
|
시장보고서
상품코드
1920862
인간 마이크로바이옴 시장(제5판) : 업계 동향과 세계 예측(-2035년) - 제품 유형별, 생물학적 제제 유형별, 투여 경로별, 약제 제형별, 대상 적응증별, 대상 치료 영역별 및 지역별Human Microbiome Market (5th Edition): Industry Trends and Global Forecasts, till 2035 - Distribution by Type of Product, Type of Biologic, Route of Administration, Drug Formulation, Target Indication, Target Therapeutic Area and Geographical Regions |
||||||
인간 마이크로바이옴 시장 : 개요
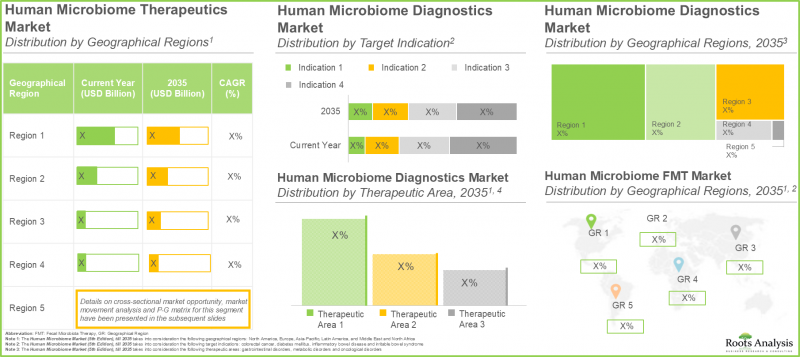
Roots Analysis의 조사에 따르면, 인간 마이크로바이옴 시장 규모는 현재 8억 5,000만 달러에서 2035년까지 32억 7,000만 달러로 성장할 것으로 예측됩니다. 예측 기간(2035년까지)의 CAGR은 16%로 예상됩니다.
인간 마이크로바이옴 시장 : 성장과 트렌드
인체 마이크로바이옴은 박테리아, 고세균, 바이러스, 곰팡이 등 다양한 미생물군으로 구성되어 있으며, 피부, 장, 구강, 기타 조직 등 인체의 다양한 부위에 서식하고 있습니다. 인간 마이크로바이옴의 다양성에 대한 연구는 Antonie van Leeuwenhoek이 구강 및 분변 미생물군집 샘플을 관찰한 1680년대까지 거슬러 올라갑니다. 현재 인간 마이크로바이옴의 상황은 매우 역동적이며, 면역, 대사, 심지어 암, 염증성 질환, 신경 퇴행성 질환 등의 질환에서 그 중요한 기능이 연구로 강조되고 있습니다. 미국 식품의약국(FDA)은 재발성 클로스트리듐 디피실 감염증(rCDI) 예방을 목적으로 하는 두 가지 분변 미생물총 치료제를 승인했습니다. 특히, Ferring Pharmaceutical이 개발한 REBYOTA는 2022년 11월 승인을 받은 최초의 인간 마이크로바이옴 치료제가 되었고, Seres Therapeutics가 개발한 VOWST도 2023년 4월 경구투여 승인을 받았습니다. 주목할 만한 점은 Nestle Health Science가 2024년 6월 VOWST의 미국 및 전 세계 판권을 획득함으로써 상업적 성공이 더욱 강화되었습니다는 점입니다.
DNA 시퀀싱, 바이오인포매틱스, 분석 도구의 지속적인 발전으로 건강 및 질병과 관련된 인간 마이크로바이옴에 대한 이해가 깊어짐에 따라 이 분야는 가까운 미래에 두 자릿수 시장 성장이 예상됩니다.
성장 촉진요인 : 시장 확대의 전략적 촉진요인
차세대 염기서열 분석, 메타유전체 등 마이크로바이옴 분석 기술의 발전으로 미생물 군집에 대한 보다 심층적인 분석이 가능해져 진단과 치료가 가속화되고 있습니다. 또한, 제약사, 바이오기업, 벤처캐피탈의 투자 증가로 위장장애, 대사질환, 암 등의 질환에 대한 임상 파이프라인의 확장을 지원하고 있습니다. 또한, 건강에서 마이크로바이옴에 대한 인식 증가, 개인 맞춤형 프로바이오틱스에 대한 수요, 예방의학, 스타트업과 학계와의 협력이 결합하여 인간 마이크로바이옴 시장의 확대를 더욱 촉진하고 있습니다.
시장 과제: 발전을 가로막는 심각한 장벽들
세계 휴먼 마이크로바이옴 시장은 괄목할 만한 성장세를 보이고 있지만, 이 산업의 발전을 저해할 수 있는 특정 제약이 존재합니다. 생체 미생물을 위한 GMP 시설과 복잡한 임상시험을 포함한 인간 마이크로바이옴 시장 분야의 높은 연구개발비용과 상업화 비용은 확장성과 보급을 가로막고 있습니다. 엄격한 규제 요건, 표준화된 가이드라인의 부재, 미생물 균주의 다양성, 승인 지연, 지역 간 진입장벽이 높아졌습니다. 또한, 일반인의 이해 부족, 마이크로바이옴 조작에 대한 윤리적 우려, 일관성 없는 증거로 인한 환자 및 의료 서비스 제공업체의 도입 지연도 진전을 가로막고 있습니다.
인간 마이크로바이옴 시장 : 주요 연구 결과
이 보고서는 인간 마이크로바이옴 시장의 현황을 분석하고 업계 내 잠재적인 성장 기회를 파악합니다. 주요 조사 결과는 다음과 같습니다.
- 현재 약 55개 개발사가 개발한 215종에 가까운 인간 마이크로바이옴 치료제가 다양한 개발단계에서 승인되었거나 연구 중에 있으며, 다양한 질환의 치료를 목적으로 하고 있습니다.
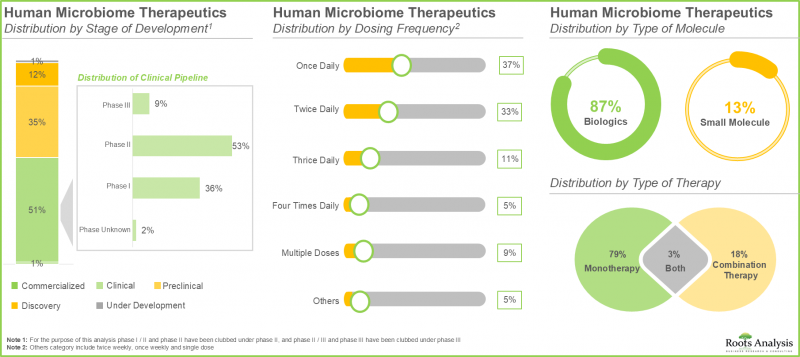
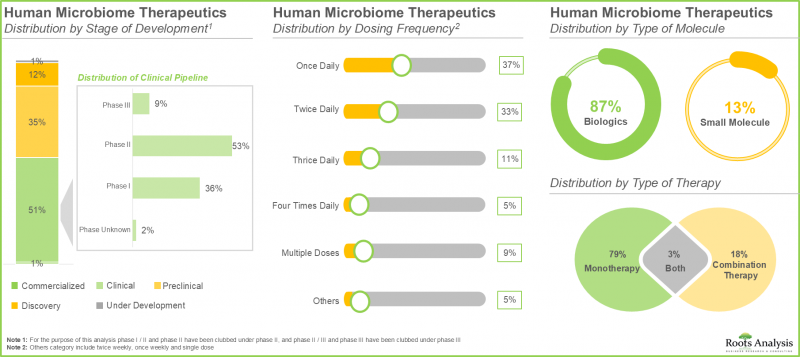
- 인간 마이크로바이옴 치료제의 50% 이상이 임상 개발 단계에 있습니다. 특히, 대부분의 약물/치료 프로그램은 생물학적 제제, 주로 살아있는 생물학적 제제 개발에 초점을 맞추었습니다.
- 이 분야는 75개 기업이 약 140여 종의 인체 마이크로바이옴 진단 검사를 개발 중이며, 주목할 만한 점은 개발 기업의 약 55%가 2015년 이후 설립되었습니다는 점입니다.
- 약 80%의 기업이 미생물총의 구성과 기능 분석을 목적으로 하는 인체 마이크로바이옴 진단을 제공하고 있으며, 55%의 기업이 시퀀싱/메타유전체 기술을 진단에 활용하고 있습니다.
- 대부분의 분변 미생물총 치료제 개발 기업들은 2000년부터 2015년 사이에 설립되었습니다. 이 중 65% 이상이 북미에 본사를 두고 있으며, 유럽(약 35%)이 그 뒤를 잇고 있습니다.
- 분변 세균 요법은 건강한 개인으로부터 채취한 분변 세균을 환자의 위, 대장/소장에 이식/주입하는 것입니다. 이들 치료법의 대부분(50%)은 임상 단계에 있습니다.
- 현재까지 다양한 분변 미생물총 요법의 안전성과 유효성을 평가하기 위한 임상시험이 205건 가까이 등록되어 있습니다. 이러한 연구의 대부분은 유럽의 다양한 시험 시설에서 수행되고 있습니다.
- 최근 몇 년 동안 스타트업들은 마이크로바이옴 치료 및 진단 포트폴리오를 강화하기 위해 여러 가지 노력을 기울이고 있습니다. 그 중 대부분은 궤양성 대장염 치료를 위한 것이었습니다.
- 이러한 시장에 대한 관심은 최근 다양한 이해관계자들 간에 맺어진 다양한 제휴 관계에도 반영되어 있습니다. 실제로 지난 2년간 45% 이상의 거래가 체결되었습니다.
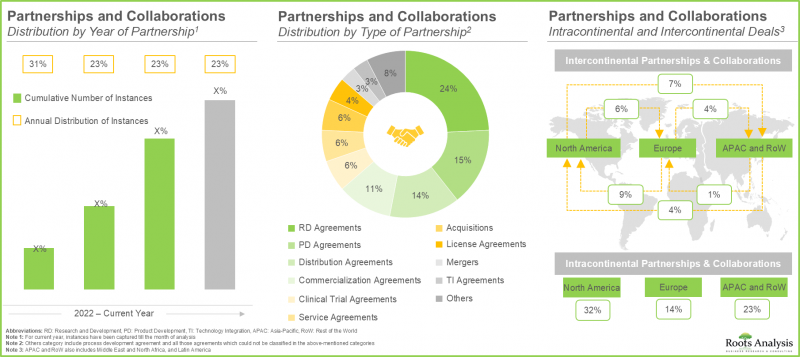
- 2022년 이후, 이 분야는 12억 달러 이상의 자금이 조달되었습니다. 특히, 보조금 및 상금도 마이크로바이옴 의약품 개발자들이 채택하는 매우 두드러진 자금 조달 모델이 되고 있습니다.
- 시장 영향력 분석은 전체 시장의 발전에 영향을 미칠 수 있는 잠재적 요인을 개괄적으로 설명하며, 특정 영역의 촉진요인, 제약요인, 기회, 과제를 파악하는 데 활용할 수 있습니다.
- 장 건강에 대한 소비자의 인식 증가, 만성 질환 증가, 마이크로바이옴 시퀀싱 기술의 발전에 힘입어 시장은 앞으로도 꾸준히 성장할 것으로 예측됩니다.
- 현재 다양한 투여 경로 중 경구 투여 경로는 안전성, 편의성, 구강 내 직접적 치료 개입에 대한 적합성으로 인해 시장에서 가장 높은 점유율(55%)을 차지하고 있습니다.
- 감염성 질환 하위 부문은 인간 마이크로바이옴 치료제가 이러한 질환의 치료에 효과적이기 때문에 올해 전체 시장 점유율을 차지할 것으로 추정됩니다.
- 특히, 여러 지역 중 북미는 예측 기간 동안 전체 인간 마이크로바이옴 시장을 지배할 것으로 예측됩니다.
- 북미에서는 인간 마이크로바이옴 시퀀싱 기술이 빠르게 발전하고 있어, 올해 북미가 전체 인간 마이크로바이옴 시장에서 큰 비중을 차지할 것으로 예측됩니다.
- 미국 내 주요 기업별 개발 및 승인된 마이크로바이옴 치료제의 빠른 성공에 힘입어 인간 마이크로바이옴 치료제 시장은 안정적인 CAGR로 성장할 것으로 예측됩니다.
인간 마이크로바이옴 시장
시장 규모 및 기회 분석은 다음 매개 변수를 기반으로 세분화됩니다.
제품 유형별
- 치료제
- 진단약
- 분변 미생물총 요법
생물학적 제제 유형별
- 생물학적 제제
- 기타
투여 경로별
- 경구 투여
- 직장 투여
약제 제형별
- 캡슐
- 서스펜션
- 관장기
- 분말
대상 적응증별
- 클로스트리디움 디피실 감염
- 괴사성 장염
- 과민성 대장 증후군
- 급성 이식편 대 숙주병
- 당뇨병
- 과민성 대장 증후군
- 대장암
치료 대상 부위별
- 감염성 질환
- 소화기 질환
- 희귀 질환
- 대사성 질환
- 종양학 영역
지역별
- 북미
- 미국
- 캐나다
- 유럽
- 독일
- 프랑스
- 이탈리아
- 영국
- 스페인
- 기타 유럽 국가
- 아시아태평양
- 호주
- 중국
- 인도
- 일본
- 라틴아메리카
- 브라질
- 중동 및 북아프리카
- 이스라엘
- 사우디아라비아
인간 마이크로바이옴 시장 : 주요 부문
제품 유형별 시장 점유율: 진단 부문이 가장 큰 점유율을 차지합니다.
올해 인간 마이크로바이옴 시장 보고서에 따르면, 대사 장애, 암 관련 질환 등 다양한 질환의 조기 발견을 위한 마이크로바이옴 중심의 진단 검사 도입 증가로 진단 부문이 가장 큰 시장 점유율(약 45%)을 차지하고 있습니다. 치료제 부문은 예측 기간 동안 32%의 높은 성장률을 보일 것으로 예측됩니다. 이는 개인 맞춤형 치료 연구개발 증가, 특정 질환에 대한 분변 미생물 이식(FMT)과 같은 기존 치료법의 효과, 그리고 개발 중인 신약 및 신치료법의 효과에 기인합니다.
인간 마이크로바이옴 기반 치료제 시장 점유율에 대한 통찰력 제공
생물학적 제제 유형별 시장 점유율: 생물학적 제제 부문이 가장 큰 점유율을 차지함.
현재 생물학적 제제 부문은 소화기 질환, 대사 장애, 암 관련 질환 등 다양한 질환의 치료에 마이크로바이옴 표적 치료제의 적용 확대로 인해 전체 인체 마이크로바이옴 시장(치료제 분야)의 점유율을 차지하고 있습니다. 특히, 기타 부문(파지 생물학적 제제, 백신, 기타 항체 등)은 예측 기간 동안 64%의 연평균 복합 성장률(CAGR)을 보일 것으로 예측됩니다.
투여 경로별 시장 점유율: 경구 투여 부문이 가장 큰 매출을 차지
올해 경구 투여 경로는 인간 마이크로바이옴 치료제 시장에서 가장 큰 시장 점유율(55%)을 차지하고 있습니다. 주목할 만한 점은 경구 투여 경로를 통한 치료법이 예측 기간 동안 38%의 높은 CAGR을 기록할 가능성이 높다는 점입니다. 이는 안전성, 편의성, 그리고 구강 내 직접적인 치료적 상호작용을 통해 전신에 대한 효과를 달성할 수 있는 능력과 관련이 있습니다.
제형별 시장 점유율: 서스펜션 부문이 더 높은 CAGR로 성장
다양한 부문 중 캡슐 부문은 2025년 인간 마이크로바이옴 치료제 시장에서 가장 큰 점유율(55%)을 차지할 것으로 예측됩니다. 이는 캡슐 형태의 마이크로바이옴 중심 치료제의 강력한 임상시험 파이프라인에 기인합니다. 특히 서스펜션 부문 시장은 예측 기간 동안 상당히 높은 CAGR(38%)로 확대될 것으로 예측됩니다. 이는 살아있는 미생물 제제의 생존성과 안정성을 유지하는 능력, 용량 조절을 용이하게 하는 특성, 그리고 다양한 치료 용도에 대한 다년간의 적용 실적에 기인합니다.
적응증별 시장 점유율: 클로스트리듐 디피실 감염증 부문이 주도하고 있음
현재 클로스트리듐 디피실 감염 질환 부문이 전체 인체 마이크로바이옴 치료제 시장 규모를 차지하고 있습니다. 이는 클로스트리디움 디피실 감염증의 발생 빈도와 중증도가 증가하고 있으며, 항생제에 의한 장내 세균총 교란과 본 감염증의 연관성이 입증되었기 때문입니다.
또한, 급성 이식편대숙주질환 치료제 시장은 2027년부터 2035년까지 비교적 높은 CAGR(35%)로 성장할 것으로 예측됩니다. 이는 소화기 관련 급성 이식편대숙주질환(GVHD) 환자의 미충족 수요를 충족시키기 위해 설계된 선구적인 미생물총 중심 치료제인 MaaT013(Xervyteg(R))의 승인에 대한 기대감이 반영된 것입니다. 이 질환은 기존 생존율이 위험할 정도로 낮고, 승인된 대체 치료법이 존재하지 않습니다.
치료 영역별 시장 점유율: 감염성 질환 부문이 시장을 독식하고 있음
현재 감염질환 분야가 전체 인체 마이크로바이옴 치료제 시장 규모를 차지하고 있으며(점유율 100%), 이러한 추세는 앞으로도 변하지 않을 것으로 보입니다. 이는 면역 기능 및 병원체 방어에 직접적으로 관여할 뿐만 아니라, 재발성 클로스트리디움 디피실 감염 등에 대한 마이크로바이옴 기반 치료의 효과가 입증되었기 때문입니다. 특히 희귀질환 분야는 2027년부터 2035년까지 비교적 높은 CAGR(35%)로 성장할 것으로 전망됩니다.
지역별 시장 점유율: 북미가 가장 큰 매출을 차지합니다.
당사의 예측에 따르면, 북미는 2025년 인간 마이크로바이옴 치료제 시장 점유율의 대부분(95%)을 차지할 것으로 예측됩니다. 이는 북미의 선진화된 의료 인프라를 통해 개발자들이 다수의 임상시험을 수행할 수 있고, 승인기관의 규제 가이드라인을 충족할 수 있기 때문입니다. 주목할 만한 점은 유럽 시장이 2025년부터 2032년까지 비교적 높은 CAGR(75%)로 성장할 것으로 예상된다는 점입니다.
인간 마이크로바이옴 시장의 대표적인 진출기업들
- Becton, Dickinson and Company
- Biome Diagnostics
- Ferring Pharmaceuticals
- GoodGut
- Infant Bacterial Therapeutics
- MaaT Pharma
- Microbiomik
- NutriPATH
- Seres Therapeutics
- Tiny Health
- OxThera
- Vedanta Bisociences
- Zybio
인간 마이크로바이옴 시장 : 조사 범위
- 시장 규모 및 기회 분석 : 본 보고서는 인간 마이크로바이옴 시장에 대해(A) 제품 유형,(B) 생물학적 제제 유형,(C) 투여 경로,(D) 제제,(E) 적응증,(F) 치료 영역,(G) 지리적 지역 등 주요 시장 부문에 초점을 맞춘 상세한 분석을 수록했습니다. 분석이 수록되어 있습니다.
- 인간 마이크로바이옴 치료제 - 시장 현황: 인간 마이크로바이옴 치료제의 현재 시장 현황에 대한 상세한 개요와 함께(A) 개발 단계,(B) 분자 유형,(C) 생물학적 제제 유형,(D) 대상 적응증,(E) 치료 영역,(F) 투여 경로,(G) 제제 유형,(H) 투여 빈도,(I) 투여 빈도,(I). 치료법 유형,(J)설립연도,(K)기업 규모,(L)본사 소재지,(M)가장 활발한 기업 관련 정보 포함.
- 인체 마이크로바이옴 진단 및 스크리닝/프로파일링 검사 - 시장 현황 2: 각종 진단 및 스크리닝/프로파일링 검사의 현재 시장 현황에 대한 상세한 개요와 관련 파라미터에 대한 정보 제공:(A) 개발 단계,(B) 분석 대상 검체 유형,(C) 스크리닝 기술 유형,(D) 대상 적응증,(E) 치료 영역,(F) 검사 목적,(G) 설립 연도 C) 스크리닝 기술 유형,(D) 대상 적응증,(E) 치료 영역,(F) 검사 목적,(G) 설립 연도,(H) 기업 규모,(I) 본사 소재지 등 관련 정보를 포함합니다.
- 인간 마이크로바이옴 분변 미생물총 치료(FMT) - 시장 상황 3: FMT 치료의 다른 관련 측면에 대한 현재 시장 상황에 대한 자세한 개요와 다음과 같은 관련 파라미터에 대한 정보 제공:(A) 개발 단계,(B) 분석 대상 샘플 유형,(C) 스크리닝 기술 유형,(D) 대상 적응증,(E) 치료 영역,(F) 검사 목적,(G) 설립 연도,(H) 기업 규모,(I) 본사 소재지.
- 기업 및 약물 프로파일:(A)설립연도,(B)본사 소재지,(C)미생물총 기반 약물 포트폴리오,(D)최근 동향,(E)향후 전망에 따라 미생물총 치료제, 미생물총 진단/스크리닝/프로파일링 검사 및 분변 미생물총 치료제 개발에 종사하는 주요 기업의 주요 기업의 상세한 프로파일.
- 임상시험 분석 : 대변 미생물총 치료 관련 완료 및 진행 중인 임상시험,(A) 임상시험 등록 연도,(B) 등록 환자 수,(C) 환자 성별,(D) 임상시험 단계,(E) 임상시험 현황,(F) 치료 영역,(G) 스폰서/협력기관 유형,(H) 연구 설계,(I) 가장 활발한 기업(임상),(J) 지역 시험 수 기준),(J) 지역.
- 매력도 및 경쟁력 분석 : 9박스 AC 매트릭스 프레임워크를 기반으로 조사 대상의 다양한 치료 영역의 주요 적응증에 대한 통찰력 있는 사업 포트폴리오 분석을 수행합니다. 또한, 가장 흔한 질환 적응증에 대한 상대적 시장 매력과 기존 경쟁 구도에 대한 고찰을 포함합니다.
- 제휴/협력 관계: 인간 마이크로바이옴 개발 기업 간 체결된 제휴/협력 관계를(A) 제휴 연도,(B) 제휴 형태,(C) 대상 적응증,(D) 치료 영역,(E) 파트너 유형,(F) 가장 활발한 기업(제휴 건수 기준) 등 관련 파라미터를 기준으로 상세하게 분석합니다.
- 자금조달 및 투자: 이 분야의 자금조달 및 투자에 대한 상세한 분석. A) 자금 조달 연도(B) 자금 조달 유형(C) 투자 금액(D) 자금 조달 목적(E) 대상 적응증(F) 치료 분야(G) 지역(H) 가장 활발한 기업(I) 주요 투자자
- 사례 연구 1: 마이크로바이옴 치료제 개발 및 제조에 관련된 각 공정에 대한 자세한 고찰과 함께 위탁생산기업(CMO) 참고 목록을 수록. 각 기업의 설립연도, 본사 소재지, 기업규모, 사업 규모, 생산제품의 유형, 제형 유형 등 상세 정보도 함께 기재. 또한 CMO/CRO 파트너 선정 시 고려해야 할 주요 사항을 중점적으로 설명합니다.
- 사례 연구 2: 빅데이터의 새로운 역할을 평가하고, 마이크로바이옴 연구에서 생성된 데이터를 분석하기 위한 다양한 알고리즘/도구 개발 및 구현에 초점을 맞춘 노력을 강조하며, 지난 10년간 이해관계자들의 마이크로바이옴 연구 지원을 위한 빅데이터 도구 활용에 대한 관심 증가를 보여주는 통찰력 있는 구글 트렌드 분석을 제시합니다.
목차
제1장 서문
제2장 조사 방법
제3장 시장 역학
- 본 장의 개요
- 예측 조사 방법
- 시장 평가 프레임워크
- 예측 툴과 테크닉
- 중요 고려사항
- 제한 사항
제4장 거시경제 지표
제5장 주요 요약
제6장 서론
- 본 장의 개요
- 미생물총과 마이크로바이옴 개념
- 장내 후로라 개요
- 마이크로바이옴과 관련 질환
- 약물 동태에 대한 미생물총 영향
- 미생물총이 치료 결과에 미치는 영향
- 마이크로바이옴 치료
- 인간 마이크로바이옴프로제크트(HMP)
- 생생물제제(LBP)에 관한 규제 가이드라인
- 마이크로바이옴 치료제 개발 주요 과제
- 향후 전망
제7장 인간 마이크로바이옴 치료제 : 시장 구도
- 본 장의 개요
- 인간 마이크로바이옴 치료제 : 시장 구도
- 인간 마이크로바이옴 치료제 : 개발 상황
제8장 인간 마이크로바이옴 진단 및 스크리닝/프로파일링 검사 : 시장 구도
- 본 장의 개요
- 마이크로바이옴 진단과 스크리닝/프로파일링 검사 개요
- 마이크로바이옴 진단 및 스크리닝/프로파일링 검사 : 시장 구도
- 마이크로바이옴 진단 및 스크리닝/프로파일링 검사 : 프로바이더 상황
제9장 인간 마이크로바이옴 분변 마이크로바이옴 요법 : 시장 구도
- 본 장의 개요
- 분변 미생물 요법(FMT) 입문
- 역사적 개요
- 변중 미생물 요법 : 절차와 임상적 의의
- 변중 미생물 요법에 관한 규제 가이드라인
- 변중 미생물 요법 보험 적용
- 분변 미생물 요법 : 시장 구도
제10장 인간 마이크로바이옴 치료제 : 기업과 약제 개요
- 본 장의 개요
- Ferring Pharmaceuticals
- Infant Bacterial Therapeutics
- MaaT Pharma
- Mikrobiomik
- Seres Therapeutics
- OxThera
- Vedanta Biosciences
- Zhiyi Biotech
제11장 마이크로바이옴 진단 및 스크리닝/프로파일링 검사 프로바이더 : 기업 개요
- 본 장의 개요
- Becton, Dickinson and Company
- Biome Diagnostics
- GoodGut
- NutriPATH
- Tiny Health
제12장 임상시험 분석
- 본 장의 개요
- 범위와 조사 방법
- 변중 미생물 요법 : 임상시험 분석
제13장 매력과 경쟁력(AC) 매트릭스
제14장 스타트업 건전성 지표
- 본 장의 개요
- 범위와 조사 방법
- 스타트업 벤치마킹
제15장 사례 연구 : 주요 치료 영역
- 본 장의 개요
- 대사장애
- 소화기계 및 위장계 질환
- 종양 학적 적응증
- 피부 질환
- 감염증
제16장 파트너십 및 협업
제17장 자금조달과 투자
제18장 사례 연구 : 마이크로바이옴 치료제 및 생체 바이오 치료제 계약 서비스
제19장 사례 연구 : 빅데이터와 마이크로바이옴 치료
제20장 인간 마이크로바이옴 치료제 시장
- 본 장의 개요
- 전제와 조사 방법
- 세계의 인간 마이크로바이옴 치료제 시장 : 예측 추정치(2035년까지)
- 인간 마이크로바이옴 시장 : 제품 유형별 분포
- 주요 시장 세분화
제21장 인간 마이크로바이옴 치료제 시장(생물학적 제제 유형별)
제22장 인간 마이크로바이옴 치료제 시장(투여 경로별)
제23장 인간 마이크로바이옴 치료제 시장(약제 제형별)
제24장 인간 마이크로바이옴 치료제 시장(대상 적응증별)
제25장 인간 마이크로바이옴 치료제 시장(대상 치료 영역별)
제26장 인간 마이크로바이옴 치료제 시장(지역별)
제27장 인간 마이크로바이옴 진단 시장
제28장 인간 마이크로바이옴 진단 시장(대상 적응증별)
제29장 인간 마이크로바이옴 진단 시장(대상 치료 영역별)
제30장 인간 마이크로바이옴 진단 시장(지역별)
제31장 인간 마이크로바이옴 분변 마이크로바이오타 치료 시장
제32장 인간 마이크로바이옴 분변 마이크로바이오타 치료 시장(지역별)
제33장 인간 마이크로바이옴 치료제 시장 기회 분석 : 북미
제34장 인간 마이크로바이옴 치료제 시장 기회 분석 : 유럽
제35장 인간 마이크로바이옴 치료제 시장 기회 분석 : 아시아태평양
제36장 인간 마이크로바이옴 치료제 시장 기회 분석 : 라틴아메리카
제37장 인간 마이크로바이옴 치료제 시장 기회 분석 : 중동 및 북아프리카
제38장 인간 마이크로바이옴 진단 시장 기회 분석 : 북미
제39장 인간 마이크로바이옴 진단 시장 기회 분석 : 유럽
제40장 인간 마이크로바이옴 진단 시장 기회 분석 : 아시아태평양
제41장 인간 마이크로바이옴 진단 시장 기회 분석 : 라틴아메리카
제42장 인간 마이크로바이옴 진단 시장 기회 분석 : 중동 및 북아프리카
제43장 결론
제44장 경영진 인사이트
제45장 부록 I : 표 데이터
제46장 부록 II : 기업 및 조직 리스트
LSH 26.02.19Human Microbiome Market: Overview
As per Roots Analysis, the human microbiome market is estimated to grow from USD 0.85 billion in the current year to USD 3.27 billion by 2035, at a CAGR of 16% during the forecast period, till 2035.
Human Microbiome Market: Growth and Trends
The human microbiome consists of a variety of microorganisms, such as bacteria, archaea, viruses, and fungi, that inhabit different regions of the human body, including the skin, gut, mouth, and other tissues. The investigation into the diversity of the human microbiome dates back to the 1680s, when Antonie van Leeuwenhoek examined his oral and fecal microbiota samples. The existing human microbiome landscape is extremely dynamic, with studies emphasizing its crucial function in immunity, metabolism, and conditions like cancer, inflammatory diseases, and even neurodegenerative disorders. The US FDA has authorized two fecal microbiota treatments to prevent recurring C. difficile infection (rCDI). Significantly, REBYOTA (created by Ferring Pharmaceutical) became the first human microbiome therapy to gain approval in November 2022, with VOWST (developed by Seres Therapeutics) also receiving approval for oral administration in April 2023. Notably, the commercial triumph of VOWST has been strengthened by Nestle Health Science's purchase of its US and worldwide rights in June 2024.
Given the continuous advancements in DNA sequencing, bioinformatics, and analytical tools that enhance our comprehension of the human microbiome in relation to health and diseases, we anticipate that this sector is poised for double-digit market growth in the near future.
Growth Drivers: Strategic Enablers of Market Expansion
Advancements in microbiome sequencing technologies, such as next-generation sequencing and metagenomics, enable deeper analysis of microbial communities, accelerating diagnostics and therapies. Further, rising investments from pharma, biotech firms, and venture capital supports expanding clinical pipelines for conditions like gastrointestinal disorders, metabolic diseases, and cancer. In addition, increasing awareness of microbiomein health, coupled with demand for personalized probiotics, preventive healthcare, and collaborations between startups and academia, further propels human microbiome market expansion.
Market Challenges: Critical Barriers Impeding Progress
While the global human microbiome market is experiencing substantial growth, there are certain restraints that can hinder the growth of this industry. High R&D and commercialization costs in the human microbiome market space including GMP facilities for live organisms and complex clinical trials, hinder scalability and adoption Stringent regulatory requirements, lack of standardized guidelines, and variability in microbial strains delay approvals and increase entry barriers across regions. Limited public understanding, ethical concerns over microbiome manipulation, and slow patient / provider adoption due to inconsistent evidence also impede progress.
Human Microbiome Market: Key Insights
The report delves into the current state of the human microbiome market and identifies potential growth opportunities within industry. Some key findings from the report include:
- Currently, close to 215 human microbiome therapeutics developed by ~55 developers are approved / being investigated across various stages of development for treating a myriad of disorders.
- Over 50% of the human microbiome therapeutics are in clinical stages of development; notably, most of the drugs / therapy programs are focused on the development of biologics, primarily live biotherapeutics.
- Close to 140 human microbiome diagnostic tests are being developed by 75 players in this domain; notably, around 55% of the developers were established post-2015.
- Around 80% of the companies are offering human microbiome diagnostics for analysis of microfloral composition and function; 55% of players are employing sequencing / metagenomics techniques for the diagnosis.
- Majority of fecal microbiota therapy developers were established between 2000-2015; of these, more than 65% are headquartered in North America, followed by Europe (~35%).
- Fecal bacteriotherapy involves the transplantation / infusion of fecal bacteria from healthy individuals to the stomach, colon / small intestine of patients; majority (50%) of these therapies are in clinical stages.
- Close to 205 clinical trials have been registered till date to evaluate the safety and efficacy of various fecal microbiota therapies; majority of these studies have been conducted across various trial sites in Europe.
- Over the last few years, start-ups have undertaken multiple initiatives to enhance their microbiome therapeutics and diagnostics portfolio; majority of the initiatives were undertaken for the treatment of ulcerative colitis.
- The rising interest in this market is reflected from the diverse partnerships established among various stakeholders in the recent past; in fact, more than 45% of the deals were inked in the last two years.
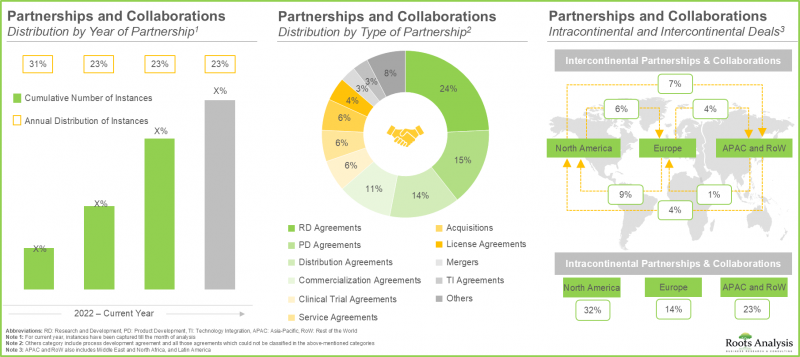
- Since 2022, more than USD 1.2 billion has been raised in this domain; notably, grants / awards are also a very prominent funding model adopted by microbiome drug developers.
- The market impact analysis outlines the potential factors that influence the evolution of the overall market; it can be employed to determine the growth drivers, restraints, opportunities and challenges within a specific domain.
- Driven by the increasing consumer awareness gut health, the rising prevalence of chronic diseases and advancements in microbiome sequencing technologies, the market is poised for a steady growth in the future.
- Currently, amongst various routes of administration, oral route captures the highest share (55%) of the market owing to its safety, convenience and the suitability for direct therapeutic intervention within the oral cavity.
- The infectious diseases sub-segment is estimated to capture of the overall market share in the current year, owing to the effectiveness of human microbiome therapeutics in the treatment of such diseases.
- Notably, amongst various geographical regions, North America is expected to dominate the overall human microbiome market during the forecast period.
- North America is expected to capture a significant share of the overall human microbiome market in the current year owing to the rapid innovations within human microbiome sequencing technologies in the region.
- Driven by the rapid success of approved microbiome therapeutic drugs developed by prominent players in the US, the human microbiome therapeutics market is expected to grow at a steady CAGR.
Human Microbiome Market
The market sizing and opportunity analysis has been segmented across the following parameters:
By Type of Product
- Therapeutics
- Diagnostics
- Fecal Microbiota Therapies
By Type of Biologic
- Live Biotherapeutics
- Others
By Route of Administration
- Oral Route
- Rectal Route
By Drug Formulation
- Capsule
- Suspension
- Enema
- Powder
By Target Indication
- Clostridium Difficile Infection
- Necrotizing Enterocolitis
- Irritable Bowel Syndrome
- Acute Graft Versus Host Disease
- Diabetes Mellitus
- Irritable Bowel Disease
- Colorectal Cancer
By Target Therapeutic Area
- Infectious Disorders
- Gastrointestinal Disorders
- Rare Disorders
- Metabolic Disorders
- Oncological Disorders
By Geographical Regions
- North America
- US
- Canada
- Europe
- Germany
- France
- Italy
- UK
- Spain
- Rest of the Europe
- Asia-Pacific
- Australia
- China
- India
- Japan
- Latin America
- Brazil
- Middle East and North Africa
- Israel
- Saudi Arabia
Human Microbiome Market: Key Segments
Market Share by Type of Product: Diagnostics Segment Hold the Largest Share
In the current year, the diagnostics segment occupies the highest market share (~45%), as per human microbiome market report, driven by increased adoption of microbiome-centered diagnostic tests for the early identification of various conditions, such as metabolic disorders and cancer-related problems. The therapeutics segment is projected to see a significant growth rate of 32% during the forecast period. This increased research and development in personalized therapies, the effectiveness of existing treatments like Fecal Microbiota Transplantation (FMT) for particular illnesses, and new drugs and therapies under development.
Human Microbiome-based Therapeutics Market Share Insights
Market Share by Type of Biologic: Live Biotherapeutics Segment Holds the Largest Market Share
Currently, the live biotherapeutics segment occupies the entire human microbiome market share (for therapeutics) owing to the increasing application of microbiome-targeted treatments for treating various conditions, such as digestive problems, metabolic disorders, and cancer-related issues. Notably, the others segment (which includes phage biologics, vaccines, and additional antibodies) is expected to experience a remarkable growth rate, with a CAGR of 64% during the forecast period.
Market Share by Route of Administration: Oral Route Segment Holds the Largest Revenue
In the current year, the oral route occupies the highest market share (55%) of human microbiome therapeutics market. It is worth noting that the therapies administered via oral route are likely to witness a substantial CAGR of 38% during the forecast period. This is associated to the safety, user-friendliness, and the ability for direct therapeutic interaction in the oral cavity to accomplish desired systemic effects.
Market Share by Drug Formulation: Suspension Segment to Grow at a Higher CAGR
Among the various segments, the capsule segment occupies the highest human microbiome therapeutics market share (55%) in 2025. This results from the strong clinical trial pipeline of microbiome-centered therapies in capsule form. It is important to highlight that the suspension segment market is expected to expand at a notably higher CAGR (38%) throughout the forecast period. This arises from its ability to maintain the viability and stability of living microbial formulations, facilitate straightforward dosage adjustments, and its long-standing application history in diverse therapeutic uses.
Market Share by Target Indication: Clostridium Difficile Infection Segment Domina
Currently, the clostridium difficile infection segment occupies the entire human microbiome therapeutics market size. This is due to the rising frequency and intensity of clostridium difficile infections, coupled with the established link between antibiotic-induced gut dysbiosis and the infection.
Further, the market for therapies targeting acute graft versus host disease is likely to grow at a relatively higher CAGR (35%), during the period 2027-2035. This is attributed to the expected endorsement of MaaT013 (Xervyteg(R)), a pioneering microbiota-centered therapy designed to meet a significant unmet need in patients suffering from GI-involved acute graft versus host disease, where existing survival rates are dangerously low and no alternative approved therapy exists.
Market Share by Target Therapeutic Area: Infectious Disorders Segment Dominates the Market
Currently, the infectious diseases segment occupies the entire human microbiome therapeutics market size (with a share of 100%) and this trend is unlikely to change in the future. This is attributed to its direct involvement in immune function and pathogen defense, along with the demonstrated effectiveness of microbiome-based treatments, such as those addressing recurrent Clostridium difficile infections. Notably, the rare disorders segment is expected to experience a comparatively higher CAGR (35%) from 2027 to 2035.
Market Share by Geographical Regions: North America with Highest Revenue
According to our projections, North America is likely to capture the majority (95%) of the human microbiome therapeutics market share in 2025, as the advanced healthcare infrastructure in North America enables developers to conduct a large number of clinical trials, in order to meet the regulatory guidelines of the approval bodies. It is worth highlighting that the market in Europe is likely to grow at a relatively higher CAGR (75%), during the period 2025-2032.
Primary Research Overview
The opinions and insights presented in the market report were also influenced by discussions held with senior stakeholders in the industry. The market report includes detailed transcripts of interviews conducted with the following individuals:
- Former Senior Manager, Corporate Development, Mid-Sized Company, UK
- Co-founder and Chairman, Small Company, US
- Chief Executive Officer, Small Company, US
- Senior Scientist, Large Company, US
- President, Small Company, Taiwan
- Vice President, Business Development, Small Company, US
- Chief Business Officer, Mid-sized Company, Israel
- Former Vice President, Sales and Business Development, Small Company, US
- Co-founder and Chief Executive Officer, Small Company, Belgium
- Co-founder and Chief Executive Officer, Small Company, US
- Co-founder and Chief Executive Officer, Small Company, US
- Ex-Co-founder and Vice President of Innovation, Mid-sized Company, US
- Vice President & Chief Operating Officer, Small Company, US
- Former Vice President, Operations, Mid-sized Company, US
- Former President and Chief Executive Officer, Small Company, US
- Former Chief Scientific Officer and Vice President, Research, Small Company, US
- Former Chief Strategy Officer, Small Company, France
Example Players in Human Microbiome Market
- Becton, Dickinson and Company
- Biome Diagnostics
- Ferring Pharmaceuticals
- GoodGut
- Infant Bacterial Therapeutics
- MaaT Pharma
- Microbiomik
- NutriPATH
- Seres Therapeutics
- Tiny Health
- OxThera
- Vedanta Bisociences
- Zybio
Human Microbiome Market: Research Coverage
- Market Sizing and Opportunity Analysis: The report features an in-depth analysis of the human microbiome market, focusing on key market segments, including [A] type of product, [B] type of biologic, [C] route of administration, [D] drug formulation, [E] target indication, [F] target therapeutic area, and [G] geographical regions.
- Human Microbiome Therapeutics - Market Landscape: A detailed overview of the current market landscape of human microbiome therapeutics, along with information on several relevant parameters, such as [A] stage of development, [B] type of molecule, [C] type of biologic, [D] target indication, [E] therapeutic area, [F] route of administration, [G] type of formulation, [H] dosage frequency, [I] type of therapy, [J] year of establishment, [K] company size, [L] location of headquarters, and [M] most active players.
- Human Microbiome Diagnostics And Screening / Profiling Tests - Market Landscape 2: A detailed overview of the current market landscape of various types of diagnostic and screening / profiling tests, along with information on several relevant parameters, such as [A] stage of development, [B] type of sample analyzed, [C] type of screening technique, [D] target indication, [E] therapeutic area, [F] purpose of test, [G] year of establishment, [H] company size, and [I] location of headquarters.
- Human Microbiome Fecal Microbiota Therapies - Market Landscape 3: A detailed overview of the current market landscape of other relevant aspects of FMT therapies, along with information on several relevant parameters, such as [A] stage of development, [B] type of sample analyzed, [C] type of screening technique, [D] target indication, [E] therapeutic area, [F] purpose of test, [G] year of establishment, [H] company size, and [I] location of headquarters.
- Company and Drug Profiles: In-depth profiles of key players engaged in the development of microbiome therapeutics, microbiome diagnostics and screening / profiling tests and fecal microbiota therapy based on [A] year of establishment, [B] location of headquarters, [C] microbiome-based drug portfolio, [D] recent developments and [E] an informed future outlook.
- Clinical Trial Analysis: An in-depth analysis of completed and ongoing clinical studies of fecal microbiota therapy, based on various relevant parameters, such as [A] trial registration year, [B] number of patients enrolled, [C] patient gender, [D] trial phase, [E] trial status, [F] therapeutic area, [G] type of sponsor / collaborator, [H] study design, [I] most active players (in terms of number of clinical trials), and [J] geography.
- Attractiveness Competitiveness Analysis: An insightful business portfolio analysis of top indications, across various therapeutic areas under investigation, based on the 9-box AC matrix framework. It further includes a discussion on the relative market attractiveness and existing competition across the most popular disease indications.
- Partnerships and Collaborations: An in-depth analysis of the partnerships and collaborations that have been inked by human microbiome developers, based on several relevant parameters, such as [A] year of partnership, [B] type of partnership, [C] target indication, [D] therapeutic area, [E] type of partner and [F] most active players (in terms of number of partnerships).
- Funding and Investments: A detailed analysis of the funding and investments made in this domain, based on several relevant parameters, such as [A] year of funding, [B] type of funding, [C] amount invested, [D] purpose of funding, [E] target indication, [F] therapeutic area, [G] geography, [H] most active players and [I] leading investors.
- Case Study 1: An elaborate discussion on the various steps involved in the development and manufacturing of microbiome therapeutics, along with an indicative list of contract manufacturers, along with details on year of establishment, location of headquarters, company size, scale of operation, type of product manufactured and type of formulation. In addition, the chapter highlights key consideration for selecting a CMO / CRO partner.
- Case Study 2: assessment of the emerging role of big data, highlighting efforts focused on the development and implementation of various algorithms / tools to analyze data generated from microbiome research along with an insightful google trends analysis to demonstrate the rising interest of stakeholders in using big data tools to support microbiome research over the past decade.
Key Questions Answered in this Report
- Which are the leading companies in human microbiome market?
- Which region dominates the human microbiome market?
- What are the key trends observed in the human microbiome market?
- What factors are likely to influence the evolution of this market?
- What are the primary challenges faced by human microbiome providers?
- What is the current and future market size?
- What is the CAGR of this market?
- How is the current and future market opportunity likely to be distributed across key market segments?
Reasons to Buy this Report
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
- The report can aid businesses in identifying future opportunities in any sector. It also helps in understanding if those opportunities are worth pursuing.
- The report helps in identifying customer demand by understanding the needs, preferences, and behavior of the target audience in order to tailor products or services effectively.
- The report equips new entrants with requisite information regarding a particular market to help them build successful business strategies.
- The report allows for more effective communication with the audience and in building strong business relations.
Additional Benefits
- Complementary PPT Insights Pack
- Complimentary Excel Data Packs for all Analytical Modules in the Report
- 15% Free Content Customization
- Detailed Report Walkthrough Session with Research Team
- Free Updated report if the report is 6-12 months old or older
TABLE OF CONTENTS
1. PREFACE
- 1.1. Introduction
- 1.2. Market Share Insights
- 1.3. Key Market Insights
- 1.4. Report Coverage
- 1.5. Key Questions Answered
- 1.6. Chapter Outlines
2. RESEARCH METHODOLOGY
- 2.1. Chapter Overview
- 2.2. Research Assumptions
- 2.2.1. Market Landscape and Market Trends
- 2.2.2. Market Forecast and Opportunity Analysis
- 2.2.3. Comparative Analysis
- 2.3. Database Building
- 2.3.1. Data Collection
- 2.3.2. Data Validation
- 2.3.3. Data Analysis
- 2.4. Project Methodology
- 2.4.1. Secondary Research
- 2.4.1.1. Annual Reports
- 2.4.1.2. Academic Research Papers
- 2.4.1.3. Company Websites
- 2.4.1.4. Investor Presentations
- 2.4.1.5. Regulatory Filings
- 2.4.1.6. White Papers
- 2.4.1.7. Industry Publications
- 2.4.1.8. Conferences and Seminars
- 2.4.1.9. Government Portals
- 2.4.1.10. Media and Press Releases
- 2.4.1.11. Newsletters
- 2.4.1.12. Industry Databases
- 2.4.1.13. Roots Proprietary Databases
- 2.4.1.14. Paid Databases and Sources
- 2.4.1.15. Social Media Portals
- 2.4.1.16. Other Secondary Sources
- 2.4.2. Primary Research
- 2.4.2.1. Types of Primary Research
- 2.4.2.1.1. Qualitative Research
- 2.4.2.1.2. Quantitative Research
- 2.4.2.1.3. Hybrid Approach
- 2.4.2.2. Advantages of Primary Research
- 2.4.2.3. Techniques for Primary Research
- 2.4.2.3.1. Interviews
- 2.4.2.3.2. Surveys
- 2.4.2.3.3. Focus Groups
- 2.4.2.3.4. Observational Research
- 2.4.2.3.5. Social Media Interactions
- 2.4.2.4. Key Opinion Leaders Considered in Primary Research
- 2.4.2.4.1. Company Executives (CXOs)
- 2.4.2.4.2. Board of Directors
- 2.4.2.4.3. Company Presidents and Vice Presidents
- 2.4.2.4.4. Research and Development Heads
- 2.4.2.4.5. Technical Experts
- 2.4.2.4.6. Subject Matter Experts
- 2.4.2.4.7. Scientists
- 2.4.2.4.8. Doctors and Other Healthcare Providers
- 2.4.2.5. Ethics and Integrity
- 2.4.2.5.1. Research Ethics
- 2.4.2.5.2. Data Integrity
- 2.4.2.1. Types of Primary Research
- 2.4.3. Analytical Tools and Databases
- 2.4.1. Secondary Research
- 2.5. Robust Quality Control
3. MARKET DYNAMICS
- 3.1. Chapter Overview
- 3.2. Forecast Methodology
- 3.2.1. Top-down Approach
- 3.2.2. Bottom-up Approach
- 3.2.3. Hybrid Approach
- 3.3. Market Assessment Framework
- 3.3.1. Total Addressable Market (TAM)
- 3.3.2. Serviceable Addressable Market (SAM)
- 3.3.3. Serviceable Obtainable Market (SOM)
- 3.3.4. Currently Acquired Market (CAM)
- 3.4. Forecasting Tools and Techniques
- 3.4.1. Qualitative Forecasting
- 3.4.2. Correlation
- 3.4.3. Regression
- 3.4.4. Extrapolation
- 3.4.5. Convergence
- 3.4.6. Sensitivity Analysis
- 3.4.7. Scenario Planning
- 3.4.8. Data Visualization
- 3.4.9. Time Series Analysis
- 3.4.10. Forecast Error Analysis
- 3.5. Key Considerations
- 3.5.1. Demographics
- 3.5.2. Government Regulations
- 3.5.3. Reimbursement Scenarios
- 3.5.4. Market Access
- 3.5.5. Supply Chain
- 3.5.6. Industry Consolidation
- 3.5.7. Pandemic / Unforeseen Disruptions Impact
- 3.6. Limitations
4. MACRO-ECONOMIC INDICATORS
- 4.1. Chapter Overview
- 4.2. Market Dynamics
- 4.2.1. Time Period
- 4.2.1.1. Historical Trends
- 4.2.1.2. Current and Forecasted Estimates
- 4.2.2. Currency Coverage
- 4.2.2.1. Major Currencies Affecting the Market
- 4.2.2.2. Factors Affecting Currency Fluctuations
- 4.2.2.3. Impact of Currency Fluctuations on the Industry
- 4.2.3. Foreign Currency Exchange Rate
- 4.2.3.1. Impact of Foreign Exchange Rate Volatility on the Market
- 4.2.3.2. Strategies for Mitigating Foreign Exchange Risk
- 4.2.4. Recession
- 4.2.4.1. Assessment of Current Economic Conditions and Potential Impact on the Market
- 4.2.4.2. Historical Analysis of Past Recessions and Lessons Learnt
- 4.2.5. Inflation
- 4.2.5.1. Measurement and Analysis of Inflationary Pressures in the Economy
- 4.2.5.2. Potential Impact of Inflation on Market Evolution
- 4.2.6. Interest Rates
- 4.2.6.1. Interest Rates and Their Impact on the Market
- 4.2.6.2. Strategies for Managing Interest Rate Risk
- 4.2.7. Commodity Flow Analysis
- 4.2.7.1. Type of Commodity
- 4.2.7.2. Origins and Destinations
- 4.2.7.3. Values and Weights
- 4.2.7.4. Modes of Transportation
- 4.2.8. Global Trade Dynamics
- 4.2.8.1. Import Scenario
- 4.2.8.2. Export Scenario
- 4.2.8.3. Trade Policies
- 4.2.8.4. Strategies for Mitigating the Risks Associated with Trade Barriers
- 4.2.8.5. Impact of Trade Barriers on the Market
- 4.2.9. War Impact Analysis
- 4.2.9.1. Russian-Ukraine War
- 4.2.9.2. Israel-Hamas War
- 4.2.10. COVID Impact / Related Factors
- 4.2.10.1. Global Economic Impact
- 4.2.10.2. Industry-specific Impact
- 4.2.10.3. Government Response and Stimulus Measures
- 4.2.10.4. Future Outlook and Adaptation Strategies
- 4.2.11. Other Indicators
- 4.2.11.1. Fiscal Policy
- 4.2.11.2. Consumer Spending
- 4.2.11.3. Gross Domestic Product
- 4.2.11.4. Employment
- 4.2.11.5. Taxes
- 4.2.11.6. Stock Market Performance
- 4.2.11.7. Cross Border Dynamics
- 4.2.1. Time Period
- 4.3. Conclusion
5. EXECUTIVE SUMMARY
- 5.1. Executive Summary: Market Landscape
- 5.2. Executive Summary: Market Trends
- 5.3. Executive Summary: Market Forecast and Opportunity Analysis
6. INTRODUCTION
- 6.1. Chapter Overview
- 6.2. Concept of Microbiota and Microbiome
- 6.2.1. Discovery of Human Microbiome
- 6.2.2. Functions of Human Microbiome
- 6.3. Overview of Gut Flora
- 6.3.1. Role of Gut Flora in Human Health
- 6.3.2. Factors Affecting Gut Flora
- 6.3.2.1. Antibiotic Consumption
- 6.3.2.2. Age and Pregnancy
- 6.3.2.2.1. Mode of Childbirth
- 6.3.2.2.2. Type of Feeding
- 6.3.2.2.3. Antibiotic Consumption by Mother
- 6.3.2.3. Stress-related Factors
- 6.3.2.4. Dietary Factors
- 6.3.2.5. Impact of Lifestyle
- 6.4. Microbiome and Associated Diseases
- 6.4.1. Cancer
- 6.4.2. Inflammatory Bowel Disease (IBD)
- 6.4.3. Obesity
- 6.4.4. Parkinson's Disease
- 6.4.5. Type 2 Diabetes
- 6.4.6. Other Disease Indications
- 6.5. Impact of Microbiota on Drug Pharmacokinetics
- 6.6. Impact of Microbiota on Therapeutic Outcomes
- 6.7. Microbiome Therapeutics
- 6.7.1. Probiotics
- 6.7.1.1. Beneficial Bacterial Strains
- 6.7.1.1.1. Lactobacilli
- 6.7.1.1.2. Bifidobacteria
- 6.7.1.1.3. Others
- 6.7.1.2. Key Therapeutic Areas
- 6.7.1.2.1. Antibiotic-Associated Diarrhea (AAD)
- 6.7.1.2.2. Bacterial Vaginosis
- 6.7.1.2.3. High Blood Pressure
- 6.7.1.2.4. Hypercholesterolemia
- 6.7.1.2.5. Infectious Childhood Diarrhea (ICD)
- 6.7.1.2.6. Inflammatory Bowel Disease (IBD)
- 6.7.1.2.7. Lactose Intolerance
- 6.7.1.2.8. Vitamin Production
- 6.7.1.2.9. Weight Management
- 6.7.1.3. Side Effects of Probiotics
- 6.7.1.1. Beneficial Bacterial Strains
- 6.7.2. Prebiotics
- 6.7.2.1. Sources of Prebiotics
- 6.7.2.2. Types of Prebiotics
- 6.7.2.2.1. Fructo-Oligosaccharides (FOS)
- 6.7.2.2.2. Galacto-Oligosaccharides (GOS)
- 6.7.2.2.3. Inulin
- 6.7.2.3. Key Therapeutic Areas
- 6.7.2.3.1. Antibiotic Associated Diarrhea (AAD)
- 6.7.2.3.2. Constipation
- 6.7.2.3.3. Gastrointestinal Diseases
- 6.7.2.3.4. Dysbiosis
- 6.7.2.4. Side Effects of Prebiotics
- 6.7.1. Probiotics
- 6.8. The Human Microbiome Project (HMP)
- 6.8.1. Project Approach
- 6.8.2. Project Initiatives
- 6.8.3. Project Achievements
- 6.9. Regulatory Guidelines for Live Biotherapeutic Products (LBPs)
- 6.10. Key Challenges Associated with the Development of Microbiome Therapeutics
- 6.11. Future Perspectives
7. HUMAN MICROBIOME THERAPEUTICS: MARKET LANDSCAPE
- 7.1. Chapter Overview
- 7.2. Human Microbiome Therapeutics: Overall Market Landscape
- 7.2.1. Analysis by Stage of Development
- 7.2.2. Analysis by Type of Molecule
- 7.2.3. Analysis by Stage of Development and Type of Molecule
- 7.2.4. Analysis by Type of Biologic
- 7.2.5. Analysis by Target Indication
- 7.2.6. Analysis by Therapeutic Area
- 7.2.7. Analysis by Route of Administration
- 7.2.8. Analysis by Type of Formulation
- 7.2.9. Analysis by Dosage Frequency
- 7.2.10. Analysis by Type of Therapy
- 7.3. Human Microbiome Therapeutics: Developer Landscape
- 7.3.1. Analysis by Year of Establishment
- 7.3.2. Analysis by Company Size
- 7.3.3. Analysis by Location of Headquarters (Region)
- 7.3.4. Analysis by Location of Headquarters (Country)
- 7.3.5. Most Active Players: Analysis by Number of Microbiome Therapeutics
8. HUMAN MICROBIOME DIAGNOSTICS AND SCREENING / PROFILING TESTS: MARKET LANDSCAPE
- 8.1. Chapter Overview
- 8.2. Overview of Microbiome Diagnostics and Screening / Profiling Tests
- 8.3. Microbiome Diagnostics and Screening / Profiling Tests: Overall Market Landscape
- 8.3.1. Analysis by Stage of Development
- 8.3.2. Analysis by Type of Sample Analyzed
- 8.3.3. Analysis by Type of Screening Technique
- 8.3.4. Analysis by Target Indication
- 8.3.5. Analysis by Therapeutic Area
- 8.3.6. Analysis by Purpose of Test
- 8.4. Microbiome Diagnostic and Screening / Profiling Tests: Provider Landscape
- 8.4.1. Analysis by Year of Establishment
- 8.4.2. Analysis by Company Size
- 8.4.3. Analysis by Location of Headquarters (Region)
- 8.4.4. Analysis by Location of Headquarters (Country)
- 8.4.5. Most Active Players: Analysis by Number of Microbiome Tests
9. HUMAN MICROBIOME FECAL MICROBOTA THERAPIES: MARKET LANDSCAPE
- 9.1. Chapter Overview
- 9.2. Introduction to Fecal Microbiota Therapies (FMT)
- 9.3. Historical Overview
- 9.4. Fecal Microbiota Therapies: Procedure and Clinical Relevance
- 9.4.1. Donor Selection
- 9.4.2. Administration Procedure
- 9.4.3. Routes of Administration
- 9.4.4. Consequences and Adverse Events
- 9.4.5. Clinical Guidelines Associated with FMT
- 9.5. Regulatory Guidelines Related to Fecal Microbiota Therapies
- 9.6. Insurance Coverage for Fecal Microbiota Therapies
- 9.7. Fecal Microbiota Therapies: Overall Market Landscape
- 9.7.1. Marketed / Development Pipeline
- 9.7.1.1. Analysis by Application Area
- 9.7.1.2. Analysis by Status of Development
- 9.7.1.3. Analysis by Target Indication
- 9.7.1.4. Analysis by Therapeutic Area
- 9.7.1.5. Analysis by Route of Administration
- 9.7.2. Fecal Microbiota Therapies: Developer Landscape
- 9.7.2.1. Analysis by Year of Establishment
- 9.7.2.2. Analysis by Company Size
- 9.7.2.3. Analysis by Location of Headquarters (Region)
- 9.7.2.4. Analysis by Location of Headquarters (Country)
- 9.7.1. Marketed / Development Pipeline
10. HUMAN MICROBIOME THERAPEUTICS: COMPANY AND DRUG PROFILES
- 10.1. Chapter Overview
- 10.2. Ferring Pharmaceuticals
- 10.2.1. Company Overview
- 10.2.2. Financial Information
- 10.2.3. Microbiome-Based Product Portfolio
- 10.2.3.1. REBYOTA
- 10.2.3.1.1. Drug Overview
- 10.2.3.1.2. Current Status of Development
- 10.2.3.1.3. Clinical Studies
- 10.2.3.1. REBYOTA
- 10.2.4. Recent Developments and Future Outlook
- 10.3. Infant Bacterial Therapeutics
- 10.4. MaaT Pharma
- 10.5. Mikrobiomik
- 10.6. Seres Therapeutics
- 10.7. OxThera
- 10.8. Vedanta Biosciences
- 10.9. Zhiyi Biotech
11. MICROBIOME DIAGNOSTIC AND SCREENING / PROFILING TEST PROVIDERS: COMPANY PROFILES
- 11.1. Chapter Overview
- 11.2. Becton, Dickinson and Company
- 11.2.1. Company Overview
- 11.2.2. Financial Information
- 11.2.3. Microbiome Diagnostic Test Portfolio
- 11.2.4. Recent Developments and Future Outlook
- 11.3. Biome Diagnostics
- 11.4. GoodGut
- 11.5. NutriPATH
- 11.6. Tiny Health
12. CLINICAL TRIAL ANALYSIS
- 12.1. Chapter Overview
- 12.2. Scope and Methodology
- 12.3. Fecal Microbiota Therapies: Clinical Trial Analysis
- 12.3.1. Analysis by Trial Registration Year
- 12.3.2. Analysis of Enrolled Patient Population by Trial Registration Year
- 12.3.3. Analysis by Trial Status
- 12.3.4. Analysis of Enrolled Patient Population by Trial Status
- 12.3.5. Analysis by Trial Registration Year and Trial Status
- 12.3.6. Analysis by Trial Phase
- 12.3.7. Analysis of Enrolled Patient Population by Trial Phase
- 12.3.8. Analysis by Type of Sponsor / Collaborator
- 12.3.9. Analysis by Study Design
- 12.3.10. Analysis by Gender
- 12.3.11. Leading Industry Players: Analysis by Number of Registered Trials
- 12.3.12. Leading Non-industry Players: Analysis by Number of Registered Trials
- 12.3.13. Analysis by Trial Status, Trial Phase and Geography
- 12.3.14. Analysis by Geography
- 12.3.15. Analysis by Trial Status and Geography
- 12.3.16. Analysis of Enrolled Patient Population by Trial Status and Geography
13. ATTRACTIVENESS COMPETITIVENESS (AC) MATRIX
- 13.1. Chapter Overview
- 13.2. AC Matrix: An Overview
- 13.2.1. Strong Business Units
- 13.2.2. Average Business Units
- 13.2.3. Weak Business Units
- 13.3. AC Matrix: Analytical Methodology
- 13.4. AC Matrix: Plotting the Information
- 13.5. AC Matrix: Analyzing the Data
- 13.5.1. Strong Business Units
- 13.5.2. Average Business Units
- 13.5.3. Weak Business Units
- 13.6. Concluding Remarks
14. START-UP HEALTH INDEXING
- 14.1. Chapter Overview
- 14.2. Scope and Methodology
- 14.3. Benchmarking of Start-ups
- 14.3.1. Analysis by Portfolio Strength
- 14.3.2. Analysis by Pipeline Maturity
- 14.3.3. Analysis by Indication Diversity
- 14.3.4. Analysis by Funding Amount
- 14.3.5. Analysis by Partnership Activity
- 14.3.6. Start-up Health Indexing: Roots Analysis Perspective
- 14.3.7. Start-up Health Indexing: Leading Companies
15. CASE STUDY: KEY THERAPEUTIC AREAS
- 15.1. Chapter Overview
- 15.2. Metabolic Disorders
- 15.2.1. Diabetes
- 15.2.1.1. Disease Description
- 15.2.1.2. Associated Health Risks / Complications
- 15.2.1.3. Epidemiology
- 15.2.1.4. Disease Diagnosis
- 15.2.1.5. Current Treatment Options
- 15.2.1.5.1. Insulin Therapies
- 15.2.1.5.2. Non-Insulin Therapies
- 15.2.1.6. Side Effects of Current Treatment Options
- 15.2.1.7. Microbiome Therapeutics for Diabetes
- 15.2.2. Lactose Intolerance
- 15.2.2.1. Disease Description
- 15.2.2.2. Epidemiology
- 15.2.2.3. Current Treatment Options
- 15.2.2.4. Microbiome Therapeutics for Lactose Intolerance
- 15.2.3. Nonalcoholic Steatohepatitis (NASH)
- 15.2.3.1. Disease Description
- 15.2.3.2. Epidemiology
- 15.2.3.3. Current Treatment Options
- 15.2.3.4. Microbiome Therapeutics for NASH
- 15.2.4. Primary Hyperoxaluria
- 15.2.4.1. Disease Description
- 15.2.4.2. Epidemiology
- 15.2.4.3. Current Treatment Options
- 15.2.4.4. Microbiome Therapeutics for Primary Hyperoxaluria
- 15.2.5. Obesity
- 15.2.5.1. Disease Description
- 15.2.5.2. Epidemiology
- 15.2.5.3. Current Treatment Options
- 15.2.5.4. Side Effects of Current Treatment Options
- 15.2.5.5. Microbiome Therapeutics for Obesity
- 15.2.1. Diabetes
- 15.3. Digestive and Gastrointestinal Disorders
- 15.3.1. Crohn's Disease
- 15.3.1.1. Disease Description
- 15.3.1.2. Epidemiology
- 15.3.1.3. Current Treatment Options
- 15.3.1.4. Side Effects of Current Treatment Options
- 15.3.1.5. Microbiome Therapeutics for Crohn's Disease
- 15.3.2. Irritable Bowel Syndrome (IBS)
- 15.3.2.1. Disease Description
- 15.3.2.2. Epidemiology
- 15.3.2.3. Current Treatment Options
- 15.3.2.4. Microbiome Therapeutics for IBS
- 15.3.3. Ulcerative Colitis
- 15.3.3.1. Disease Description
- 15.3.3.2. Epidemiology
- 15.3.3.3. Current Treatment Options
- 15.3.3.4. Side Effects of Current Treatment Options
- 15.3.3.5. Microbiome Therapeutics for Ulcerative Colitis
- 15.3.1. Crohn's Disease
- 15.4. Oncological Indications
- 15.4.1. Colorectal Cancer
- 15.4.1.1. Disease Description
- 15.4.1.2. Epidemiology
- 15.4.1.3. Current Treatment Options
- 15.4.1.4. Side Effects of Current Treatments
- 15.4.1.5. Microbiome Therapeutics for Colorectal Cancer
- 15.4.2. Lung Cancer
- 15.4.2.1. Disease Description
- 15.4.2.2. Epidemiology
- 15.4.2.3. Current Treatment Options
- 15.4.2.4. Side Effects of Current Treatment Options
- 15.4.2.5. Microbiome Therapeutics for Lung Cancer
- 15.4.1. Colorectal Cancer
- 15.5. Dermatological Disorders
- 15.5.1. Acne Vulgaris
- 15.5.1.1. Disease Description
- 15.5.1.2. Epidemiology
- 15.5.1.3. Current Treatment Options
- 15.5.1.4. Side Effects of Current Treatment Options
- 15.5.1.5. Microbiome Therapeutics for Acne Vulgaris
- 15.5.1. Acne Vulgaris
- 15.6. Infectious Diseases
- 15.6.1. Clostridium Difficile Infections (CDIs)
- 15.6.1.1. Disease Description
- 15.6.1.2. Epidemiology
- 15.6.1.3. Disease Diagnosis
- 15.6.1.4. Current Treatment Options
- 15.6.1.5. Side Effects of Current Treatment Options
- 15.6.1.6. Microbiome Therapeutics for CDI
- 15.6.2. Bacterial Vaginosis
- 15.6.2.1. Disease Description
- 15.6.2.2. Epidemiology
- 15.6.2.3. Current Treatment Options
- 15.6.2.4. Side Effects of Current Treatment Options
- 15.6.2.5. Microbiome Therapeutics for Bacterial Vaginosis
- 15.6.1. Clostridium Difficile Infections (CDIs)
16. PARTNERSHIPS AND COLLABORATIONS
- 16.1. Chapter Overview
- 16.2. Partnership Models
- 16.3. Human Microbiome: List of Partnerships and Collaborations
- 16.4. Analysis by Year of Partnership
- 16.5. Analysis by Type of Partnership
- 16.6. Analysis by Year and Type of Partnership
- 16.7. Analysis by Target Indication
- 16.8. Analysis by Therapeutic Area
- 16.9. Analysis by Type of Partnership and Therapeutic Area
- 16.10. Analysis by Type of Partner
- 16.11. Most Active Players: Analysis by Number of Partnerships
- 16.12. Analysis by Geography
- 16.12.1. Local and International Deals
- 16.12.2. Intracontinental and Intercontinental Deals
17. FUNDING AND INVESTMENTS
- 17.1. Chapter Overview
- 17.2. Funding Models
- 17.3. Human Microbiome: List of Funding and Investments
- 17.3.1. Analysis of Funding Instances by Year of Funding
- 17.3.2. Analysis of Amount Invested by Year of Funding
- 17.3.3. Analysis by Type of Funding
- 17.3.4. Analysis by Year and Type of Funding
- 17.3.5. Analysis by Amount Invested and Type of Funding
- 17.3.6. Analysis by Purpose of Funding
- 17.3.7. Analysis by Target Indication
- 17.3.8. Analysis by Therapeutic Area
- 17.3.9. Analysis by Geography
- 17.3.10. Most Active Players: Analysis by Number of Instances
- 17.3.11. Most Active Players: Analysis by Amount Invested
- 17.3.12. Leading Investors: Analysis by Number of Instances
- 17.3.13. Funding and Investment Summary
18. CASE STUDY: CONTRACT SERVICES FOR MICROBIOME THERAPEUTICS AND LIVE BIOTHERAPEUTICS
- 18.1. Chapter Overview
- 18.2. Manufacturing Microbiome Therapeutics
- 18.2.1. Key Steps Involved
- 18.2.2. Associated Challenges
- 18.2.3. Growing Demand for Contract Manufacturing Services
- 18.2.4. Contract Manufacturing Organizations (CMOs)
- 18.2.4.1. Introduction to Contract Manufacturing
- 18.2.5. Microbiome Therapeutics: List of Contract Manufacturing Providers
- 18.2.5.1. Analysis by Year of Establishment
- 18.2.5.2. Analysis by Company Size
- 18.2.5.3. Analysis by Location of Headquarters
- 18.2.5.4. Analysis by Scale of Operation
- 18.2.5.5. Analysis by Type of Product Manufactured
- 18.2.5.6. Analysis by Type of Formulation
- 18.2.5.7. Analysis by Scale of Operation and Type of Formulation
- 18.3. Key Considerations for Selecting a CMO / CRO Partner
19. CASE STUDY: BIG DATA AND MICROBIOME THERAPEUTICS
- 19.1. Chapter Overview
- 19.2. Introduction to Big Data
- 19.3. Internet of Things (IoT)
- 19.4. Growing Interest in Big Data: Google Trends Analysis
- 19.5. Key Application Areas
- 19.6. Big Data in Microbiome Research
- 19.6.1. Microbiome Data and Personalized Medicine
- 19.6.2. Microbiome-related Data Management Challenges
- 19.6.3. National Microbiome Data Center
- 19.7. Big Data Services for Microbiome Research: List of Companies
- 19.8. Big Data Services for Microbiome Research: Profiles of Key Players
- 19.8.1. Human Longevity
- 19.8.1.1. Company Overview
- 19.8.1.2. Technology and Service Portfolio
- 19.8.1.3. Recent Developments and Future Outlook
- 19.8.2. Resilient Biotics
- 19.8.2.1. Company Overview
- 19.8.2.2. Technology and Service Portfolio
- 19.8.2.3. Recent Developments and Future Outlook
- 19.8.3. Resphera Biosciences
- 19.8.3.1. Company Overview
- 19.8.3.2. Technology and Service Portfolio
- 19.8.3.3. Recent Developments and Future Outlook
- 19.8.1. Human Longevity
20. HUMAN MICROBIOME THERAPEUTICS MARKET
- 20.1. Chapter Overview
- 20.2. Assumptions and Methodology
- 20.3. Global Human Microbiome Therapeutics Market: Forecasted Estimates (till 2035)
- 20.4. Human Microbiome Market: Distribution by Type of Product
- 20.4.1. Therapeutics: Forecasted Estimates (till 2035)
- 20.4.2. Diagnostics: Forecasted Estimates (till 2035)
- 20.4.3. Fecal Microbiota Therapies: Forecasted Estimates (till 2035)
- 20.4.4. Scenario Analysis
- 20.4.4.1. Conservative Scenario
- 20.4.4.2. Optimistic Scenario
- 20.5. Key Market Segmentations
21. HUMAN MICROBIOME THERAPEUTICS MARKET, BY TYPE OF BIOLOGIC
- 21.1. Chapter Overview
- 21.2. Assumptions and Methodology
- 21.3. Distribution by Type of Biologic
- 21.3.1. Live Biotherapeutics: Forecasted Estimates (till 2035)
- 21.3.2. Others: Forecasted Estimates (till 2035)
- 21.4. Data Triangulation and Validation
22. HUMAN MICROBIOME THERAPEUTICS MARKET, BY ROUTE OF ADMINISTRATION
- 22.1. Chapter Overview
- 22.2. Key Assumptions and Methodology
- 22.3. Distribution by Route of Administration
- 22.3.1. Oral Route: Forecasted Estimates (till 2035)
- 22.3.2. Rectal Route: Forecasted Estimates (till 2035)
- 22.4. Data Triangulation and Validation
23. HUMAN MICROBIOME THERAPEUTICS MARKET, BY DRUG FORMULATION
- 23.1. Chapter Overview
- 23.2. Key Assumptions and Methodology
- 23.3. Distribution by Drug Formulation
- 23.3.1. Capsules: Forecasted Estimates (till 2035)
- 23.3.2. Suspensions: Forecasted Estimates (till 2035)
- 23.3.3. Enemas: Forecasted Estimates (till 2035)
- 23.3.4. Powders: Forecasted Estimates (till 2035)
- 23.4. Data Triangulation and Validation
24. HUMAN MICROBIOME THERAPEUTICS MARKET, BY TARGET INDICATION
- 24.1. Chapter Overview
- 24.2. Key Assumptions and Methodology
- 24.3. Distribution by Target Indication
- 24.3.1. Clostridium Difficile Infection
- 24.3.2. Necrotizing Enterocolitis
- 24.3.3. Irritable Bowel Syndrome
- 24.3.4. Acute Graft Versus Host Disease
- 24.4. Data Triangulation and Validation
25. HUMAN MICROBIOME THERAPEUTICS MARKET, BY TARGET THERAPEUTIC AREA
- 25.1. Chapter Overview
- 25.2. Key Assumptions and Methodology
- 25.3. Distribution by Therapeutic Area
- 25.3.1. Infectious Diseases
- 25.3.2. Gastrointestinal Disorders
- 25.3.3. Rare Disorders
- 25.4. Data Triangulation and Validation
26. HUMAN MICROBIOME THERAPEUTICS MARKET, BY GEOGRAPHICAL REGIONS
- 26.1. Chapter Overview
- 26.2. Key Assumptions and Methodology
- 26.3. Distribution by Geographical Regions
- 26.3.1. North America
- 26.3.2. Europe
- 26.3.3. Asia-Pacific
- 26.3.4. Latin America
- 26.3.5. Middle East and North Africa
- 26.4. Market Dynamics Assessment
- 26.4.1. Penetration Growth (P-G) Matrix
- 26.4.2. Market Movement Analysis
- 26.5. Data Triangulation and Validation
27. HUMAN MICROBIOME DIAGNOSTICS MARKET
- 27.1. Chapter Overview
- 27.2. Assumptions and Methodology
- 27.3. Global Human Microbiome Diagnostics Market: Forecasted Estimates (till 2035)
- 27.3.1. Scenario Analysis
- 27.4. Key Market Segmentations
28. HUMAN MICROBIOME DIAGNOSTICS MARKET, BY TARGET INDICATION
- 28.1. Chapter Overview
- 28.2. Key Assumptions and Methodology
- 28.3. Market Distribution by Target Indication
- 28.3.1. Diabetes Mellitus: Forecasted Estimates (till 2035)
- 28.3.2. Irritable Bowel Syndrome: Forecasted Estimates (till 2035)
- 28.3.3. Irritable Bowel Disease: Forecasted Estimates (till 2035)
- 28.3.4. Colorectal Cancer: Forecasted Estimates (till 2035)
- 28.4. Data Triangulation and Validation
29. HUMAN MICROBIOME DIAGNOSTICS MARKET, BY TARGET THERAPEUTIC AREA
- 29.1. Chapter Overview
- 29.2. Key Assumptions and Methodology
- 29.3. Market Distribution by Therapeutic Area
- 29.3.1. Gastrointestinal Disorders: Forecasted Estimates (till 2035)
- 29.3.2. Metabolic Disorders: Forecasted Estimates (till 2035)
- 29.3.3. Oncological Disorders: Forecasted Estimates (till 2035)
- 29.4. Data Triangulation and Validation
30. HUMAN MICROBIOME DIAGNOSTICS MARKET, BY GEOGRAPHICAL REGIONS
- 30.1. Chapter Overview
- 30.2. Key Assumptions and Methodology
- 30.3. Market Distribution by Geographical Regions
- 30.3.1. North America: Forecasted Estimates (till 2035)
- 30.3.2. Europe: Forecasted Estimates (till 2035)
- 30.3.3. Asia-Pacific: Forecasted Estimates (till 2035)
- 30.3.4. Latin America: Forecasted Estimates (till 2035)
- 30.3.5. Middle East and North Africa: Forecasted Estimates (till 2035)
- 30.4. Data Triangulation and Validation
- 30.5. Market Dynamics Assessment
- 30.5.1. Penetration Growth (P-G) Matrix
- 30.5.2. Market Movement Analysis
- 30.6. Data Triangulation and Validation
31. HUMAN MICROBIOME FECAL MICROBIOTA THERAPIES MARKET
- 31.1. Chapter Overview
- 31.2. Assumptions and Methodology
- 31.3. Global Market: Forecasted Estimates (till 2035)
- 31.3.1. Scenario Analysis
- 31.3.1.1. Conservative Scenario
- 31.3.1.2. Optimistic Scenario
- 31.3.1. Scenario Analysis
- 31.4. Key Market Segmentations
32. HUMAN MICROBIOME FECAL MICROBIOTA THERAPIES MARKET, BY GEOGRAPHIC REGIONS
- 32.1. Chapter Overview
- 32.2. Key Assumptions and Methodology
- 32.3. Distribution by Geographic Regions
- 32.3.1. North America
- 32.3.2. Europe
- 32.3.3. Asia-Pacific
- 32.3.4. Latin America
- 32.3.5. Middle East and North Africa
- 32.4. Data Triangulation and Validation
- 32.5. Market Dynamics Assessment
- 32.5.1. Penetration Growth (P-G) Matrix
- 32.5.2. Market Movement Analysis
- 32.6. Data Triangulation and Validation
33. HUMAN MICROBIOME THERAPEUTICS MARKET OPPORTUNITY ANALYSIS: NORTH AMERICA
- 33.1. Market Distribution by Type of Biologic
- 33.2. Market Distribution by Route of Administration
- 33.3. Market Distribution by Drug Formulation
- 33.4. Market Distribution by Target Indication
- 33.5. Market Distribution by Target Therapeutic Area
34. HUMAN MICROBIOME THERAPEUTICS MARKET OPPORTUNITY ANALYSIS: EUROPE
- 34.1. Market Distribution by Type of Biologic
- 34.2. Market Distribution by Route of Administration
- 34.3. Market Distribution by Drug Formulation
- 34.4. Market Distribution by Target Indication
- 34.5. Market Distribution by Target Therapeutic Area
35. HUMAN MICROBIOME THERAPEUTICS MARKET OPPORTUNITY ANALYSIS: ASIA-PACIFIC
- 35.1. Market Distribution by Type of Biologic
- 35.2. Market Distribution by Route of Administration
- 35.3. Market Distribution by Drug Formulation
- 35.4. Market Distribution by Target Indication
- 35.5. Market Distribution by Target Therapeutic Area
36. HUMAN MICROBIOME THERAPEUTICS MARKET OPPORTUNITY ANALYSIS: LATIN AMERICA
- 36.1. Market Distribution by Type of Biologic
- 36.2. Market Distribution by Route of Administration
- 36.3. Market Distribution by Drug Formulation
- 36.4. Market Distribution by Target Indication
- 36.5. Market Distribution by Target Therapeutic Area
37. HUMAN MICROBIOME THERAPEUTICS MARKET OPPORTUNITY ANALYSIS: MIDDLE EAST AND NORTH AFRICA
- 37.1. Market Distribution by Type of Biologic
- 37.2. Market Distribution by Route of Administration
- 37.3. Market Distribution by Drug Formulation
- 37.4. Market Distribution by Target Indication
- 37.5. Market Distribution by Target Therapeutic Area
38. HUMAN MICROBIOME DIAGNOSTICS MARKET OPPORTUNITY ANALYSIS: NORTH AMERICA
- 38.1. Distribution by Target Indication
- 38.2. Distribution by Target Therapeutic Area
39. HUMAN MICROBIOME DIAGNOSTICS MARKET OPPORTUNITY ANALYSIS: EUROPE
- 39.1. Distribution by Target Indication
- 39.2. Distribution by Target Therapeutic Area
40. HUMAN MICROBIOME DIAGNOSTICS MARKET OPPORTUNITY ANALYSIS: ASIA-PACIFIC
- 40.1. Distribution by Target Indication
- 40.2. Distribution by Target Therapeutic Area
41. HUMAN MICROBIOME DIAGNOSTICS MARKET OPPORTUNITY ANALYSIS: LATIN AMERICA
- 41.1. Distribution by Target Indication
- 41.2. Distribution by Target Therapeutic Area
42. HUMAN MICROBIOME DIAGNOSTICS MARKET OPPORTUNITY ANALYSIS: MIDDLE EAST AND NORTH AFRICA
- 42.1. Distribution by Target Indication
- 42.2. Distribution by Target Therapeutic Area
43. CONCLUDING INSIGHTS
44. EXECUTIVE INSIGHTS
- 44.1. Chapter Overview
- 44.2. Company A (Mid-sized Company, UK)
- 44.2.1. Company Snapshot
- 44.2.2. Interview Transcript: Former Senior Manager, Corporate Development
- 44.3. Company B (Small Company, US)
- 44.3.1. Company Snapshot
- 44.3.2. Interview Transcript: Co-founder and Chairman
- 44.4. Company C (Small Company, US)
- 44.4.1. Company Snapshot
- 44.4.2. Interview Transcript: Chief Executive Officer
- 44.5. Company D (Large Company, US)
- 44.5.1. Company Snapshot
- 44.5.2. Interview Transcript: Senior Scientist
- 44.6. Company E (Small Company, Taiwan)
- 44.6.1. Company Snapshot
- 44.6.2. Interview Transcript: President
- 44.7. Company F (Small Company, US)
- 44.7.1. Company Snapshot
- 44.7.2. Interview Transcript: Vice President, Business Development
- 44.8. Company G (Mid-sized Company, Israel)
- 44.8.1. Company Snapshot
- 44.8.2. Interview Transcript: Chief Business Officer
- 44.9. Company H (Small Company, US)
- 44.9.1. Company Snapshot
- 44.9.2. Interview Transcript: Former Vice President, Sales and Business Development
- 44.10. Company I (Small Company, Belgium)
- 44.10.1. Company Snapshot
- 44.10.2. Interview Transcript: Co-founder and Chief Executive Officer
- 44.11. Company J (Small Company, US)
- 44.11.1. Company Snapshot
- 44.11.2. Interview Transcript: Co-founder and Chief Executive Officer
- 44.12. Company K (Small Company, US)
- 44.12.1. Company Snapshot
- 44.12.2. Interview Transcript: Co-founder and Chief Executive Officer
- 44.13. Company L (Mid-sized Company, US)
- 44.13.1. Company Snapshot
- 44.13.2. Interview Transcript: Ex-Co-founder and Vice President of Innovation
- 44.14. Company M (Small Company, US)
- 44.14.1. Company Snapshot
- 44.14.2. Interview Transcript: Vice President & Chief Operating Officer
- 44.15. Company N (Mid-sized Company, US)
- 44.15.1. Company Snapshot
- 44.15.2. Interview Transcript: Former Vice President, Operations
- 44.16. Company O (Small Company, US)
- 44.16.1. Company Snapshot
- 44.16.2. Interview Transcript: Former President and Chief Executive Officer
- 44.17. Company P (Small Company, US)
- 44.17.1. Company Snapshot
- 44.17.2. Interview Transcript: Former Chief Scientific Officer and Vice President, Research
- 44.18. Company Q (Small Company, France)
- 44.18.1. Company Snapshot
- 44.18.2. Interview Transcript: Former Chief Strategy Officer





