 15.2.2 TUTTNAUER (PART OF FORTISSIMO CAPITAL)
- 15.2.3 ANDERSEN STERILIZERS
- 15.2.4 NOXILIZER, INC.
- 15.2.5 DE LAMA S.P.A
- 15.2.6 C.B.M. S.R.L. MEDICAL EQUIPMENT
- 15.2.7 E-BEAM SERVICES, INC.
- 15.2.8 LIFE SCIENCE OUTSOURCING, INC.
- 15.2.9 SYSTEC GMBH & CO. KG
- 15.2.10 CONTINENTAL EQUIPMENT COMPANY
- 15.2.11 MIDWEST STERILIZATION CORPORATION
- 15.2.12 BGS BETA-GAMMA-SERVICE GMBH & CO. KG
- 15.2.13 ACMAS TECHNOLOGIES (P) LTD.
- 15.2.14 CELITRON MEDICAL TECHNOLOGIES KFT
- 15.2.15 PRINCE STERILIZATION SERVICES, LLC
16 RESEARCH METHODOLOGY
- 16.1 RESEARCH DATA
- 16.1.1 SECONDARY DATA
- 16.1.1.1 List of secondary sources
- 16.1.1.2 Key data from secondary sources
- 16.1.2 PRIMARY DATA
- 16.1.2.1 List of primary sources
- 16.1.2.2 Key data from primary sources
- 16.1.2.3 Key industry insights
- 16.1.2.4 Breakdown of primary interviews
- 16.1.1 SECONDARY DATA
- 16.2 MARKET SIZE ESTIMATION
- 16.2.1 TOTAL MARKET SIZE: STERILIZATION EQUIPMENT MARKET
- 16.2.2 BOTTOM-UP APPROACH
- 16.2.2.1 Approach 1: Revenue estimation of key players
- 16.2.2.2 Approach 2: Study of annual reports and investor presentations
- 16.2.2.3 Approach 3: Primary interviews
- 16.2.2.4 Growth forecast
- 16.2.2.5 CAGR projections
- 16.2.3 TOP-DOWN APPROACH
- 16.3 DATA TRIANGULATION
- 16.4 RESEARCH ASSUMPTIONS
- 16.4.1 STUDY-RELATED ASSUMPTIONS
- 16.5 STUDY ASSUMPTIONS
- 16.5.1 PARAMETRIC ASSUMPTIONS
- 16.5.2 GROWTH RATE ASSUMPTIONS
- 16.6 RESEARCH LIMITATIONS
- 16.7 RISK ASSESSMENT
17 APPENDIX
- 17.1 DISCUSSION GUIDE
- 17.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 17.3 CUSTOMIZATION OPTIONS
- 17.4 RELATED REPORTS
- 17.5 AUTHOR DETAILS

